Beyond the Pill: Expanding the Definition of Treatment
Ryan Varghese Mathew, Philadelphia College of Pharmacy, Saint Joseph's University
The article elucidates a paradigm shift from conventional pharmacological interventions to a holistic, patient-centric healthcare model. It unveils a future where prevention takes center stage, personalized care becomes the norm, and digital innovations redefine what’s possible. With the advent of wearable biosensors for real-time health monitoring, state-of-the-art precision medicine, and patient empowerment initiatives, healthcare is evolving from a reactive to a proactive model. As the rise of value-based care challenges old paradigms, we’re witnessing a bold new era of treatments that are not only more effective but also more accessible, putting patients at the heart of their healing journeys like never before.
In an era of rapid medical advancements, we often find ourselves tethered to the simplicity of banking on a pill for a quick fix. While pharmaceuticals have undeniably transformed our capacity to treat and manage ailments, the concept of healthcare transcends the conventional boundary of prescriptions and treatments. For it is rightly said, "True health is not just the mere absence of disease, but a unison of the harmony of the mind, body, and spirit". As we stand at the threshold of a new post-COVID era in medicine, the focus has metamorphosized from a symptomatic treatment to a more holistic treatment approach. This integrative perspective acknowledges the byzantine interplay between psychological, physiological, and metaphysical dimensions, which emphasizes the uniqueness of each individual's health journey.
This evolving healthcare landscape demands a recalibration of our existing notion of well-being, from reactive care to proactive holistic approaches - where disease prevention; mental health and well-being; accessible and affordable healthcare; and personalized care provision take the center stage. This approach has catalyzed developments that empower personalized care and early diagnosis, such as digital health innovations, telemedicine, and AI-driven diagnostics. Some recent breakthroughs have been discussed herewith, and is illustrated in Fig.1 and Fig. 2.
Fig 1: Expanding Pharmaceutical Treatment beyond the conventional pill-centric approaches
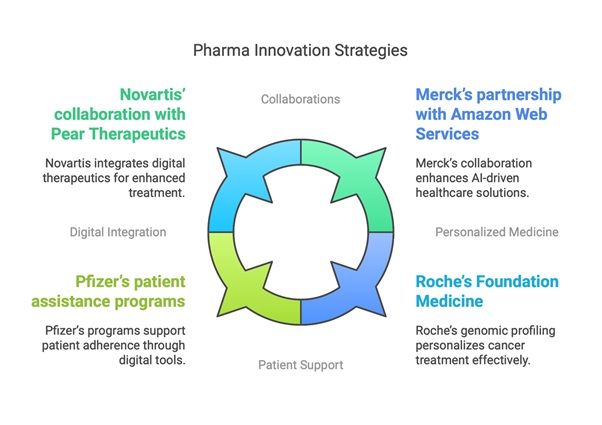
Fig 2: Contemporary Examples to Comprehensive Patient-Centered Care
1. Fostering Compassionate Care: The Patient-Centric Paradigm
Since the early 2000s, several authoritative bodies, including the National Academy of Medicine (NAM) (formerly Institute of Medicine (IoM)), have accentuated the paramount significance of patient-centric approaches in ameliorating healthcare outcomes and satisfaction. Principally, patient-centric approaches focus on orchestrating systems of healthcare delivery around the dynamic needs and predilections of each individual patient.
The amalgamation of key components such as interdisciplinary teams, integrated health systems, electronic health records (EHRs), and care managers, facilitates the mitigation of errors and confers enhanced satisfaction. Moreover, patient engagements plays a pivotal role, often buttressed by educational programs, digital tools, and continuous feedback systems, thereby augmenting care quality, minimizing associated costs, and fostering a more efficacious healthcare system.
A notable example is the Changing Diabetes in Children® (CDiC) program [1], which stands as a paragon of corporate social responsibility, meticulously designed to ameliorate the lives of juveniles and adolescents afflicted with Type 1 Diabetes in low- and middle-income countries. This multifaceted initiative, spearheaded by a consortium of esteemed organizations including Novo Nordisk, Roche, the World Diabetes Federation (WDF), and the International Society for Pediatric and Adolescent Diabetes (ISPAD), has set an ambitious target of extending its beneficence to 100,000 children by the year 2030. The program facilitates access to vital insulin, disseminates educational resources and interactive webinars for families, implements digital solutions for caregivers, and conducts professional development for healthcare practitioners. This multifaceted approach ensures that pediatric diabetes patients receive optimal care and requisite resources to efficiently manage their condition.
