The Role of Artificial Intelligence in Advancing Pharmacovigilance Systems
Rahul Jaiswal, Faculty of Pharmacy, Integral University, Hygia College of Pharmacy, and Dr. A.P.J. Abdul Kalam Technical University
Artificial Intelligence (AI) is revolutionising pharmacovigilance by streamlining drug safety processes, enabling real-time adverse event monitoring, and enhancing decision-making. This article examines the fundamentals of AI in healthcare, its application in pharmacovigilance, associated challenges, and future prospects, highlighting its transformative impact on patient safety and pharmaceutical innovation.
1. Introduction to Pharmacovigilance and its Significance
Pharmacovigilance refers to the detection, assessment, monitoring, and prevention of adverse drug reactions. It is an essential part of scientific research and healthcare and has paramount importance in monitoring the safety and effectiveness of pharmaceuticals. Pharmacovigilance can be traced back to the 20th century, and it has grown rapidly since then. By the end of the 20th century, pharmacovigilance was being performed in various countries. Today, pharmacovigilance is prevalent all around the globe and is governed by several regulations. The main role of these regulations is to ensure the safety and quality of pharmaceuticals and is enforced to protect public health. A core component of pharmacovigilance is the Adverse Drug Reaction (ADR). ADR is defined as "an appreciably harmful or unpleasant reaction resulting from an intervention related to the use of a medicinal product."
ADR can be classified into six categories: Type A, B, C, D, E, and F. There are multiple ADR reporting systems in place, including ADR monitoring within healthcare settings, the healthcare professional reporting system, the pharmaceutical company reporting system, and the regulatory authority reporting system. The various stakeholders involved in pharmacovigilance include healthcare professionals, patients and consumers, pharmaceutical industries and companies, and regulatory authorities, establishing pharmacovigilance centers. Traditional pharmacovigilance systems face many challenges, such as underreporting, qualitative and quantitative data, availability of broad data, data management, and data regulations; thus, pharmacovigilance systems have an urgent need for further development to ensure drug safety. Figure 1, a flowchart illustrating the steps involved in the pharmacovigilance process.
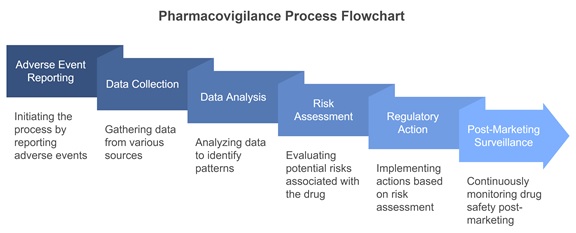
Figure 1: A flowchart showing the pharmacovigilance process
2. Fundamentals of Artificial Intelligence in Healthcare
Artificial Intelligence (AI) fundamentally is the ability of a machine to display human-like cognitive functions or perform tasks requiring human intelligence. The AI revolution has helped in solving real-world problems and has found varied verticals. The phenomenon of this intelligent solution seamlessly encroached on healthcare – a domain where we don’t want half-baked solutions. The introduction of AI was not instantaneous in healthcare, and the domain primarily focused on diagnostic healthcare. The use of AI in diagnostics can detect patterns, correlations, and provide reasoning that humans make. The domain has evolved, and today the AI tools support diagnostics, manage, and treat healthcare. Machine learning, a sub-segment of AI, helps the model learn from existing data without human intermediate contribution. Natural language processing supports understanding human speech, while data analytics classify and predict outcomes. This Figure 2 provides a visual representation of common applications of AI, highlighting key areas where artificial intelligence is widely utilised.
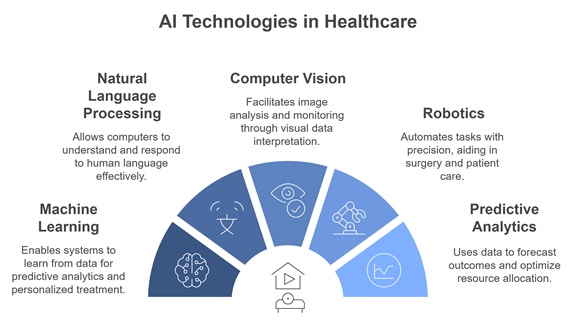
Figure 2: A visual diagram illustrating common AI technologies
AI in healthcare is for innovative technology, from behind-the-scenes administrative capabilities up to the point of care and onto recovery processes. The world has seen new platforms and solutions centered on transforming the patient experience into better prevention, diagnostics, and the strengthening of patient management. The inclusion of AI in healthcare strengthens precision, personalisation, and democratisation of healthcare. AI-based products can help streamline patient management systems in healthcare, as the days of dumb records will perish and digitisation will boom. The AI platforms can handle a lot of redundant tasks of practitioners and hospitals, coming into the decision assistive role. AI provides maturity and experience with a base of millions of cases across physical or virtual barriers quicker than traditional medical research.
