Lipid Based Drug Delivery Systems
A New Wave to Improve Bioavailability of Class II and IV Drugs
Venu Madhav Katla, Professor, Department of Pharmaceutics, St. Paul's College of Pharmacy
Sridhar Babu Gummadi, Professor, Department of Pharmaceutical Chemistry, Sri Shivani College of Pharmacy
Somnath De, Professor, Department of Pharmacology, St.Paul's College of Pharmacy
In current situation of pharmaceutical industries, drug development and technology most of the drugs comes under either BCS class II or IV which effects on drug solubility and its permeability thereby bioavailability. It is difficult to prepare these drugs in the form of conventional dosage forms because these drugs will not dissolve in GIT which creates solubility issues thereby poor bioavailabilty of the drug after taking orally. To overcome this aspect Lipid-based drug delivery systems (LBDDS) is one of the novel techniques for delivering the orally either by dissolving or dispersing or incorporation or encapsulation in an lipid excipient which results in improving the oral bioavailability. This review gives an importance of LBDDS, its mechanisms, applications and types of dosage forms available for different delivery aspects.
In the recent progressive era of pharmaceutical research and technology investigated new chemical entity (NCEs) is generally from Biopharmaceutical classification system (BCS) class II (low solubility and high permeability) and IV (high solubility and low permeability). From the discovered chemical entities, most of the bio actives have high molecular weight, low solubility and high permeability. These properties limit the absorption of the drug and also its oral bioavailability [1].
The oral bioavailability of drugs is mainly dependent on aqueous solubility of drugs especially in the GIT. These are directly proportional to each other which effects the dose of drug and response variability in different subjects [2]. To bypass or overcome such problems, different formulation aspects are being come into account such as use of surfactants, cosolvancy, use of permeation enhancers, micronisation, co-crystallisation, salt formation, cyclodextrins, complexation, nanoparticles, solid dispersions and recently, much importance and attention was given to lipid-based formulations [3].
Lipid-based drug delivery system (LBDDS) in recent days gained much attention as a novel drug delivery in improving of drug solubility, dissolution and bioavailability of low or poorly aqueous soluble drugs. These LBDDS also play major role in drug targeting especially in case of treating tumors. These preparations show many advantages such as biocompatibility and biodegradability. LBDDS can reduce or overcome the problem associated with slow or incomplete dissolution of poorly aqueous soluble drugs by formation of soluble structure of core drug after digestion in the gastro intestinal tract (GIT) which facilitates drug absorption and thereby improving its oral bioavailability [4]. These lipid formulations also protect the drugs from enzymatic and biological degradation which in turn improve drug potency. These formulations also reduce the toxicity of most drugs. Most lipid delivery systems used as drug carriers which increases drug stability and also capable of incorporating hydrophobic and hydrophilic substances, have feasibility of administration into various routes i.e., conventional, parenteral, topical and pulmonary route [5].
LBDDS are mainly consisting of lipids; surfactant, co-surfactant and also sometimes contain co solvents. These are of many types of formulations with wide range of properties in which can incorporate various oils (mono, di and tri glycerides), surfactants (water soluble and insoluble) with co-surfactants / co-solvents [6].
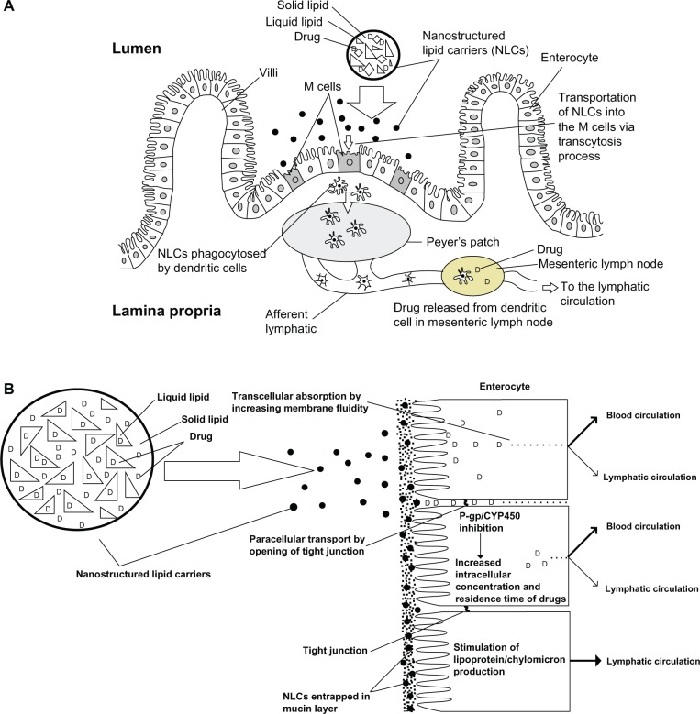
Fig. 1 Mechanisms of intestinal drug transport from lipid based formulations (adopted from [7]).
