Tiny Vesicles, Big Impact
Unlocking Exosomes in cancer chemotherapy
Disha Marwaha, Institute of Pharmaceutical Research, GLA University
Neha Singh, Lloyd School of Pharmacy
The rapidly evolving pharmaceutical industry frequently adopts innovative concepts in microscopic packaging. Exosomes, once regarded as cellular waste, are now the subject of intense investigation. Pharmaceutical experts are diligently striving to comprehend the potential of these tiny messengers in laboratory and clinical trials. With the progression of precision medicine, exosomes may emerge as a prevalent instrument.
Introduction
Exosomes have emerged as a promising area of research with significant potential in drug delivery and diagnosis in cancer. These tiny particles, previously regarded as cellular waste, are now acknowledged for their crucial function in intercellular communication and their ability to transport molecular cargo, such as nucleic acids and proteins.
These vesicles, typically ranging from 30 to 150 nm in size, derive from the endosomal pathway and possess a lipid bilayer that encompasses biomolecules such as proteins, lipids, and nucleic acids. Their biocompatibility, ability to traverse biological barriers, and minimal immunogenicity render them exceptionally appropriate for drug delivery, especially in cancer treatment. Exosome-mediated drug delivery is emerging as a potential technique owing to their inherent capacity to target specific cells, facilitated by surface proteins and ligands that recognise and bind to these target cells.
Engineered exosomes, as an innovative drug delivery platform, may enhance the efficacy of anticancer therapies while mitigating side effects associated with treatments like chemotherapy and radiotherapy. Intravenous administration of free chemotherapeutics is the predominant method of systemic cancer therapy[1]. Nonetheless, the limited targeting capability of this strategy typically results in a range of therapy-related adverse effects, such as leukocytopenia, hypothyroidism. Engineered exosomes have garnered heightened interest as a novel drug delivery platform to address the aforementioned concerns [2].
Mechanistic Insights into Exosome formation and cargo loading
Exosomes are diminutive extracellular vesicles (EVs) derived from the endosomal system. Their biogenesis entails several stages, commencing with the production of early endosomes by plasma membrane invagination, thereafter maturing into multivesicular bodies (MVBs). In this process, the inward folding of the MVB membrane generates intraluminal vesicles (ILVs), which transform into exosomes upon the fusion of MVBs with the plasma membrane, subsequently releasing them into the extracellular environment. The synthesis of exosomes is governed by the endosomal sorting complexes required for transport (ESCRT) mechanism, essential for the sorting of cargo molecules, including proteins, nucleic acids, and lipids, into intraluminal vesicles (ILVs) [3].
The release of exosomes is meticulously regulated by cellular signalling pathways and fluctuates according to the cell's physiological condition. Consequently, exosome biogenesis and secretion are regarded as potential therapeutic targets, as altering these processes could disrupt detrimental interactions between tumour cells and their microenvironment. Exosome biogenesis is a highly dynamic and complex process, characterised by multiple regulatory checkpoints that guarantee for the accurate loading and release of bioactive cargoes.
Recent developments in exosome delivery systems have allowed researchers to modify the cargo loading mechanism for therapeutic applications. Both passive and active loading techniques are employed to integrate bioactive molecules into exosomes. Passive loading depends on the inherent absorption of molecules by exosomes during their formation, whereas active loading methods, including electroporation and chemical alterations, improve the encapsulation effectiveness of larger or charged molecules. Engineered exosomes exhibit significant potential as drug carriers, facilitating site specific targeting, minimising toxicity, and enhancing therapeutic efficacy in preclinical settings. Comprehending the mechanics of cargo selection and loading is crucial for enhancing exosome application in disease therapy, particularly for cancer and neurological disorders[4].
The power of exosomes
Strong biocompatibility with reduced immunogenicity
Exosomes, being naturally occurring vesicles derived from the body's cells, reduce potential risks associated with immunological responses. In contrast to synthetic nanocarriers, exosomes are less prone to induce adverse reactions, rendering them a safer alternative for therapeutic applications. The natural origin of exosomes enables prolonged circulation within the body, hence augmenting their efficacy in drug delivery and diminishing the likelihood of immune clearance. Exosome-based drug delivery systems provide great biodegradability and minimal toxicity, essential for preventing accumulation in the body. Cancer cell-derived exosomes have been investigated for their ability to transport medications directly to tumours, utilising their low toxicity and immunogenicity to enhance precision medicine strategies [5]. The biocompatibility characteristic is crucial for its prospective application in transporting drugs across several physiological barriers, including the blood-brain barrier, which poses significant challenges for conventional drug delivery systems.