Many legacy systems are not equipped to handle large volumes of data nor the volumetric speed of the data. AI-infused algorithms use the 5Vs of big data to convert IoT enabled digital data in the area of healthcare for better decision-making. The advancements in sensors and data reporting standardisation open numerous opportunities for continuous and unobstructed patient data creation. The arrival of AI would augment new technologies, ensuring that data integrity, patient identification, and privacy are maintained at all times. Ethical, moral, and legal issues that govern the responsible deployment of AI need to be discussed at different forums. Predictive modeling could support or improve clinicians’ diagnostic capabilities and can be used to predict health trends, prevent diseases, facilitate diagnosis and management, support clinical treatment and decision-making, and optimise patient care management. AI model training is a data-consuming process, increasing the responsibility and data privacy in the development of AI applications in healthcare. AI is supported by existing AI platforms such as machine learning, deep learning algorithms, and differentially analysing health data. AI will also improve the efficiency and effectiveness of activities. AI models can assist in detecting an excessive number of cases or deaths, assuring immediate regulatory alerts. A few independent evaluations are carried out to determine the safety of AI applications in any field. This Table 1 compares traditional and AI-driven healthcare approaches, highlighting differences in speed, accuracy, scalability, and data handling.
Table 1: Comparison of traditional vs. AI-driven approaches in healthcare
| Aspect | Traditional Approach | AI-Driven Approach |
| Speed | Slower data processing and analysis, reliant on manual review. | Faster processing and analysis, real-time data handling. |
| Accuracy | Subject to human error and biases in diagnosis. | Increased accuracy through machine learning algorithms. |
| Scalability | Limited by resources, labor-intensive for large datasets. | Highly scalable, able to handle vast amounts of data. |
| Data Handling | Manual collection and analysis of patient data. | Automates data extraction, integration, and analysis. |
| Decision-Making | Decisions may be slower due to reliance on clinical judgment. | AI models assist in faster decision-making with evidence-based predictions. |
| Cost Efficiency | Higher due to manual labor, slower processes, and longer wait times. | Can reduce costs by automating processes and improving efficiency. |
3. Integration of AI Technologies in Pharmacovigilance Systems
Integration of AI Technologies in Pharmacovigilance Systems: Actually, there is a convergence stage where many of these real systems might not be driven purely by artificial intelligence, but they will have AI tools integrated. In fact, there is great potential in how AI-driven data analysis can improve upon traditional pharmacovigilance systems. Within the drug development framework, the pharmacovigilance stage is where adverse drug reactions begin to be detected. Using AI technologies such as machine learning algorithms, the ability of pharmacovigilance systems to detect adverse drug reactions can be enhanced. Often, pharmacovigilance databases contain vast amounts of interlinked clinical history data that, when perturbed by a drug, seem unrelated to the newcomer. AI technologies can be utilised to help pick out the true signal of adverse events by pattern finding and predicting potential risks. This can lead to a quicker updating of safety and efficacy summaries, and hence, faster communication to healthcare professionals. The following Table 2 highlights key AI applications in pharmacovigilance, showcasing their functionalities and real-world implementations.
