The Emergence of Organ-on-chips for Diseases Studies and Drug Development
Giuseppe Maruccio, Full Professor, Omnics Research Group @ University of Salento, CNR-Nanotec and INFN-Lecce
Anna Grazia Monteduro, Researcher, Omnics Research Group @ University of Salento, CNR-Nanotec, and INFN-Lecce
Silvia Rizzato, Researcher, Omnics Research Group @ University of Salento, CNR-Nanotec and INFN-Lecce
Giusi Caragnano, PhD Student, Omnics Research Group @ University of Salento, CNR-Nanotec and INFN-Lecce
Organ-on-chips have recently emerged as microphysiological systems enabling to overcome the limitations of traditional cell culture methods and the use of animal models [1, 2]. They can simulate the functional unit of an organ, account for multi-organ interplay, and facilitate the drug development process and progress in the present knowledge on pathological mechanisms responsible for human illnesses.
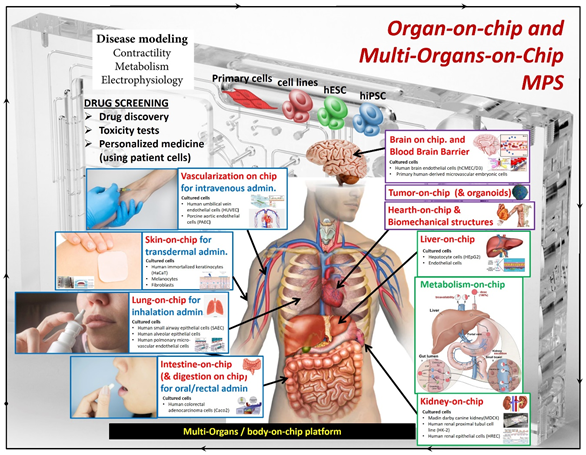
Organ-on-chip models with details about their involvement in different drug delivery routes and the employed cell lines [reproduced from A. G. Monteduro, S. Rizzato, G. Caragnano, A. Trapani, G. Giannelli and G. Maruccio, Organs-on-chips technologies – A guide from disease models to opportunities for drug development, Biosensors and Bioelectronics 2023, 231, 115271, http://dx.doi.org/https://doi.org/10.1016/j.bios.2023.115271]
A new frontier of technology: Organ-on-chips
In recent years, Organ-on-chips (OOC) attracted large interest as a new enabling technology to better understand human pathophysiological mechanisms and develop new drug treatments [1, 2]. These microfluidic devices can recapitulate the functional unit of an organ, reproducing the tissue architecture and mechanics of the organ in question. Differently from 2D cultures, Organ-on-chips can allow 3D growth and account for cell-cell interactions, and can be subjected to mechanical stimuli that mimic those present in vivo such as the movement of intestinal peristalsis, lung respiration, and heartbeat. Compared to animal models, OOC does not have ethical issues and does not have the problem of limited translatability of results between humans and animals. In addition, sensors can be integrated into OOCs to monitor cell activity, such as cell proliferation, or changes in oxygen concentration or pH that are reflected in cell culture conditions. OOCs are made by microfabrication techniques such as photolithography and soft lithography.[1, 2]
A new tool for drug discovery
The process of identifying a new drug is very long, costly, and partially inefficient. On average, it takes about 15 years, highly specialized personnel is needed, millions of dollars are spent and the success rate in identifying a new drug is very low, less than 5%, resulting in a waste of many resources. OOC microfluidic devices could speed up this process, being faster and better at testing the efficacy of a drug on a target organ and also assessing its toxicity on other organs through which the drug passes [1, 3]. In the search for new orally administered drugs, the compound toxicity on the involved organs can be investigated on the chip, including the effect on the liver where it is metabolized, and the kidneys by which it is eliminated [2, 4]. For this purpose, researchers developed multi-organ-on-chip devices, having interconnected compartments for each target organ.
