Laser Vaccine Adjuvants
Corona virus, Influenza and beyond
Alex Nivorozhkin, Alex Nivorozhkin, Ph.D. is a Boston-based biopharma scientist, consultant and entrepreneur. He is a recognized team builder in life science enterprises with track record in early technologies transfer and development. Trained at the top academic institutions such as Harvard University, Dr. Alex Nivorozhkin has a prolific publications’ record and extensive patents’ portfolio.
Recent medical research has proved that low power nondestructive lasers boost vaccine immune response for most threatening infections of our time. Enabled by recently developed inexpensive devices and involving just a minute-long treatment, this novel approach breaks existing stereotypes and appears ready for wide clinical testing and practice.
Vaccine adjuvants represent an essential tool to augment human immune response. Despite significant advances in medical research, practical progress in commercial development of new modalities has been slow till recently. Alum salts have remained a gold standard adjuvant for almost one hundred years since its introduction in 1920s. Alum salts and other particulate adjuvants act by enhancing the antigen presentation to the innate immune cells and improving uptake of the antigen. In modern times, discovery of Pattern Recognition Receptors led to advancement of new type of adjuvants. They potentiate specific innate immune signals through Toll-like Receptors (TLRs). Combination of both approaches allows for better tuning of the immune response to a specific antigen. Majority of current clinical development candidates in Phase II, III clinical trials are TLR adjuvants.
Industrial developments of novel adjuvanted vaccines remain a very laborious endeavor. The problems stem from safety of adjuvant components and their formulation aspects. Alum vaccines cannot be frozen due to particle aggregations. Some popular lipid-based adjuvants contain naturally derived squalene oil that is hard to purify and standardize. Translation of the vaccine adjuvants between different vaccines has many challenges. Safety and efficacy data are hard to separate between two components of the vaccine formulation, i.e. a vaccine part and its adjuvant. Therefore the whole package cannot be easily decoupled from a specific target disease of the original vaccine. For example, a widely publicized recent agreement by GSK to make their flu vaccine adjuvant AS03 (squalene, tocopherol, polysorbate) available to the CEPI non-profit group for coronavirus vaccine is marred by controversy about observed narcolepsy cases.
Inherent problems of the ‘chemical’ vaccine adjuvants are apparent. Simple and safer ‘physical’ adjuvants like a beam of light applied to skin vaccination sites have attracted attention of immunologists.
Some of the laser vaccine adjuvant concept studies were conducted in the Russian labs in late 1990s led by Dr. Sergei Onikienko. These studies demonstrated high adjuvant efficacy in the patients’ flu vaccination. The author of this article has introduced these findings to Harvard Medical School (HMS) researchers in 2005. Two HMS groups confirmed and expanded the original findings from a Cu nanosecond pulsed laser (operated at a dual wavelength 510/578 nm), funded by NIH and the Bill & Melinda Gates Foundation. One of the principal investigators, Prof. Satoshi Kashiwagi at Massachusetts General Hospital (MGH), has published the most recent review that described the launch of these studies, key results and subsequent work by other researchers.
Early MGH mice studies adopted a modern semiconductor Q-switched Nd:YAG (532 nm) nanosecond pulsed laser and showed a robust antibody generation to both intradermally administered influenza vaccines and ovalbumin. This work has supported original findings of increased motility of antigen presenting cells (APC), along with increased antigen and helper T-cell priming in draining lymph nodes. Influenza vaccine’s humoral immune response increased 4x, getting inline with usable ‘chemical’ adjuvants. The laser irradiation lasted 1-2 min and the irradiation zone had a diameter of about 5 mm, with an average energy density delivered 1 W/cm². The studies have been calibrated to control the skin temperature upon laser exposure not to exceed 43oC to prevent onset of thermal damage.
Lasers within visible part of the spectrum are known to interact with melanin. It leads to variable skin heating dependent on the skin types. So the infrared lasers were adopted to standardize thermal outcomes. These are inexpensive portable non-pulsed machines, many of which used in cosmetic procedures, in contrast to highly priced(tens of thousands dollars) pulsed laser systems that are heavy stationary objects requiring engineering support. Additionally, fractional cosmetic lasers designed to induce some thermal skin damage (for stimulating collagen production) were also tested as vaccine adjuvants.
Presently, there are published data for 4 (four) laser types used as vaccine adjuvant: (i) ultra short pulsed lasers (UPLs), (ii) continuous wavelasers (CWL), (iii) non-ablative fractional lasers (NAFL), and (iv) Ablative Fractional Lasers (AFL).