Table 2: Examples of AI Applications in Pharmacovigilance
| AI Application | Description | Real-World Example |
| Adverse Event Detection | AI systems analyse large datasets, including social media and EHRs, to identify potential adverse drug reactions. | IBM Watson Health uses AI to detect adverse events from electronic health records. |
| Predictive Modeling | AI predicts the likelihood of adverse events based on patient demographics, drug interactions, and usage patterns. | AstraZeneca employs machine learning to predict drug safety risks during development. |
| Signal Detection Tools | AI algorithms identify signals of potential safety concerns by processing pharmacovigilance databases. | FDA Sentinel System uses AI to enhance signal detection in pharmacovigilance. |
| Natural Language Processing (NLP) | NLP extracts adverse event reports from unstructured data, such as clinical notes or social media posts. | BioPharm Solutions leverages NLP to analyse social media for drug safety insights. |
| Automation of Reporting | AI automates the creation and submission of regulatory safety reports to streamline compliance. | ArisGlobal’s LifeSphere automates case processing and regulatory reporting tasks. |
| Risk-Benefit Analysis | AI assesses drug safety profiles by balancing risks and benefits across patient populations. | Bayer uses AI-driven platforms for comprehensive drug safety evaluations. |
There is actual application; for example, an application proposed in the development of an AI-powered integrated pharmacovigilance and medication surveillance framework. The theoretical use of AI technologies should only be trusted as far as the surrounding human processes. Finally, for pharmacovigilance systems, it is essential that they are capable of being fully interoperable with existing databases. This is important for the exchange of data and to allow the acquisition of evidence chains for causal analysis and monitoring. As with other applications, such as mobile applications and chatbots for healthcare delivery, the relevant infrastructure must be built, and training and capacity-building would be needed to operationalise these systems successfully. In terms of investment of time and resources, it is currently too early to make a valuation of their collective benefit. But further development and implementation offer benefits, not only in terms of refining drug safety and efficacy parameters but also in reducing the cost to pharmaceutical companies in terms of investment per blockbuster medication. In Figure 3, the workflow of an AI-enhanced pharmacovigilance system is illustrated, showcasing the integration of AI technologies in detecting, analysing, and managing adverse drug reactions efficiently.
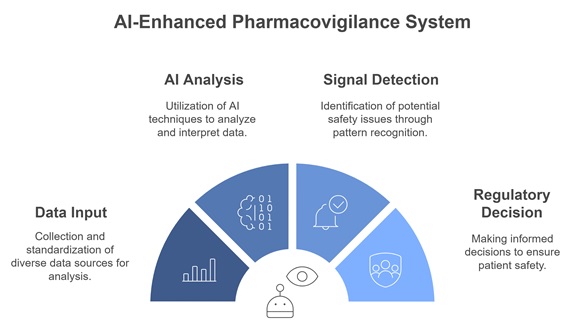
Figure 3: Workflow of an AI-enhanced pharmacovigilance system
4. Challenges and Opportunities in Implementing AI in Pharmacovigilance
The application of artificial intelligence (AI) to pharmacovigilance systems poses several challenges, some of which include the quality of data in the databases. Furthermore, there is a need to have a sizeable dataset in order to train a model, and this patient safety and pharmacovigilance data are completely reliant on spontaneous reports. Moreover, setting up the gold standard in this field is difficult due to the fact that spontaneous reports do not often undergo appropriate confirmation. The implementation of AI in pharmacovigilance comes with several barriers, including harmonisation of standards, data integration of AI with the legacy database, regulatory and legal compliance, as well as the attitudes of medicine regulators and manufacturers. A significant collaboration among various stakeholders is required to address all proposals and establish a prototype framework for implementing AI in pharmacovigilance. AI should also help increase curation efficiency, thus partly addressing the rising demand for resources. Machine learning algorithms should converge to purpose more granular side effect reports, tailored and statistically parameterised to be transformed directly into case reports. This would substantially accelerate AE report integration and, at the same time, streamline data processing in the workforce and tailor resource allocation to their proven added value. AI would eventually deliver systems that are more than mere Health Action Systems in that they add value by delivering the correct answer in less time and with lower operational costs.
Improvement of different AI methodologies should be tested, validated, and have extensive research done through collaborative work among different pharmacovigilance stakeholders and more data. With AI in pharmacovigilance, it is better to show the whole application and have it validated through post-marketing activities to further dilute measurement bias than validating little by little in the application settings. The following Table 3 highlights key challenges in integrating AI into pharmacovigilance and the corresponding opportunities to address them.
