Every patient deserves to receive the treatment they need. In pulmonology, however, where it is becoming increasingly clear that diseases such as asthma and COPD have many phenotypes, it is difficult to prescribe individualised treat-ments. The gold standard for diagnosis, spirometry, is unable to differentiate between pulmonary disease phenotypes, but Functional Respiratory Imaging does have that ability. It is a technique that makes better follow-up of diagnostics and treatment selection possible.
‘COVID-19 has made the demand for innovative diagnostic techniques greater than ever.“We are having difficulty bringing the pandemic under control, partly because the tools we use to detect and treat the disease are not sufficiently responsive to the situation. Hence there is a substantial need for innovation in the respiratory field”, says Dr Jan De Backer, CEO of Fluidda. De Backer developed Functional Respiratory Imaging (FRI) in collaboration with his father, Dr Wilfried De Backer, Emeritus Professor of Respiratory Medicine at the University of Antwerp. FRI shows the anatomy of the airways and blood vessels in the lungs and the flow of air through them at millimetre-level resolution.
A radiologist will examine a CT scan of the lungs purely by eye and then describe it, with the result that only larger abnormalities and abnormal patterns are detected. ‘It is very difficult even for a practised eye to see small abnormalities or create a three-dimen-sional reconstruction of the lungs in the mind’s eye. With our technique we can do that’, continues Jan De Backer. ‘FRI is a quantitative technique that produces a 3D map of all the clinically relevant structures in the lungs, such as the alveoli, blood vessels and lung volume, using images from a standard CT scanner‘, says Jan De Backer. FRI even shows small abnormalities. The technique developed by the De Backers is able to show diffuse disease-related changes at very high resolution and monitor the changes over time. FRI thus enhances a radiologist‘s performance, and outperforms spirometry, which at present is still the gold standard for analysing lung function.
FRI analysis starts with two CT scans, the first after deep inspiration and the second after normal expiration. The patient uses a mouthpiece that records his or her breathing to enable the scans to be timed correctly. After that, the work of the hospital and the patient is done. The two scans are sent to Fluidda in the Belgian village of Kontich, only walking distance from the university hospital in Antwerp. Jan De Backer: ‘Our systems are based on machine learning and artificial intelligence. Every CT scan that we analyse improves our algorithms. We analyse and model the images using computational fluid dynamics, a flow simulation system that has its origins in the aerospace industry. That yields a three-dimensional image of the lungs, showing the flow of air and revealing areas of increased air resistance or places where inhaled particles have been deposited.
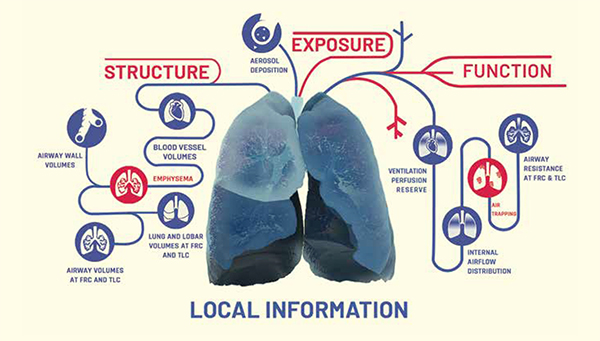
Jan De Backer gives an example of just how sensitive the technique is. ‘Pulmonary hypertension can be due to constriction of the smallest blood vessels in the lungs. This causes the blood to accumulate in the feeder blood vessels, which expand slightly. We can see the expansion even in blood vessels with a diameter of only one millimetre. That makes FRI a very patient-friendly option for diagnosing and monitoring pulmonary hypertension, which at present is done using invasive pressure measurement in the pulmonary artery.’ Using FRI, scientists have discovered that thrombosis in small blood vessels plays a major role in severe COVID-19. The finding made using FRI has now been confirmed by autopsies (see the interview with Muhunthan Thillai).
Like the field of Computational Fluid Dynamics (CFD), Jan de Backer also comes from a background in aerospace. De Backer first encountered CFD while studying aerospace engineering at Delft University of Technology in the Netherlands. It was his father, Wilfried De Backer, who then hit on the idea of using the technique to model the flow of air through the lungs. The big challenge for the Fluidda researchers was to adapt the computer technology to use lung data – after all, a lung is rather different from the smooth surface of an aircraft. Jan De Backer: ‘A lung – especially a diseased lung – has an irregular structure. You need to set the right boundary conditions to map flow in the lungs using computational fluid dynamics. We managed to obtain those boundary conditions directly from the CT scans, so that each measurement is individual.’ Wilfried De Backer adds: ‘FRI yields an accurate, personal representation of the anatomy of the airways and blood vessels in the lungs and the flow of air through them, so it is not based on a general model.’
The motivation for developing FRI was EUROSCOPE (European Respiratory Society Study on Chronic Obstructive Pulmonary Disease), a study conducted in the 1990s to explore the effects of inhaled corticosteroids on COPD. Wilfried De Backer: ‘In order to reach a worthwhile conclusion, we had to monitor 1,200 patients over three years. Although the study produced significant results, it gave rise to lengthy discussions among respiratory physicians on the value of inhaled corticosteroids for treating COPD. The EUROSCOPE study set me thinking about an alternative to spirometry. My son Jan then told me about computational fluid dynamics, and I thought we could obtain the requisite information from CT scans. We started developing FRI in 2005, and thanks to huge progress in computer technology and artificial intelligence we’ve managed to develop a technique that works well.’
