
Our proprietary CleanCap technology is the fastest and easiest way to produce a 5’ Cap 1 mRNA with 95% efficiency, offering a modern alternative to enzymatic capping. With GMP grade now available, CleanCap reagents are an immediate solution for swiftly scaling from clinical to commercial therapeutics production.
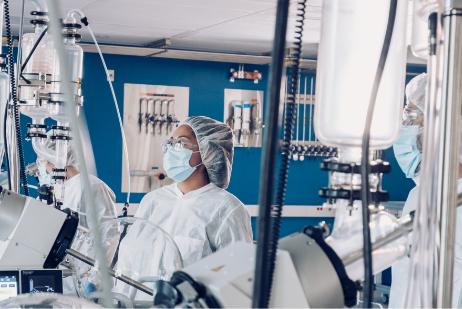
Our state-of-the-art Current Good Manufacturing Practice (cGMP) facility operates at the highest quality standards. Covering 118,000 sq. ft, the facility contains multiple specialized manufacturing suites and support labs, ensuring success across a wide variety of synthesis scales.
Dedicated ISO 8 or better manufacturing suite
Over 10 validated analytical methods per product
Multi-kg/month production scale
Certified ISO 9001:2015
We are dedicated to providing our customers with industry-leading products and services that enhance their mRNA therapeutics production.
With decades of experience, innovative technology, and a team committed to your success, we are the ideal partner to support you as you scale from clinical to commercial.

Trusted by Top Pharma & Biotech Companies
In 33 Therapeutic Programs Worldwide

Simplified Supply Chain Solutions

Dedicated Program Team & Day-to-Day Communication

25+ Years of Experience