Application of Artificial Intelligence in Development Safety Update Report Preparation
Maria Carolina Braga, Senior PV Reporting Associate, ICON plc
Artificial intelligence is increasingly integrated into pharmacovigilance to optimise data analysis and streamline aggregate report generation. A study mapped DSUR processes, reviewed regulations, and analysed available AI tools. This article presents selected findings and aims to initiate discussion on AI’s role in improving aggregate safety report generation.
Pharmacovigilance (PV) is the science responsible for monitoring safety of diverse products (drugs, cosmetics, devices, etc). As part of the PV workflow there are the following steps: identification, collection, processing and reporting of adverse events. Naturally, this is a very brief resume of how PV workflow works; however, it is possible to translate the entire workflow with basically two words: data analysis.
PV professionals, at different levels and through different methods, are responsible for performing data analysis of safety information of medicinal products. To do that, it is necessary to collect large amounts of information, from early stages of drug development to continuous monitoring after marketing approval until the product is withdrawn from the market.
Based on World Health Organization Uppsala Monitoring Centre 2021-2022 Annual Report1: VigiBase received 31,326,511 ICSRs by 30 June 2022. The report from 20232 provided an updated number, with the database reaching 36 million reports by 31 Dec 2023 and nearly 40 million reports by 31 Dec 2024 (WHO-UMC 2024 Report)3. The increase in the number of reports became a trend that is also possible to notice within other databases, such as FAERs (US-FDA). With the number of reports increasing each year, it is humanly impossible to analyse all this information.
One of the methods employed for analysing safety information of medicinal products is through aggregate reports, regulatory documents that summarize and evaluate the cumulative safety information of medicinal products over a defined period. One of them is the Development Safety Update Report (DSUR), an annual report that provides a comprehensive summary of the safety profile and clinical data for drugs under development. It is used for:
- Communicating/Assessing benefit-risk profile updates during drug development
- Make informed decisions on the continuation, modification or termination of a trial
- Ensure trial participants' safety.
While the aggregate report used for approved drugs is the Periodic Benefit-Risk Evaluation Report (PBRER). Currently, two entities provide guidelines for writing these documents: International Council Harmonisation (ICH) and Council for International Organizations of Medical Sciences (CIOMS).
Seeking alternatives that allow this data analysis in an optimised way, Artificial Intelligence (AI) has been studied as an optional tool to help PV professionals in this task. Although it seems to be like a novelty, AI was first developed in the 1950’s as a branch of computer science that enables machines to emulate human behavior and handle tasks that typically require human intelligence. Over the years, it has been evolving, and among its various applications, AI is being studied to be applied in PV.
Applications for AI in Pharmacovigilance
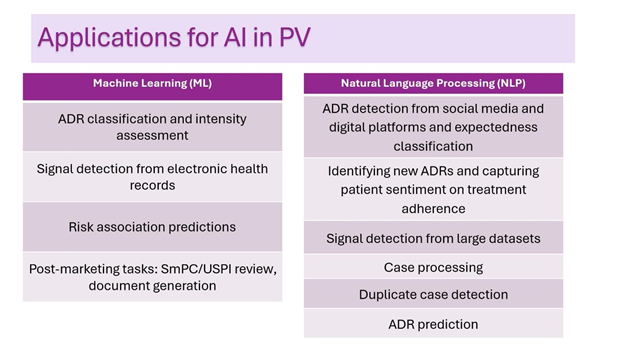
AI has a promising future in PV in activities such as data extraction, case processing, signal detection, and even predictive modeling, projecting potential safety issues based on historical data. There are various subfields of AI with distinct applications; in the context of PV, two relevant types include machine learning (ML) and natural language processing (NLP).
Machine learning can be applied in PV for optimisation of processes, as it can be used for analyzing structured data and making predictions. In PV, ML is being used for different things, such as classification of adverse drug reaction intensity and identification of adverse drug reactions. ML can also be applied during the post-marketing phase for review of SmPC and USPI, as well as for generating updated documents.