Lipid formulation classification system
A working model of a lipid formulation classification system (LFCS) was introduced by Pouton et al., 2000 [8] and further an extra formulation type was added in 2006 [9]. The in vivo performance of various drugs and its physico chemical properties, formulations can be interpreted easily by using of LFCS. Most of the marketed formulations are comes under Type III systems which are composed of wide range of oil & water soluble substances [10].
Type I lipid formulations are suitable for potent drugs or drugs having high solubility in oils. Type II formulations contain high concentration of surfactant. Above 65% concentration of surfactant, there is formation of liquid crystalline gel phases at o/w interphase. Such systems when dispersed in water produce droplet size above 250nm (SEDDS). Type III formulations produce droplet size 50-250nm (SMEDDS/SNEDDS).
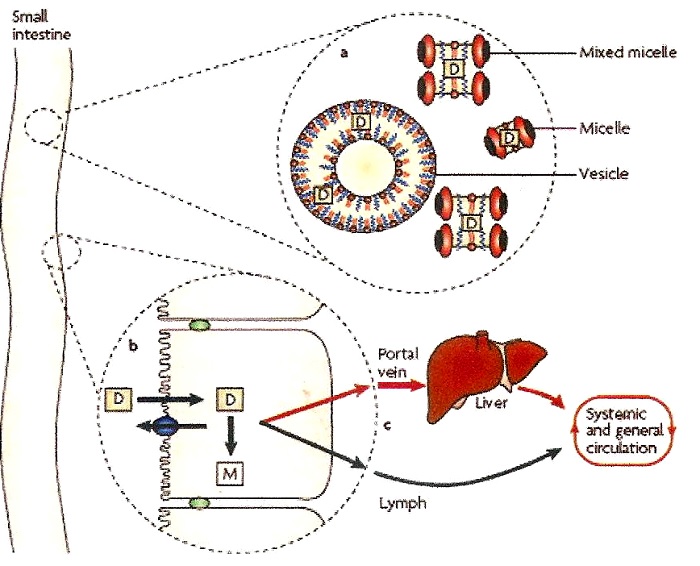
Fig.2 Various mechanisms of enhancement of drug bioavailability in the presence of lipids (adopted from [12]).
Table 2: Lipid based formulations for improving of oral bioavailability
| Strategy | Description |
| More time of retention in stomach | Reaching of lipids into the GIT will result in slower peristaltic movement, gastric emptying, and simultaneously increase of retention time in upper part of intestine, where absorption occurs. |
| Improved solubility | Presence of lipids in the GIT stimulate and increase excretion of bile lipids and bile salts (including cholesterol and phospholipids), this leads to emulsification of the lipids thereby drug solubilisation. |
| Changes in physical and biochemical barrier | Presence and absence of physical and chemical barriers in the intestine plays important role in absorption of drugs GI lumen. These will inhibit some metabolic enzymes which are involved in reducing of absorption of drugs before giving its action. Presence of intestinal vectors in GI wall (P-glycoprotein). Sometimes combination of lipids and surfactant will promote intestinal absorption of drugs. |
| Stimulation of intestinal lymphatic transport | Lipids are of either Long-chain triglycerides (LCT) or medium-chain triglycerides (MCT) in which MCT is transported directly by the portal blood into the systemic circulation but where as LCT stimulates lipoproteins which facilitates lymphatic transport. |
Classification of Lipid-based systems:
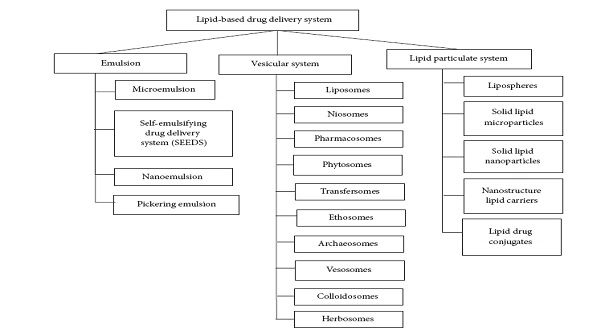
Fig. 3 Classification of Lipid based systems (adopted from 13).