2. Synergizing Digital Health: A Nexus of Innovation
With the advent of technology, digitization has become an integral part of healthcare, precipitating a paradigm shift in the delivery of medical services. This digital transformation has engendered increasing acceptance of and interest in digital health solutions. This encompasses a broad spectrum of telemedicine platforms, mobile applications, and wearable devices that revolutionize the healthcare industry. Digital health solutions play a pivotal role in advancing healthcare delivery through continuous monitoring, personalized care, and improved patient engagement. For example, wearable devices such as smartwatch-integrated heart rate and blood pressure trackers or continuous blood glucose monitors (CGMs) exemplify this paradigm shift. These instruments continuously track, store, and analyze real-time health metrics, empowering healthcare providers to proactively tailor their treatment plans. This continuous feedback loop mitigates the complications associated with overdosage and optimizes therapeutic outcomes in stark contrast to the conventional “one-size-fits-all” approach.
Furthermore, in recent years, there has been an upsurge in prescription digital therapeutics (PDTs), which are FDA-approved software-based interventions designed to manage, treat, or prevent medical conditions. These digital modalities, prescribed by healthcare providers, function as adjuncts or alternatives to traditional treatment. PDTs harness evidence-based algorithms, robust datasets, and cutting-edge technologies to confer tailored care, thereby improving patient outcomes and augmenting treatment adherence. While superficially analogous to mobile health applications, PDTs are distinguished by their mandatory regulatory approval, which guarantees clinical safety and efficacy. For instance, Novartis’ Sandoz division has recently forged a strategic alliance with Pear Therapeutics to develop and commercialize PDTs, with reSET as their vanguard initiative [2]. reSET, an FDA-approved PDT for substance use disorders, is designed to be utilized over a 12-week period as an adjunct to other clinician-directed behavioral interventions. With this innovative platform, reSET aims to deliver cognitive behavioral therapy (CBT) via a smartphone interface, leveraging digital technology to enhance treatment accessibility. Clinical trials have demonstrated that reSET, compared with conventional therapy, elicited an approximately 40% higher abstinence rate and significantly mitigated relapse [3]. This synergistic collaboration facilitates Novartis in integrating PDTs into their existing portfolio, ensuring a confluence of digital and pharmaceutical interventions, augmenting medication adherence, ameliorating patient outcomes, and optimizing cost-to-value ratios. Furthermore, this initiative underscores the potential of collaborative regulatory efforts, closed-loop data sharing on patient outcomes, and perpetual post-marketing surveillance to ensure long-term therapeutic efficacy.
3. The Dawn of Personalized Medicine
Over the decades, personalized medicine (PM) has emerged as a paradigm-shifting approach in healthcare that transcends the conventional notion of the “one-size-fits-all” approach. PM leverages genetic and molecular data to tailor therapeutic interventions for individual patients to optimize treatment efficacy while mitigating adverse effects. By leveraging advanced genomic sequencing and diagnostic tools, PM-based algorithms can identify biomarkers that predict patient treatment responses and outcomes, enabling the tailoring of therapies to each patient’s unique genetic profile.
PM offers hope for more efficacious targeted therapies with higher precision and improved patient outcome. This approach is poised to revolutionize the delivery of healthcare across various domains, including oncology, orphan diseases, and chronic ailments. The implementation of PM represents a significant advancement in medical practice, promising for use in an era of more effective personalized healthcare solutions. Roche’s FoundationOne® epitomizes the pinnacle of PM in oncology, catalyzing a paradigm shift in cancer therapeutics [4]. FoundationOne® offers a comprehensive genomic profiling (CGP) test that elucidates the DNA landscape of an individual’s tumour. CGP facilitates the identification of salient genetic mutations and alterations across 324 genes implicated in oncogenesis and tumor progression. By delineating mutations such as ALK, BRAF, and EGFR, the FoundationOne® CGP provides clinicians with pivotal insights into the genetic drivers of cancer, which guides the judicious implementation of PM delivery. A seminal study published in the Journal of the American Medical Association (JAMA) Oncology [5] reported that 51% of patients receiving genomically guided therapy demonstrated profound clinical benefit, juxtaposed with 29% receiving standard treatment. With over a million tests performed globally, FoundationOne® has achieved widespread acceptance and clinical integration, serving as a testament to inexorable progress in precision oncology. This extensive adoption ensures that patients with cancer receive efficacious therapeutic interventions that are optimally tailored to their unique genomic profiles.