Table 3: Key Challenges and Opportunities in AI-Driven Pharmacovigilance
| Challenges | Opportunities |
| Data Privacy Concerns | Development of robust regulatory frameworks to ensure data security. |
| Algorithm Bias | Creation of advanced, unbiased algorithms through diverse datasets. |
| Integration with Legacy Systems | Opportunities to modernise and streamline existing pharmacovigilance systems. |
| Regulatory and Ethical Issues | Establishment of clear guidelines and global harmonisation efforts. |
| Data Quality and Standardisation | Implementation of AI-driven tools for data cleaning and harmonisation. |
| High Implementation Costs | Cost reductions through scalable cloud-based AI solutions. |
| Limited AI Expertise | Training programs and interdisciplinary collaborations to enhance skills. |
5. Future Directions and Potential Impact of AI in Pharmacovigilance
The evolution of AI in the world contributes an unprecedented opportunity for the pharmacovigilance system. The background indicates that the basic feature of AI is the ability to continuously learn and adapt based on continuously detected signals. To this end, a new generation of continuous learning algorithms can be imagined that can continuously refresh their pharmacovigilance knowledge based on the latest data points available. The learned associations made can be structured around safety and efficacy. A new era of continuous learning algorithms and extendable computing platforms can now be combined with simpler continuous learning agents to perform AI-powered PRR signals directly from clinical data streams.
It is clear that AI algorithms can create new challenges for regulators who are responsible for ensuring the protection of patient safety. An important consideration is the lack of transparency in AI algorithms which is being researched. The reported technologies may evolve yet further to provide full transparency in associations and record associations in an irreversible digital ledger, making inauthentic changes detectable. In 10 years, we may see a world with many companies sharing pseudonymised information encapsulated in a blockchain ecosystem leading to a single global standard for data and standardised AI behavior. We will be tool providers competing on feature performance rather than ownership of proprietary information. There is a broader strategic view that AI's impact on haphazard pharmacovigilance can extend to drug therapy in general. Some haphazard or "environmental" effects of drugs acting on a population basis can have value in favorable clinical settings with administration titrated based on personalized drug parties and may drive toward such an adjustment. This would be one response to demonstrate how AI pharmacovigilance applications would eventually converge into an AI personalised medicine platform. Clearly, the attendant ethical questions would demand explicit consideration.
6. Conclusion
In conclusion, the introduction of artificial intelligence into pharmacovigilance is transforming drug safety and monitoring. AI technologies can streamline data processing, detect adverse drug reactions more effectively, and support real-time decision-making, ultimately improving patient safety. Despite challenges such as data privacy concerns and the need for regulatory adaptation, the future of pharmacovigilance looks promising with AI at its core. As AI continues to evolve, it will play an increasingly vital role in shaping the future of drug safety and ensuring better healthcare outcomes globally.
References:
1. Zehravi, M., Maqbool, M., & Ara, I. (2021). An overview about safety surveillance of adverse drug reactions and pharmacovigilance in India. The Indian Journal of Nutrition and Dietetics, 58(3), 408-18. researchgate.net
2. Hamid, A. A. A., Rahim, R., & Teo, S. P. (2022). Pharmacovigilance and its importance for primary health care professionals. Korean Journal of Family Medicine. nih.gov
3. Montané, E. & Santesmases, J. (2020). Adverse drug reactions. Medicina Clínica (English Edition). [HTML]
4. Sasuphan, S., & Ullah, M. F. (2023). Pharmacovigilance Monitoring of Herbal and Traditional Medicine Products: A Survey of Challenges and Solutions. Contemporary Issues in Behavioral and Social Sciences, 7(1), 16-34. researchberg.com