At present, GPs and respiratory physicians diagnose lung diseases based on symptoms, blood tests, and usually spirometry. Jan De Backer: ‘The treatment for most pulmonary diseases is the same for a substantial majority of patients, who are given medication via an inhaler, for instance. Spirometry is then used to check whether their lung function has improved. Spirometry is the gold standard, but it only gives a general idea of lung function, whereas FRI gives a detailed, personalised picture.’ Pulmonologist Wilfried De Backer adds: ‘Take people with end-stage COPD, for example: they all have an FEV1 of 25 per cent, but they‘re not all the same. If you treat a patient based on that one FEV1 value, the treatment is not really targeted, as you have no information on the pathophysiology in the lungs. FRI provides pathophysiological information on such things as blood vessels, pulmonary hypertension and the sites of resistance in the distal alveoli.’
The FDA cleared Fluidda's device Broncholab, which is a diagnostic support tool. Jan De Backer: ‘We expect to receive European approval in the first quarter of 2021. It is not Fluidda’s aim to test all patients using FRI immediately. Most patients respond well to the treatment they receive from their GP or respiratory physician, and for them spirometry is adequate to analyse their lung function. We are focusing initially on patients whose treatment options have currently been exhausted: the question is whether the respiratory physician can find out, using FRI, whether there are reasons for the failure of their treatment. FRI also lends itself to predicting clinical outcomes. In COVID-19 trials , for instance, we find that vascular problems in the lungs detected using FRI are predictive of a poorer clinical outcome. The physician can then opt for more aggressive treatment, based on the FRI information.’
Wilfried De Backer goes on: ‘With FRI we can see sooner whether treatment goals are being met. At present it can take a year for treatment using biologics to produce an improvement in the FEV1 in patients with pulmonary disease. With FRI we can see after only one or two months whether there are improvements in geometry and/or function that spirometry does not detect. This has the advantage that we can review expensive treatments quickly and discontinue them if necessary, thus avoiding unnecessary adverse effects on patients and saving on the high cost of biologics.’
As for the cost savings from FRI, Jan De Backer says: ‘Cardiac catheterisation to diagnose pulmonary hypertension costs between five and twenty thousand dollars in the USA. An FRI scan costs only a few hundred dollars. COPD exacerbations in the USA cost over ten billion dollars a year. We know that a substantial proportion of those people are struggling with a treatable but undiagnosed vascular problem in the lungs. With FRI it can be diagnosed. If the treatment prevents 10% of exacerbations, that already yields a billion dollars in savings.’
FRI is proving its value not only in patient care but also in pharma-ceutical research. Wilfried De Backer’s personal experience again comes into play: ‘As a member of the European Medicines Agency, I noticed that a lot of products were failing to be approved because there was insufficient evidence of their efficacy based on spirometry, while there was indirect evident that they worked. There was huge frustration about the fact that we were missing a lot of innovations because there were no good quantitative endpoints for clinical trials. There was an intensive search going on for markers of that kind, for example biomarkers in the blood, but those markers suffer from huge variability. If you want to reach a clinically relevant conclusion about a particular patient, such variable markers will not do. FRI solves the problem, as we show the individual patient‘s anatomy as it really is. We have never seen any significant variability unrelated to the disease or the effect of medication in our studies. So FRI provides stable parameters for clinical trials.’
A recent example of the success of FRI in clinical research is from the Dutch respiratory physician Maarten van den Berge (see the interview with him). Wilfried De Backer: ‘The contrast between Van den Berge‘s study and EUROSCOPE could not be greater. Whereas EUROSCOPE needed three years and 1,200 patients to show a significant difference, Van den Berge achieved that in one month with just 23 patients.’ Jan De Backer: ‘The study looked at the difference between LABA/LAMA and LABA/LAMA plus inhaled corticosteroids for people with COPD. Thanks to FRI, Dr Van den Berge was able to show convincingly that adding inhaled corticosteroids works significantly better than treating them solely with LABA/LAMA. He also showed where in the lungs the drugs act and what differences there are between patients in which the treatment does and does not work well. Spirometry did not yield any significant results in that study, given the small number of patients involved.’
FRI was behind the development of smallparticle inhalers that send the particles deeper into the lungs. Jan De Backer: ‘Starting in 2005, we carried out ten to fifteen studies resulting in the approval of drugs using those small particles.’ Several completed and current Phase 1-4 clinical trials of drugs for pulmonary disease thus owe their success (or potential future success) to the Fluidda technique.
Researchers are increasingly embracing Functional Respiratory Imaging for clinical trials into new drugs for pulmonary disease. Broncholab is currently being used as a routine diagnostic support of patients with pulmonary diseases in American hospitals, and Europe will probably follow in 2021. As soon as doctors start making widespread use of FRI, the days of non-specific diagnosis using spirometry will be over. FRI is able to map each patient‘s disease at millimetre-level resolution and monitor the effects of treatment, thus making the prospect of a new scientific and diagnostic gold standard a real possibility.
‘It’s all coming together now’, concludes Jan De Backer. In recent years the radiation exposure from CT scans has decreased substantially, treatments have become more and more expensive and the possibilities of data analysis have improved to unprece-dented levels. Furthermore, a pandemic is showing the world that we need innovations in pulmonary medicine. The introduction of FRI as a diagnostic technique has come at just the right time.