Advanced Drug Delivery Systems for Chronic Liver Disease Management
Jyothi U. Menon, Associate Professor, College of Pharmacy at University of Rhode Island
Chronic liver disease (CLD) has emerged as major health issue globally, leading to millions of diagnoses and deaths from complications each year. The unmet need for safe and effective therapies has led to the exploration of advanced drug delivery systems as a possible alternative, for targeted and localised treatment.
Introduction
In the past two decades, chronic liver disease (CLD) has become increasingly prevalent across various age groups. Although the elderly were considered to be at high risk earlier, the increasing rates of obesity, diabetes, hepatitis and excessive alcohol consumption have contributed to increasing CLD occurrences among other age groups as well.
The primary function of the liver is to filter the blood and break down/ metabolise substances, including toxins, impurities, drugs, dietary amino acids, lipids, vitamins and carbohydrates in the body. Consequently, liver disease and inflammation are closely linked to metabolic disorders. Metabolic dysfunction-associated steatotic liver disease (MASLD), previously known as nonalcoholic fatty liver disease (NAFLD), results from fat buildup in the liver due to conditions like obesity, type-2 diabetes and insulin resistance; it is estimated to affect about 30% of the adult population worldwide [1]. Alcoholic liver disease (ALD) occurs due to chronic alcohol consumption and prolonged exposure of the liver and gut cells to alcohol. Viral hepatitis occurs due to infection from hepatitis viruses while drug-induced liver injury (DILI) is caused by accumulation of chemotherapeutic drugs, antibiotics and other toxic medications in the liver over time. Autoimmune hepatitis is another condition where the patient’s own immune system attacks liver cells leading to chronic inflammation and potential liver damage. The liver inflammation observed across all of these above conditions, if not managed adequately, can progress to chronic liver fibrosis, cirrhosis and end-stage liver disease. In some cases, these conditions can eventually lead to hepatocellular carcinoma (HCC) – the fastest growing cause of cancer-related deaths both in the United States and worldwide.
Due to the widespread prevalence of CLDs and the limitations of current therapies as described below, alternative strategies such as advanced drug delivery systems for targeted and localised treatment are now being explored. This article will focus on some of the considerations for development of advanced drug delivery systems-based therapeutic approaches in CLD treatment and the future outlook of this critical research area.
Challenges in the Treatment of CLDs
CLD diagnosis often occurs at the later stages of the disease due to its asymptomatic nature in the early stages, overlap of symptoms with other disorders and variability in presentation. While treatment options for viral hepatitis typically include antiviral medications, and lifestyle modifications are recommended for MAFLD, there are limited or no treatments available currently for other CLDs [2]. Once the disease progresses to advanced fibrosis and cirrhosis, liver transplantation is the only therapeutic option available, but this is severely constrained by the organ shortage crisis and therefore, the lack of suitable donor livers for transplantation. Off-target side effects of some of the treatments pose an additional challenge. Furthermore, due to the high variability in CLD presentation in different individuals, there is an unmet need for personalized treatment approaches that can address the variable manifestations of CLDs in each patient. To treat CLDs and prevent further complications, it is essential to develop alternative approaches that can provide targeted, effective and rigorous treatment.
Key Biomarkers for Therapy
In order to provide targeted treatment to inflamed and fibrotic liver cells, the key biomarkers expressed in these cells need to be identified. This is an area that has received considerable attention in the past decade, particularly as CLD cases began to increase. Hepatocytes constitute about 70-80% of the cells of the liver and are therefore the primary cells that are exposed to alcohol and other hepatotoxic agents arriving in the liver. Therefore, incorporating ligands that can specifically recognise and bind to markers upregulated in damaged hepatocytes will enable us to deliver therapies in a site-specific manner. Hepatocyte-specific targets that have been explored so far include the asialoglycoprotein receptor, farnesoid-X receptor (FXR), Organic Anion Transporting Polypeptides (OATP) and Solute Carrier Family 39 Member 14 (SLC39A14) [3,4]. Stressed and dying hepatocytes release danger-associated molecular patterns (DAMPs) as well as other protein biomarkers into the blood including transaminases alanine aminotransferase (ALT) and aspartate aminotransferase (AST); elevated levels of the latter two biomarkers are indicators of liver disease. The DAMPs are recognised by other cells in the liver, including the Kupffer cells and Hepatic Stellate cells, both of which play a key role in the initiation and progression of liver inflammation and fibrosis.
