Thrombopoiesis is a dynamic process which has varying physiologic responses in different clinical conditions. In newly diagnosed thrombocytopenic patients, or patients that have thrombocytopenia secondary to disease or drug therapy, it is crucial for a clinician to understand its underlying mechanism. Thrombocytopenia results when the bone marrow has slowed or stopped production of new platelets or it could be a destructive or consumption problem such as ITP. Care pathways differ in each case.
Immature Platelet Fraction
Immature platelet fraction (IPF) is an index of thrombopoiesis and can help determine the mechanism of thrombocytopenia. An increased IPF in the presence of thrombocytopenia is indicative of platelet destruction or consumption. A decreased/low normal IPF in the presence
of thrombocytopenia is indicative of decreased marrow production thereby getting to the pathogenesis of thrombocytopenia. Thus, IPF values serves as differentiator between increased consumption and decreased production.
Highest values of IPF are seen in DIC, AITP, TTP followed by patients with reconstituting marrows post chemo and bone marrow transplantation. IPF is a potential tool for monitoring thrombocytopenia in Dengue fever case and acts as a platelet transfusion regulator.
Most often an impaired production is due to bone marrow insufficiency. After chemotherapy, radiation therapy, stem cell or bone marrow transplantation, it takes a certain while until the bone marrow produces platelets at a sufficient rate again. Normally, the immature platelets count rises two to three days earlier than the total platelet concentration. Hence measurement of immature platelets fraction will be helpful to check whether the bone marrow's platelet production has started again. (See Fig) Possibly, this approach will help to save expensive platelet transfusions in the future.
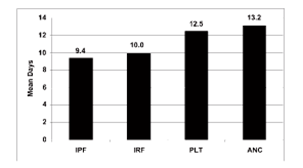
Fig 1: Zucker showed that IPF is an earlier indicator of platelet recovery at 9 days, and the difference is statistically significant when compared to IRF at 10 days, Platelet count at 12.5, and ANC at 13.2 days.
Immature Platelet Fraction Measurement on Sysmex XN Analyzer
A reportable parameter IPF can now be measured on the Sysmex XN /XN-L series as a part of Complete Blood Count. It has now become possible to monitor IPF in all cases of thrombocytopenia.
Transasia Bio-Medicals Ltd., India’s Largest In-vitro Diagnostic Company, is the sole distributor of the Sysmex hematology instruments in India.
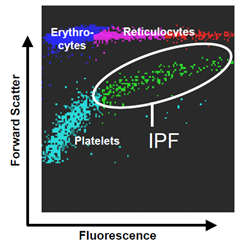
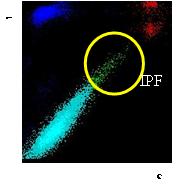
Fig 2 : IPF on XE / XN-L Analyzer Fig 3: IPF on XN Analyzer
Conclusion
Since destruction-mediated thrombocytopenic diseases are fairly rare, IPF is most useful in environments that service a large number of patients. Good examples include laboratories of large hospitals with haemato-oncologic units, paediatric units / neonatology units for differential diagnosis of juvenile thrombocytopenia and/or monitoring of the course of thrombocytopenia.
IPF is most useful in laboratories /hospitals with intensive care units, haemato-oncologic units, pediatric units / neonatology units for differential diagnosis of juvenile thrombocytopenia and/or monitoring of the course of thrombocytopenia in dengue cases.
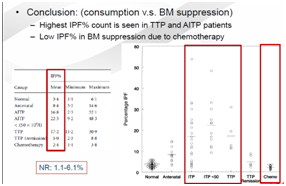
Fig 4: IPF levels in Consumptive v BM suppression
References:
Sysmex Website
Assessment of an immature platelet fraction (IPF) in peripheral thrombocytopenia.- Briggs C1, Kunka S, Hart D, Oguni S, Machin SJ.
Zucker, M. Laboratory Hematology, 2006 12:125 – 130