Targeted delivery
Another notable benefit of exosomes drug delivery systems is their improved targeting capabilities. Exosomes inherently possess surface proteins and ligands that facilitate their recognition and binding to specific cells or tissues, hence ensuring efficient delivery of therapeutic drugs. This targeting capability can be augmented using bioengineering methods, wherein specific ligands or receptors are integrated into the exosome membrane to boost its affinity for particular tumour cells or sites. This enables exosome-based therapies to deliver drugs precisely to the target region while reducing off-target effects, which is crucial in cancer treatment, as high drug specificity can markedly diminish toxicity to healthy tissues.
Drug stability
Exosomes boost drug stability by safeguarding therapeutic molecules from breakdown in the circulation. The entrapment of pharmaceuticals within the lipid bilayer of exosomes protects them from enzymatic breakdown and adverse biological conditions, enhancing their bioavailability and efficacy. This property has proven especially advantageous for the administration of nucleic acid-based treatments which are highly vulnerable to degradation. Utilising exosomes as carriers enables the intact delivery of these molecules to target cells, hence enhancing their therapeutic efficacy. The inherent capacity of exosomes to maintain drug stability, along with their targeting proficiency, renders them a compelling platform for advanced delivery systems.
Notwithstanding their benefits, tumour-derived exosomes can also potentially stimulate angiogenesis, metastasis, and tumour growth via a variety of pathways. “They transport oncogenic proteins and RNAs that can modify recipient cell phenotypes, breakdown the extracellular matrix to promote invasion, create pre-metastatic niches in remote tissues, and augment angiogenesis through pro-angiogenic factors. Moreover, they can inhibit host immunological responses, facilitating tumour cells' evasion of immune surveillance. The tumour-promoting characteristics provide considerable safety issues when evaluating tumour-derived exosomes as drug delivery systems. Selecting a suitable cellular source for exosome formation is essential to solve these challenges. Progressing exosome-based therapeutics necessitates meticulous screening and alteration of exosome composition to reduce hazards. Methods like genetic engineering or the encapsulation of exosomes with therapeutic compounds to mitigate tumour-promoting factors can optimise treatment efficacy while reducing toxic effects. The subsequent steps for advancing exosome-based drug delivery systems involve standardizing isolation techniques, comprehensive characterisation, and guaranteeing safety in preclinical and clinical evaluations. Engineered exosomes, designed for focused drug delivery and enhanced safety, signify a revolutionary advancement in oncology, facilitating more effective and tailored cancer therapies[6].
The significance of exosomes in the real world is a connection between the scientific community and Society.
In addition to being investigated by researchers in laboratories, exosomes have the potential to bring about a change in the manner in which healthcare is provided in general. There is a possibility that cancer treatments will become not only more effective but also safer if exosomes are utilised. Because of this, patients would experience fewer adverse effects and would have a higher quality of life while they were receiving treatment. It is possible that their application in diagnostics would make early cancer identification easier, which will in turn make it possible to perform life-saving therapies during the most favourable stages of the disease. It is possible that drugs based on exosomes could help reduce the costs of long-term medical care by reducing the need for multiple rounds of potentially harmful chemotherapy and hospital stays. It is safe to say that exosomes are at the forefront of both wellness and new ideas. They claim that they will not only alter cancer treatment, but also the way we think about patients receiving care that is individualised to their needs.