Kupffer cells, which are resident liver macrophages, constitute about 15% of the cells in the liver. The activation of Kupffer cells by DAMPs triggers the release of proinflammatory cytokines to recruit more immune cells to the region to repair liver damage (Fig. 1). However, persistent damage to the liver leads to prolonged release of proinflammatory cytokines by these cells, leading to greater tissue damage, inflammation and fibrosis. Targeting the Kupffer cells to “switch off” the proinflammatory cytokine release and promote anti-inflammatory phenotype is one possible method to prevent disease progression. Commonly targeted markers on Kupffer cells for drug delivery include the mannose receptor, the G-protein coupled bile acid receptor (Gpbar1) and some toll-like receptors (TLRs) [5]. Some of the proinflammatory cytokines that are released, such as platelet-derived growth factor (PDGF) and transforming growth factor-β (TGF- β), can also promote hepatic stellate cell activation, which leads to initiation of fibrinogenesis [6].
Activated hepatic stellate cells are of great interest in liver-specific drug development due to their role in initiating liver fibrosis and secreting excess collagen and extracellular matrix proteins. In addition to the proinflammatory cytokines released by Kupffer cells, hepatic stellate cells can also detect DAMPs released by hepatocytes, which leads to their activation. CD34 and CD44 are transmembrane phosphoglycoproteins frequently used to target HSCs for delivering therapies [7]. Mannose receptors have also been used but their expression on Kupffer cells as well reduces their specificity. Upregulation of platelet-derived growth factor receptor alpha (PDGFRα) is commonly seen in HSCs in cirrhotic livers and may also be used as a possible target. Targeting other cells such as the liver sinusoidal endothelial cells have also been explored, albeit to a lesser extent, than the hepatocytes, Kupffer cells and hepatic stellate cells. In addition, components of the extracellular matrix such as fibrillin-1 – a large protein of the extracellular matrix upregulated in stressed collagen matrices, may also be targeted for drug delivery [8].
Advanced Drug Delivery Systems and their Role in CLD Treatment
Advanced drug delivery systems such as nanoparticles are being developed and investigated to improve the efficacy of therapies and target them towards specific cell types in the liver. The nanoparticle formulations can be prepared using lipids (e.g. micelles, liposomes), polymers (e.g. polyesters, polysaccharides), inorganic materials (e.g. iron, gold, mesoporous silica) or hybrids of these materials. A diameter of about 200 nm is preferred for liver targeting. The nanoparticles thus prepared can be used to encapsulate and deliver anti-fibrotic or anti-inflammatory therapies. Hybrid nanoformulations such as lipid-polymer core-shell formulations can be used to encapsulate multiple therapies to be delivered at varying rates from the different layers for an additive or synergistic effect. A substantial portion (~90%) of systemically delivered nanoparticles have been reported to reach the liver. However, to target the drug-loaded nanoparticles to the intended cell type or site of action in the liver and maximise their therapeutic effects, surface modification of the particles may be done. More recently, biomimetic nanoparticle formulations coated with cell membranes have also been synthesized and investigated for treatment of CLDs. The presence of the cell membrane coating can potentially keep the particles in circulation longer and prevent their rapid clearance. For example, all-trans retinoic acid (ATRA)-containing cell membrane-coated polymer nanoparticles surface conjugated with the tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) have been studied to deplete fibrosis-causing hepatic stellate cells [9]. The ligand successfully caused apoptosis of the cells while ATRA induced hepatic stellate cell quiescence, demonstrating the potential of this formulation for targeted anti-fibrotic therapy. Cell membrane-coated formulations have also been developed and studied successfully for the treatment of fatty liver disease [10]. In the future, patients diagnosed with CLDs may be evaluated for patient-specific markers, which can then be targeted using surface modifications on drug-loaded nanoparticles to enable precision therapy.