Natural language processing uses machine learning to enable computers to understand and communicate with human language; it can identify patterns, trends, and sentiments from large datasets and learn from it, by adapting its text generation when used for communication. It can also extract and summarize structured and unstructured data and perform categorisation of vast amounts of text, being applied for in the whole workflow of case processing: performing identification of adverse events in different datasets (EHRs, social media), and once an adverse drug event is identified, performing the case intake, triage, and initial assessment (classifying case seriousness and expectedness); data entry into the preferable PV system; medical assessment, including causality; narrative writing; and coding the adverse event (AE) into standardized terminology. identifying ADR reported via social media and other digital platforms. Beyond ADR detection in social media, NLP is also being employed for:
- Signal detection
- Adverse drug reaction prediction
- Adverse drug reaction identification from social media
- Duplicate case detection
It is important to highlight that even if AI can perform the tasks listed above, it does not replace the PV professional. For the current systems available on the market and the programs and algorithms described in the literature, human oversight is always required. The human factor remains essential to ensure contextual interpretation, regulatory compliance, and the validation of AI-generated outputs.
AI in Aggregate Report generation

The study focused on DSUR to streamline the analysis on what is required to write the document. CIOMS and ICH guidelines have a well-structured orientation on what information is needed to prepare the document. Both guidelines require the inclusion of aggregated clinical information, commonly presented as line listings and summary tabulations. Since clinical trial and safety data are often collected through diverse systems and platforms, the application of artificial intelligence can facilitate the automated extraction, standardization, and organisation of these data, thereby improving the efficiency and consistency of report preparation.
AI tools developed for aggregate report generation could incorporate country-specific templates to facilitate document preparation. Use of NLP for this task is promising, as this technology can extract and synthesize information from both structured and unstructured datasets. This is also important considering that not all countries adopted the ICH or CIOMS format; Mexico, for example, has its own report format (Report Periodico de Seguridad - RPS), although it is very similar to the CIOMS report, it is prepared in a different time period. With AI it is possible to optimise this report preparation, as it has overlapping content in relation to the DSUR sent to other authorities.
One of the main advantages of applying AI to aggregate report generation is the substantial reduction in preparation time. Traditionally, DSUR development may require 4 to 6 weeks, depending on data complexity and the number of review cycles, which is challenging given the typical 60-day submission deadline from the DLP. With AI-assisted tools, DSUR preparation can be shortened to approximately 3 to 7 days, allowing pharmacovigilance teams sufficient time to conduct thorough reviews and ensure timely submission to regulatory authorities.
AI implementation enables PV professionals to focus better on high-level tasks instead of routine ones that are mostly time-consuming but mechanical. Which promotes a more efficient workflow and improves adherence to reporting timelines.
Conclusion
A key advantage of AI implementation is the automation of critical yet repetitive tasks, such as data extraction, formatting, version control, and template alignment. This not only accelerates the preparation process but also ensures greater uniformity and quality across submissions. Automation is especially beneficial for pharmaceutical companies expanding their pharmacovigilance operations across multiple markets, where diverse reporting formats, submission timelines, and local expectations create complexity.
However, while AI can efficiently manage technical and repetitive components of report development, human oversight remains indispensable. PV professionals provide the scientific expertise necessary to interpret clinical context, assess benefit–risk profiles, and make informed safety decisions that AI systems cannot independently achieve. Their judgment ensures that AI-generated outputs are accurate, meaningful, and aligned with regulatory expectations.
Ultimately, the integration of AI should be viewed not as a replacement for human expertise, but as a tool that augments it: enabling PV professionals to focus on higher-level scientific evaluation while maintaining the rigor and accountability required for safeguarding patient safety.
Footnotes:
- https://who-umc.org/media/cgnlrs5v/umc-annual-report-2021-22.pdf
- https://who-umc.org/media/yhunvrcz/annual-report-2023.pdf
- https://view.publitas.com/uppsala-monitoring-centre/annual-report-2024/page/8-9