AFL cosmetic lasers work by creating a tunable array of wells in the skin, few tens of microns wide and few hundreds micron deep, with a density of 100-500 wells/cm². The wells are generated via explosive water superheating and are lined up with coagulated dead tissue. NAFL lasers use lesser energies and limit skin damage to production of the columns containing mostly coagulated tissues. AFL lasers are believed to have a dual benefit of both an immune activation and effectively delivering the antigen via topical application (through the formed micro-channels) eliminating the need for injection.
A complex mechanistic picture that emerged regarding the specific pathway for enhancing vaccine response is still evolving and may be briefly conceptualized as follows. NAFL/AFL treatment induces skin cell death releasing damage-associated molecular patterns (DAMPs) including dsDNA that are taken up by antigen-presenting cells (APCs) and recognized by intracellular pattern recognition receptors (PRRs). PRRs then transduce signals to STING activating IRF3 and NF-κB. Enhanced maturation and migration of APCs is stimulated by overexpressed Type I interferon, pro-inflammatory cytokines and chemokines. In CWL lasers, the key role is presumably played by reactive oxygen species (ROS), which start the immune response cascade in the skin. Non-damaging UPLs induce extracellular release of HSP70 and cause disarray in extracellular matrices thus activating Langerhans cell and facilitating migration of DCs.
Human data to support effective vaccination using lasers are still lacking and require more industry players. Pantec Biosolutions (Ruggell, Liechtenstein) in collaboration with University of Vienna conduct a Phase I human study, in which 4 adjacent areas of 2 cm² each (14 x 14 mm) will be pretreated with an Er:YA Glaser (2940 nm, 22.7 J/cm², 2 pulses, density: 5%) generating micro-pores with a depth of ca. 91 microns. 0.1 ml of Intanza (15 µg, seasonal trivalent influenza vaccine) will be topically administered on the laser-treated area. The immunogenicity of the vaccine is planned to be assessed by evaluating activation of B-cell mediated and T-cell mediated immune responses of the laser-assisted epidermally administered seasonal influenza vaccine versus needle-based intradermal administration.
MGH group has recently published a small set of human data on the safety of the 1064 nm CWL laser (IPG Photonics, Oxford, MA) that was able to delivery energy about 1 W/cm² to a skin spot of 5 mm diameter over 1 min exposure time without sensory disturbances. However, this study didn't include any vaccination protocols.
Alternative Innovative Technologies, LLC (Boston, MA) developed a first in class inexpensive portable UPL laser operating at 930 nm (independent of the skin type) and conduct an ongoing clinical testing in Russia to enhance the efficacy of cancer vaccination (Fig. 1, 2). In these studies, the oncolytic properties of non-pathogenic for humans Sendai virus have been dramatically enhanced via laser-stimulated delivery and viral accumulation/propagation at the tumor and inflammation sites.
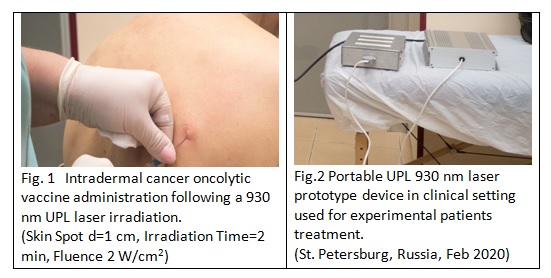
Notably, Sendai virus has been used in early commercial preparations of interferon in chicken eggs and found stimulating human interferon production, incubated ex vivo with leucocytes. Recently published data indicate that Corona virus suppresses the immune system in general and interferon production specifically, and that may be one of the key factors to its spread, high morbidity and mortality. Thus a therapeutic approach of using ‘laser-activated’ Sendai virus as a COVID-19 antagonist may be a viable hypothesis for future clinical testing.
In summary, laser vaccine adjuvants show promise in multiple animal models and early human clinical testing, both as a standalone or combined with ‘chemical’ adjuvants. There are several laser engineering platforms and specific devices that could be easily (re)configured to specific vaccination types and rapidly and economically produced on a commercial scale to enable mass vaccination. That could boost further development of intradermal vaccines that in general allow for significant dose sparing and accelerated speed to market, an essential attribute for any effective measures to fast respond to rapidly evolving and high dangerous pathogens such as COVID-19. We are lagging behind in wider adoption of the new intradermal vaccine delivery platforms other than intradermal needle (e.g., micro-needle arrays, jet injectors, etc.) that will be an important roadblock to overcome.