4. The Genesis of Patient Support Programs (PSPs)
Patient Support Programs (PSPs) are multifaceted initiatives within the pharmaceutical industry that are designed to help patients access medications, mitigate treatment costs, and improve patient outcomes. These comprehensive programs span a continuum of services encompassing financial assistance, insurance navigation, educational resources, and round-the-clock customer support. By addressing the key barriers to accessing therapeutic agents, such as affordability constraints or insufficient comprehension of treatment regimens, PSPs aim to augment adherence and overall therapeutic efficacy.
With escalating investment in personalized care, PSPs serve as a crucial nexus between complex therapies, caregivers, and patients. This interconnection paves the way for improved outcomes and ensures an equitable access to innovative treatment modalities. For instance, companies such as Pfizer and Merck have implemented comprehensive patient-assistance programs to improve access to vital medications. Pfizer’s Patient Assistance Program [6] offers gratis or discounted pharmaceuticals to eligible individuals, encompassing over 70 drugs across various therapeutic domains including oncology, immunology, and cardiology. Pfizer’s RxPathways initiative [7] facilitates patient navigation through insurance intricacies, co-payment challenges, and financial aid procurement.
Analogously, Merck’s Access Program [8] aids eligible patients in procuring their prescribed medications, notably cancer immunotherapies such as Keytruda®, by offering co-payment assistance, insurance support, and coverage resources. In 2020, Merck’s program provided assistance to more than 100,000 patients. Both companies reported enhanced patient adherence and satisfaction, underscoring the efficacy of comprehensive support in augmenting treatment accessibility and outcomes.
5. Collaborations and Partnerships: Forging Pathways to Shared Success
In the dynamic landscape of the pharmaceutical sector, partnerships with tech companies and artificial intelligence (AI)-based startups are imperative for delivering value-added services to patients. Strategic collaboration enables the integration of cutting-edge AI, machine learning (ML), and cloud computing advancements into drug development and patient care. These alliances not only streamlined operations but also ameliorated patient outcomes by enhancing personalized medicine, improving access to medications, and expanding digital health services. By synergizing the best of both worlds, Pharma can augment efficiency, expedite research and development (R&D) efforts, and foster a holistic ecosystem that prioritizes patient-centric care. While several companies have started adapting AI and ML-based technologies, one example that stands out is Merck’s strategic alliance with Amazon Web Services (AWS) and Accenture [9]. This collaboration leverages the AWS’s cloud infrastructure and AI capabilities to expedite drug discovery, process voluminous datasets, and rapidly identify and screen drug candidates. AWS’s high-performance computing empowers Merck to model complex biological processes, enables real-time data sharing, optimizes clinical trials, and enhances patient engagement through digital health platforms. Notably, AWS played a pivotal role in streamlining and accelerating Merck’s COVID-19 vaccine R&D in 2020, achieving remarkable efficiency and cost-effectiveness.
6. The Advent of Value-Based Healthcare Paradigms
In recent years, pharmaceutical companies have increasingly transitioned from conventional sales-based reimbursement models to outcome-based pricing. Remuneration in the outcomes-based model is contingent upon the efficacy of treatments in real-world patient outcomes. As this model confers value-based care, it aligns with the interests of the healthcare providers, patients and insurance bodies. Furthermore, by tethering reimbursements to quantifiable results, such as amelioration of symptoms or cessation of disease progression, companies are incentivized to innovate and commercialize therapies that truly improve patient health. Thus, outcomes-based pricing engenders greater transparency in health outcomes, mitigates healthcare expenditure, and ultimately propels more patient-centric care, harmonizing business objectives with improved health outcomes.
Amgen’s cholesterol-lowering drug, Repatha®, exemplifies the implementation of an outcome-based pricing model in action [10]. In contrast to conventional flat-rate pricing, Amgen instituted an innovative pricing model that ties reimbursements to a drug’s efficacy in lowering cholesterol levels and mitigating cardiovascular events. This contractual arrangement stipulates that insurers pay for Repatha® if prescribed patients achieve predefined outcomes, such as reduced low-density lipoprotein (LDL) cholesterol levels. Consequently, this model ensures that patients and healthcare systems incur costs only when the drug demonstrates measurable clinical benefits. In the United States, Amgen collaborated with insurers such as Cigna to offer rebates contingent on patient outcomes. For instance, if patients fail to achieve targeted cholesterol level reductions, insurers may receive a rebate, aligning costs more closely with the drug’s effectiveness. This outcomes-based approach has proven efficacious in driving the adoption of Repatha®, while concurrently offering cost savings and augmented confidence to healthcare providers, patients, and payers.