A. Micro-emulsion: These are transparent and thermodynamically stable preparations. It consists of oil, surfactant and co-surfactant. When compared with simple or normal emulsion, micro-emulsions are formed by simple mixing and have particle size between 10-200 nm but where as normal emulsion have particle size between 1-20 μm [14].
B. Self-Emulsifying Drug Delivery System (SEDDS): These are isotropic mixtures of oil, surfactant and co-surfactant and thermodynamically stable. These will form an emulsion when they come in contact with aqueous fluid of GI tract. Depending on variation in particle sizes these are differentiated as SMEDDS (100 – 250 nm) and SNEDDS (< 100 nm). [15].
C. Nano-emulsion: These are primarily made up of oil and water which are stabilised by adding up of either surfactant or alcohol. These are oil-in-water (O/W) type of emulsion with droplet size of between 10 to 200 nm and water-in-oil (W/O) forms, with droplet size of more than 200 nm [16].
D. Pickering Emulsion: It is an O/W type of emulsion which is stabilised by solid particles that are present at the interface of two systems, which leads to reduction of surface energy [17].
E. Liposomes: Liposomes are artificial vesicles consisting of lipid bilayer which is usually made up of phospholipids and cholesterol, have particle diameter from 0.01 and 100 μm. These are classified as multilamellar vesicles (MLVs), small unilamellar vesicles (SUVs) and large unilamellar vesicles (LUVs) [18].
F. Niosomes: These are also known as non-ionic surfactant vesicles which are alternative of liposomes and have size between 10 to 100 nm [19].
G. Pharmacosomes: These are termed as colloidal dispersions in which drugs are covalently bonded to the lipids components. These systems are made up of drug (pharmakon), carrier (soma), combinely termed as Pharmacosomes.
H. Phytosomes: In other way it is expressed as Herbosomes, which is a combination of two words in which phyto means as plant and somes means as cell-like structure.
I. Transfersomes: Transfersomes means carrying of the body. In which transferred means “take across‟ and soma means “body‟ [20].
J. Ethosomes: These are malleable soft vesicular structures basically used for delivery of drugs by transdermal route. These are vesicular systems chiefly comprises of phospholipids and the solvent system (hydroalcoholic mixture) contains an active ingredient in a core system.
K. Archaeosomes: These are vesicular systems containing archebacteria lipids and usually made up of one or more fully saturated bipolar tetra ether lipids. These are less sensitive to oxidative stress, alkaline pH and high temperature.
L. Vesosomes: These are of vesicular structure which consists of one or more bilayers enclosed in aqueous core. Along with drug it contain unilamellar vesicles as an internal compartment.
M. Colloidosomes: These are small microcapsules in which shell consists of closely packed colloidal particles. Their physical properties, such as its mechanical strength, permeability, biocompatibility need to be controlled by using of proper colloids, excipients for formulation.
N. Liposphere: These are particulate dispersion which are of solid spherical particles having diameter from 0.2 to 100 μm The contains triglycerides or fatty acids as solid lipophilic core which are stabilised by monolayer of phospholipids.
O. Solid-Lipid Microparticles: Micro systems are used as sustained release drug delivery systems; but its safety remains uncertain, this lends for the development of solid lipid microparticles (SLM).
P. Solid Lipid Nanoparticles (SLNs): These were developed in 1990 as an alternative of other lipid based systems like liposomes, nanoemulsion and polymeric particles. These are having size ranges from 50-1000 nm.
Q. Lipid Drug Conjugates (LDC): These are developed especially for hydrophilic drug molecules. Here the drug and lipid conjugate are prepared by either covalent linking or salt formation.