5. Sree Sudha, T. Y., Meena, B., Pareek, S., & Singh, H. (2024). Enhancing pharmacovigilance for robust drug safety monitoring: addressing underreporting and collaborative solutions. Therapeutic Advances in Drug Safety, 15, 20420986241285927. sagepub.com
6. Alomar, M., Tawfiq, A. M., Hassan, N., & Palaian, S. (2020). Post marketing surveillance of suspected adverse drug reactions through spontaneous reporting: current status, challenges and the future. Therapeutic advances in drug safety, 11, 2042098620938595. sagepub.com
7. Singh, R. P., Hom, G. L., Abramoff, M. D., Campbell, J. P., & Chiang, M. F. (2020). Current challenges and barriers to real-world artificial intelligence adoption for the healthcare system, provider, and the patient. Translational Vision Science & Technology, 9(2), 45-45. arvojournals.org
8. Mudgal, S. K., Agarwal, R., Chaturvedi, J., Gaur, R., & Ranjan, N. (2022). Real-world application, challenges and implication of artificial intelligence in healthcare: an essay. Pan African Medical Journal, 43(1). ajol.info
9. Haakman, M., Cruz, L., Huijgens, H., & Van Deursen, A. (2021). AI lifecycle models need to be revised: An exploratory study in Fintech. Empirical Software Engineering, 26(5), 95. springer.com
10. Xu, Z. (2022). Human judges in the era of artificial intelligence: challenges and opportunities. Applied Artificial Intelligence. tandfonline.com
11. Agbehadji, I. E., Awuzie, B. O., Ngowi, A. B., & Millham, R. C. (2020). Review of big data analytics, artificial intelligence and nature-inspired computing models towards accurate detection of COVID-19 pandemic cases and contact tracing. International journal of environmental research and public health, 17(15), 5330. mdpi.com
12. Mohamadou, Y., Halidou, A., & Kapen, P. T. (2020). A review of mathematical modeling, artificial intelligence and datasets used in the study, prediction and management of COVID-19. Applied Intelligence. springer.com
13. Pham, Q. V., Nguyen, D. C., Huynh-The, T., Hwang, W. J., & Pathirana, P. N. (2020). Artificial intelligence (AI) and big data for coronavirus (COVID-19) pandemic: a survey on the state-of-the-arts. IEEE access, 8, 130820-130839. ieee.org
14. Salas, M., Petracek, J., Yalamanchili, P., Aimer, O., Kasthuril, D., Dhingra, S., ... & Bostic, T. (2022). The use of artificial intelligence in pharmacovigilance: a systematic review of the literature. Pharmaceutical medicine, 36(5), 295-306. pharmacovigilance.institute
15. Sadaf, D. S., & Sameer, S. (2023). PHARMACOVIGILANCE AND ADVERSE EVENT DETECTION: A COMPREHENSIVE REVIEW OF ARTIFICIAL INTELLIGENCE APPLICATIONS. EUROPEAN JOURNAL OF MODERN MEDICINE AND PRACTICE, 3(12), 97-104. inovatus.es
16. Oluwaseyi, J. (2024). AI in Predicting Adverse Drug Reactions: Enhancing Patient Safety. easychair.org
17. Ahire, Y. S., Patil, J. H., Chordiya, H. N., Deore, R. A., & Bairagi, V. A. (2024). Advanced Applications of Artificial Intelligence in Pharmacovigilance: Current Trends and Future Perspectives. Journal of Pharmaceutical Research, 23(1), 24. amazonaws.com
18. Kalita, M. J. K., Bora, M. G., Mohan, M. H. R., & Das, P. J. (2024). Innovations in Pharmacovigilance: Harnessing AI for Enhancing Drug Safety and Efficacy. Modern Trends in Multi-Disciplinary Research (Vol-1). academia.edu
19. Khinvasara, T., Tzenios, N., & Shanker, A. (2024). Post-market surveillance of medical devices using AI. Journal of Complementary and Alternative Medical Research, 25(7), 108-122. article2publish.com
20. Hauben, M. (2023). Artificial intelligence and data mining for the pharmacovigilance of drug–drug interactions. Clinical Therapeutics. clinicaltherapeutics.com
21. Gamaleldin, M., Abdelhalim, S., & Abraham, I. (2024). Perspective Chapter: Application of Artificial Intelligence in Pharmacovigilance Practices. intechopen.com
22. Egon, K. & KARL, L. (2023). Machine Learning in Drug Safety Monitoring: Enhancing Pharmacovigilance Efforts. researchgate.net
23. Maliha, G., Gerke, S., Cohen, I. G., & Parikh, R. B. (2021). Artificial intelligence and liability in medicine: balancing safety and innovation. The Milbank Quarterly. nih.gov
24. Mennella, C., Maniscalco, U., De Pietro, G., & Esposito, M. (2024). Ethical and regulatory challenges of AI technologies in healthcare: A narrative review. Heliyon. cell.com
25. Van Kolfschooten, H. (2022). EU regulation of artificial intelligence: Challenges for patients' rights. Common Market Law Review. ssrn.com