Conclusions and Future Outlook
Advanced drug delivery systems hold immense potential for delivering therapies to the site of interest, while minimising the possibility of toxic side-effects. These systems are versatile and can be used to encapsulate multiple therapies and surface-modified to target specific cell types, overcome liver tissue-related barriers, and improve their residence time and prevent immune recognition. This versatility of the advanced drug delivery formulations can be harnessed to develop improved therapies against specific cell types that are key players in CLDs, dramatically improving patient outcomes and paving the way for the development of personalized therapies.
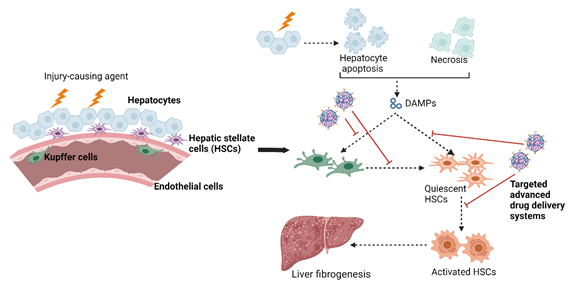
Figure 1: Schematic representation of the potential of advanced drug delivery systems to target different liver cell types for therapy.
This figure was created using BioRender.com
References
[1] Liu J, Ayada I, Zhang X, Wang L, Li Y, Wen T, Ma Z, Bruno MJ, de Knegt RJ, Cao W. Estimating global prevalence of metabolic dysfunction-associated fatty liver disease in overweight or obese adults. (2022), 20(3), pp.e573-e582.
[2] Xu J, Yu Y, Xu X. Management of chronic liver diseases and cirrhosis: current status and future directions. Chin Med J (2020), 133(22), pp.2647-2649.
[3] Zhang H, Guo Y, Jiao J, Qiu Y, Miao Y, He Y, Li Z, Xia C, Li L, Cai J. A hepatocyte-targeting nanoparticle for enhanced hepatobiliary magnetic resonance imaging. (2023), 7(3), pp.221-235.
[4] Porterfield JE, Sharma R, Jimenez AS, Sah N, McCracken S, Zhang L, An H, Lee S, Kannan S, Sharma A. Galactosylated hydroxyl‐polyamidoamine dendrimer targets hepatocytes and improves therapeutic outcomes in a severe model of acetaminophen poisoning‐induced liver failure. (2023), 8(3), pp.e10486.
[5] Unagolla J, Das S, Flanagan R, Oehler M, Menon JU. Targeting chronic liver diseases: Molecular markers, drug delivery strategies and future perspectives. Int J Pharm (2024), pp.124381.
[6] Matsuda M, Seki E. Hepatic stellate cell–macrophage crosstalk in liver fibrosis and carcinogenesis. (2020). ,40 (03),307-320.
[7] Ezhilarasan D. Hepatic stellate cells in the injured liver: Perspectives beyond hepatic fibrosis. J Cell Physiol (2022), 237(1), pp.436-449.
[8] Dubuisson L, Lepreux S, Bioulac-Sage P, Balabaud C, Costa AMA, Rosenbaum J, Desmoulière A. Expression and cellular localization of fibrillin-1 in normal and pathological human liver. J Hepatol (2001), 34(4), pp.514-522.
[9] Xia S, Liu Z, Cai J, Ren H, Li Q, Zhang H, Yue J, Zhou Q, Zhou T, Wang L. Liver fibrosis therapy based on biomimetic nanoparticles which deplete activated hepatic stellate cells. J Controlled Release (2023), 355, pp.54-67.
[10] Zahid AA, Chakraborty A, Shamiya Y, Wilson RB, Borradaile N, Paul A. Cell Membrane-Derived Nanoparticles as Biomimetic Nanotherapeutics to Alleviate Fatty Liver Disease. (2024).