Multi-organs platforms
Multi-organ on-chip (MOC) devices are intended to recapitulate the communication between the various organs, to represent the interface of different tissues that make up biological barriers using different closely connected compartments. These models can also contribute to investigating the kinetic and dynamic course of a drug (PK-PD) by monitoring the various responses of multiple organs to pharmaceutical compounds under development [1, 5]. MOC platforms can be used to study the absorption, distribution, metabolism, and elimination (ADME) process of a drug. For example, the administration of an inhaled drug can be analyzed by recreating the pulmonary microenvironment and that of the liver, the main organ where drugs are metabolized, and connecting them. It is also possible to explore the pathway of an orally administered drug by simulating the intestinal, hepatic, and renal compartments [6]. Multi-organ platforms can be useful for predicting the response of a cancer drug on the target organ and for investigating the side effects it may cause in other organs [1, 7]. The challenge of the future will be to move from organ-on-chip to body-on-chip to obtain the ultimate platform for pharmaceutical experimentation.[8]
Organ-on-chips with integrated sensors
Beyond cell cultures, Organ-on-chip devices should be coupled with high-performance analytical methods to fully exploit the opportunities provided by these technologies. Due to the small volumes within OOC platforms, traditional methods such as high-performance liquid chromatography (HPLC) or assays based on endpoint analysis present limitations in their use. However, miniaturized sensors can be integrated into Organ-on-chips to collect data in an automated and continuous manner [1, 9, 10]. Using sensors, it is possible to obtain information on cell culture, cell proliferation, and cell responses following the introduction of a molecule, drug, or another external stimulus. Among the different transducer approaches, electrical sensors are the most commonly employed in OOCs, and trans-epithelial/endothelial electrical resistance (TEER) assays are widely used to assess the permeability of biological barriers. Electrochemical sensors can also provide tools to monitor glucose and metabolites
Take home messages
Organ-on-chips represent an alternative to classical cell culture methods and animal experimentation for the study of human physiology, disease mechanisms, and drug development. These micro-engineered systems comprise a series of microfluidic channels/compartments, sometimes integrated with sensors, in which to recapitulate the major functions of a single organ or even multi-organ interplay. In this way, it is possible to recreate a cellular microenvironment similar to that present in a pathophysiological situation and collect information similar to what would be obtained in vivo. This new technology can lead to a paradigm shift in research. For more information, the interested readers are referred to recent reviews in the field such as ref. [1].
References
- Monteduro, A.G., et al., Organs-on-chips technologies – A guide from disease models to opportunities for drug development. Biosensors and Bioelectronics, 2023. 231: p. 115271.
- Bhatia, S.N. and D.E. Ingber, Microfluidic organs-on-chips. Nat Biotechnol, 2014. 32(8): p. 760-72.
- Bovard, D., et al., A lung/liver-on-a-chip platform for acute and chronic toxicity studies. Lab Chip, 2018. 18(24): p. 3814-3829.
- Lee, H., et al., A pumpless multi-organ-on-a-chip (MOC) combined with a pharmacokinetic-pharmacodynamic (PK-PD) model. Biotechnol Bioeng, 2017. 114(2): p. 432-443.
- Zhao, Y., et al., Multi-Organs-on-Chips: Towards Long-Term Biomedical Investigations. Molecules, 2019. 24(4).
- Choe, A., et al., Microfluidic Gut-liver chip for reproducing the first pass metabolism. Biomed Microdevices, 2017. 19(1): p. 4.
- Li, Z., et al., Evaluation of hepatic drug-metabolism for glioblastoma using liver-brain chip. Biotechnol Lett, 2021. 43(2): p. 383-392.
- Lacombe, J., M. Soldevila, and F. Zenhausern, From organ-on-chip to body-on-chip: The next generation of microfluidics platforms for in vitro drug efficacy and toxicity testing. Prog Mol Biol Transl Sci, 2022. 187(1): p. 41-91.
- Fuchs, S., et al., In-Line Analysis of Organ-on-Chip Systems with Sensors: Integration, Fabrication, Challenges, and Potential. ACS Biomater Sci Eng, 2021. 7(7): p. 2926-2948.
- Clarke, G.A., et al., Advancement of Sensor Integrated Organ-on-Chip Devices. Sensors (Basel), 2021. 21(4).