Conclusion and Outlook
The healthcare landscape is undergoing a paradigm shift from a reactive, pill-centric model to one that embraces a more holistic, integrated approach. This metamorphosis is catalyzed by technological advancements and an evolving comprehension of health, emphasizing more on prevention, personalized care, and psychological well-being as quintessential components of overall health. As we move beyond the pill, it becomes evident that healthcare is no longer confined to disease treatment but encompasses patient empowerment through innovative solutions and bespoke care strategies. Looking ahead, we anticipate further breakthroughs shaping this novel healthcare paradigm. Digital health technologies such as wearable devices and health applications will continue to provide real-time monitoring and actionable insights, facilitating more individualized care and preemptive intervention. For instance, wearable devices like the Apple Watch already monitor vital signs and alert users to potential health anomalies, before they become critical. Concomitantly, AI-powered platforms, such as IBM Watson, are poised to revolutionize diagnostics and treatment planning, offering tailored healthcare solutions based on individual genetic profiles and real-time data.
The trajectory forward necessitates cross-sectoral collaborations – from pharma to tech, and healthcare providers to insurers – to build ecosystems that prioritize patient well-being over mere symptom management. Through this paradigm shift, the healthcare landscape can metamorphose into a proactive system that fosters “holistic health” rather than just treating the illness.
References
[1] Novo Nordisk Global. Changing Diabetes® in Children. Press Release 2024. https://www.novonordisk.com/sustainable-business/access-and-affordability/changing-diabetes-in-children.html (accessed January 17, 2025).
[2] Novartis. Sandoz and Pear Therapeutics announce launch of reSET® for treatment of patients with Substance Use Disorder. Press Release 2018. https://www.novartis.com/news/media-releases/sandoz-and-pear-therapeutics-announce-launch-reset-treatment-patients-substance-use-disorder (accessed January 17, 2025).
[3] Maricich YA, Nunes E V, Campbell ANC, Botbyl JD, Luderer HF. Safety and efficacy of a digital therapeutic for substance use disorder: Secondary analysis of data from a NIDA clinical trials network study. Subst Abus 2022;43:937–42. https://doi.org/10.1080/08897077.2022.2060425.
[4] Ross JS, Sokol ES, Moch H, Mileshkin L, Baciarello G, Losa F, et al. Comprehensive Genomic Profiling of Carcinoma of Unknown Primary Origin: Retrospective Molecular Classification Considering the CUPISCO Study Design. Oncologist 2021;26:e394–402. https://doi.org/10.1002/onco.13597.
[5] Cobain EF, Wu Y-M, Vats P, Chugh R, Worden F, Smith DC, et al. Assessment of Clinical Benefit of Integrative Genomic Profiling in Advanced Solid Tumors. JAMA Oncol 2021;7:525–33. https://doi.org/10.1001/jamaoncol.2020.7987.
[6] Pfizer. Patient Assistance. Press Release 2024. https://www.pfizerpro.com/patient-assistance (accessed January 17, 2025).
[7] Pfizer. Pfizer RxPathways. Press Release 2024. https://www.pfizerrxpathways.com (accessed January 17, 2025).
[8] Merck. The Merck Access Program. Press Release 2024. https://www.merckaccessprogram.com (accessed January 17, 2025).
[9] Accenture. AWS and Accenture Help Merck Use Cloud Technology to Reduce Drug Discovery Time and Accelerate Clinical Trial Development. Press Release 2023. https://newsroom.accenture.com/news/2023/aws-and-accenture-help-merck-use-cloud-technology-to-reduce-drug-discovery-time-and-accelerate-clinical-trial-development (accessed January 17, 2025).
[10] Amgen. Amgen And Harvard Pilgrim Agree To First Cardiovascular Outcomes-Based Refund Contract For Repatha® (Evolocumab). Press Release 2017. https://www.amgen.com/newsroom/press-releases/2017/05/amgen-and-harvard-pilgrim-agree-to-first-cardiovascular-outcomesbased-refund-contract-for-repatha-evolocumab (accessed January 17, 2025).