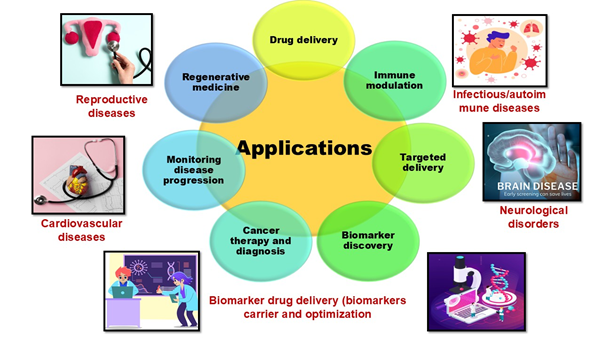
Future perspectives
Recent advancements in exosome research have revealed their considerable potential in diagnosis, therapy, and prevention across several diseases. Exosomes are becoming recognised as significant non-invasive markers for the early diagnosis and monitoring of diseases, including cancer, cardiovascular problems, and neurological disorders. Their capacity to transport illness-specific proteins, RNA, and other molecular markers has facilitated the advancement of liquid biopsy techniques, providing a less intrusive and more accessible approach for clinical screening and monitoring disease progression. Exosomes are being investigated for their function in immunomodulation within the field of prophylaxis. They possess the capacity to elicit immunological responses or modulate immune processes, which is especially pertinent in the realm of vaccines and immunotherapeutic interventions. Exosome-based vaccines have been studied for their capacity to convey antigens to immune cells, potentially improving the effectiveness of cancer immunotherapy and the prevention of infectious diseases.
The recent advancements in exosome research are propelling their utilisation as essential instruments in customised medicine, with considerable ramifications for diagnostics, targeted therapies, and preventive healthcare approaches.
As research on exosomes progresses, they are set to assume a more prominent function in cancer therapy, propelled by their distinctive biological characteristics and adaptability. The emergence of personalized medicine allows for the engineering of exosomes to provide customised therapies according to the unique molecular and genetic characteristics of an individual's tumour. This personalization would provide precisely targeted therapies with less adverse effects and enhanced effectiveness. Isolating exosomes from a patient's own cells minimises the danger of immunological rejection, rendering exosome-based therapeutics a compelling choice for customised cancer treatment.
Furthermore, they may be utilised to develop novel cancer vaccines. Exosomes generated from tumours, which contain tumour-associated antigens, could be modified to elicit an immune response, thereby enabling the patient's immune system to identify and eliminate cancer cells. This exosome-based vaccine strategy and may enhance current immunotherapies such as CAR-T cell therapy and checkpoint inhibitors. Another intriguing future application of exosomes in cancer therapy is their capacity to modify the tumour microenvironment (TME). Exosome-based therapeutics may inhibit cancer development by changing the tumour microenvironment to be less conducive to tumour growth and spread. Exosomes may transport therapeutic chemicals that impede angiogenesis, obstruct communication among cancer cells, or promote immune cell infiltration into tumours, so establishing an inhospitable environment for cancer cell proliferation.
Conclusion
The research on exosome-based drug delivery systems have introduced a revolutionary method for cancer therapy, presenting remarkable potential for personalized medicine, improved therapeutic efficacy, and the capacity to address several enduring challenges of traditional treatments. The distinctive characteristics of exosomes, including their exceptional biocompatibility, capacity to transport diverse therapeutic compounds, and inherent targeting skills, render them an optimal instrument in the battle against cancer. Research has shown that exosomes can be designed to transport chemotherapeutic drugs, gene treatments, and immunomodulators with great specificity to cancer cells, thereby limiting harm to healthy organs and decreasing systemic toxicity. These advances enhance patient outcomes and provide a persuasive argument for the further investigation of exosome-mediated therapeutics in clinical environments. The future of exosome-mediated drug delivery appears promising, with potential applications extending beyond cancer to various other disorders, signifying a new frontier in precision medicine.
References
- Zhang, Y., et al., Recent advances in exosome-mediated nucleic acid delivery for cancer therapy. Journal of nanobiotechnology, 2022. 20(1): p. 279.
- Avgoulas, D.I., et al., Therapeutic and diagnostic potential of exosomes as drug delivery systems in brain cancer. Pharmaceutics, 2023. 15(5): p. 1439.
- Han, Q.-F., et al., Exosome biogenesis: machinery, regulation, and therapeutic implications in cancer. Molecular cancer, 2022. 21(1): p. 207.
- Li, J., J. Wang, and Z. Chen, Emerging role of exosomes in cancer therapy: progress and challenges. Molecular Cancer, 2025. 24(1): p. 13.
- Sadeghi, S., et al., Exosome engineering in cell therapy and drug delivery. Inflammopharmacology, 2023. 31(1): p. 145-169.
- Herrmann, I.K., M.J.A. Wood, and G. Fuhrmann, Extracellular vesicles as a next-generation drug delivery platform. Nature nanotechnology, 2021. 16(7): p. 748-759.