Future prospects
In future, more consideration will to be paid for the characterisation of various lipid formulations, which plays an important role in improving drug solubility and their by its bioavailability by using of suitable excipients and formulations in an early stage of product development. Methods need to be sought for tracking the solubilisation state of the drug in vivo, and there is a need for in vitro methods for predicting the dynamic changes, which are expected to take place in the gut. Attention to the physical and chemical stability of drugs within lipid systems and the interactions of lipid systems with the components of capsule shells will also be required. By this leads in selection or use of lipid formulations as potential drug delivery systems. The priority for future research should be to conduct human bioavailability studies and to conduct more basic studies on the mechanisms of action of this fascinating and diverse group of formulations.
References
1. S. Kalepu, M. Manthina, V. Padavala, Oral lipid-based drug delivery systems–an overview, Acta. Pharm. Sin. B. 3 (2013) 361-372.
2. Z. Sheikh, M. Nabila, Optimizing oral drug delivery using lipid based formulations, Int. Res. J. Pharm, 5 (2014) 514-522.
3. B.J. Aungst, Novel formulation strategies for improving oral bioavailability of drugs with poor membrane permeation or presystemic metabolism, J. Pharm. Sci. 82 (1993) 979-987.
4. A.K. Bansal, K. Sarpal, Y.B. Pawar, Self emulsifying drug delivery systems: A strategy to improve oral bioavailability, CRIPS. 11 (2010) 42-49.
5. K. Čerpnjak, A. Zvonar, M. Gašperlin, F. Vrečer, Lipid-based systems as a promising approach for enhancing the bioavailability of poorly water-soluble drugs, Acta. Pharm. Sin. B. 63 (2013) 427-445.
6. A. Rahman, Oral lipid based drug delivery system (LBDDS): formulation, characterization and application, Curr. Drug. Deliv. 8 (2011) 330-345.
7. A. Ali Khan, J. Mudassir, N. Mohtar, Y. Darwis, Advanced drug delivery to the lymphatic system: lipid-based nanoformulations, Int. J. Nanomed. 8 (2013) 2733-2744.
8. C.W. Pouton, Lipid formulations for oral administration of drugs: non-emulsifying, self emulsifying and ‘self-micro emulsifying’ drug delivery systems, Eur. J. Pharm. Sci. 11 (2000) 93–98.
9. C.W. Pouton, Formulation of poorly water-soluble drugs for oral administration: physico chemical and physiological issues and the lipid formulation classification system, Eur. J. Pharm. Sci. 29 (2006) 278–287.
10. C.W. Pouton, Formulation of self-emulsifying drug delivery systems, Adv. Drug. Deliv. Rev. 25 (1997) 47–58.
11. C.W. Pouton, C.J.H. Porter, Formulation of lipid-based delivery systems for oral administration: Materials, methods and strategies, Adv. Drug. Deliv. Rev. 60 (2008) 625–637.
12. S. Chakraborty, D. Shukla, B. Mishra, S. Singh, Lipid - an emerging platform for oral delivery of drugs with poor bioavailability, Eur. J. Pharm. Biopharm. 73 (2009) 1-15.
13. H. Shrestha, B. Rajni, A. Sandeep, Lipid-Based Drug Delivery Systems, J. Pharm, 8 (2014) 1-10.
14. B. Shamant, Lipid based drug delivery system in arthritis and allied conditions, World J. Pharm. Res. 4 (2016) 61-68.
15. S. Rani, Self-Emulsifying oral lipid drug delivery systems: advances and challenges, AAPS Pharm. Sci. Tech. 20 (2019) 1-12.
16. S. Chime, I. Onyishi, Lipid-based drug delivery systems (LDDS): Recent advances and applications of lipids in drug delivery, AJPP. 7 (2013) 3034-3059.
17. R. L. McMullen, M. Gorcea M, S. Chen, Emulsions and their characterization by texture profile analysis., Formulating Topical Applications-A Practical Guide (2013) 131-153.
18. B.J. Boyd, T. H. Nguyen, A. Müllertz, Lipids in oral controlled release drug delivery, Cont. oral. Drug. Deliv. 10 (2011) 299–327.
19. M. Oliveira, Hydrophobic ion pairing as a strategy to improve drug encapsulation into lipid nanocarriers for the cancer treatment, Exp. Opinion. Drug. Deliv 14 (2017) 983-995.
20. M. Rajabi, S. Mousa, Lipid nanoparticles and their application in nanomedicine, Cur. Pharm Biotech 17 (2016) 662-672.














