Combined chemo-photo (photothermal/photodynamic) therapy is gaining attraction as modern art to treat diverse types of cancer. Researchers in the field focusing on the delivery agents that could place the chemo drugs at the site of desire along with producing photothermal effect upon exposure to light irradiation. This not only fastening the release of the drug at site but also killing the cancerous cells employing a sudden rise in temperature from normal to higher (~ 45°C). Such agent includes gold nanoparticles, graphitic carbon-based nanostructures, carbon nano-tubes and silver nanoparticles.
Cancer: Current statistics and Hurdles in treatment
Cancer is one of the leading causes of death in both developed and developing countries and also a major public health problem. This dreadful disease is spreading with continuance and increasing incidences in recent times [1]. In 2012, 8.2 million cancer patients died as per the reports of World Health Organization (WHO) which also estimated the increase in annual cancer cases from 14 million to 22 million within coming 20 years[2].
In 2018, 18.1 million new cancer cases were estimated (excluding non-melanoma skin cancer) and 9.6 million were estimated deaths due to cancer, from which 70 per cent of deaths occur in low and middle income countries. Most commonly diagnosed cancer in both male and females is the lung cancer[3]. Lung cancer is the most common diagnosed cancer (11.6 per cent of total cases) and the leading cause of cancer deaths (18.4 per cent of total deaths due to cancer), closely followed by female breast cancer (11.6 per cent), prostate cancer (7.1 per cent), and colorectal cancer (6.1 per cent) for incidence and colorectal cancer (9.2 per cent), liver cancer (8.2 per cent), and stomach cancer (8.2 per cent) for mortality. Among males, lung cancer, prostate cancer and colorectal cancer are most frequent for incidence and the stomach cancer and liver cancer are most frequent for mortality[4]. Among females, most frequently diagnosed is breast cancer followed by lung and colorectal cancer for incidence and stomach and liver cancer for mortality. Cervical cancer ranks at the fourth in both cases i.e. incidence and mortality in both sexes. The rating of cancers according to most commonly diagnosed and leading to death cases may vary country to country depending upon the degree of economic development, social, life style factors and also eating habits [5].
Some states are home to both the wealthiest and the poorest counties, suggesting the opportunity for more equitable dissemination of effective cancer prevention, early detection, and treatment strategies. A broader application of existing cancer control knowledge with an emphasis on disadvantaged groups would undoubtedly accelerate progress against cancer[6].
Photo therapy is an emerging and noninvasive technique that has been employed for the treatment of several cancer types. However, many hurdles still exist that greatly reduce the efficacy of photodynamic therapy and also impairs its overall performance for the clinical translation[7]. The main challenges faced by conventional phototherapeutic agents include easy recognition by the immune system, low accumulation in target sites and rapid clearance from blood circulation. Tumour associated hypoxia is another important factor responsible for the poor responses in the cancer treatment by photo therapy. Tumour associated hypoxia has also been recognised as a hallmark of solid tumour and also plays a key role in the resistance to conventional anti-cancer therapies[8].
Combined chemo-photo therapy: Need of hour
Chemo-photo therapy for cancer is a phototherapy combined with chemotherapy. Phototherapy including both, photodynamic and photothermal therapies is known to be effective in cancer treatment. Photodynamic therapy is a photochemistry based therapy, it utilises photosensitisers which are based on light energy to produce cytotoxic species by transfer of electrons to the biological substrates and to transfer energy to molecular oxygen [9]. Whereas, photothermal therapy utilises those substances which have ability to convert the light energy into heat energy for the efficient tumour excision. Due to its localised and selective laser-induced therapeutic effect photothermal therapy promotes effective treatment for cancer[10]. Light-induced heat in photothermal therapy increases thetemperature of body above normal of the irradiated area and destroys malignant tumor’s. In treatment of cancer, heating at high temperature i.e. 80°C can ablate the tumour, whereas, heating at the range of 41-45°C may achieve death of cancer cells without causing any serious injury to the adjoining normal tissues. Alternatively, at 39–41°C temperature fever-range effects canbe achieved do not damage any tissues[11].
Photodynamic therapy is an alternative option for tumour excision and function sparing oncologic intervention. The treatment modality includes administration of tumour-localising photosensitiser (PS), which is illuminated locally with light that activates the photosensitiser to generate cytotoxic molecules including free radicals and singlet oxygen. When used individually, light, photosensitiser and oxygen are non-toxic to the cells, but when used incombination, may generate high reactive singlet oxygen that are capable to destroy cancer cells[12].
Discussion on current research outcomes: Mapping the future road
Cancer phototherapy require the development of biomimetic cell membrane-camouflages nanoparticles [13]. Phototherapeutic nanoparticles can be cell membrane coated which may be equipped with some additional physiological features such as immune escape, homologous targeting or in-vivo prolongs circulation time depending on the cell sources. However, cell membrane coated on phototherapeutic nanoparticles can be removed under laser irradiation, leading to drug release and hence, known as synergetic therapy. By combining other supplementary agents to normalise tumor microenvironment, cell-membrane coating can further enhance the therapeutic efficacy against cancer[1, 14].
Tan et al.,2019 developed a strategy based on photothermal stimulation for mitochondria responsive drug release along with heat shock, to maximise the antitumour effects with minimum side effects. Their strategy depends upon mitochondrial targeted delivery of doxorubicin (DOX) through a lipophilic and photothermal agent IR-780 iodide (IR780)-modified glycolipid conjugates (CSOSA), which have capacity to kill the tumour cells by triggering high level reactive oxygen species synergistically. The study found that the photothermal conversion by IR780-CSOSA can weaken the hydrophobic interaction between DOX and the core of micelles. It can release DOX in mitochondria by triggering micelle swelling for ROS amplification and can also promote the fast evolution of ROS at the same locus by inducing mitochondria specific heat shock to eradicate cancer cells in more efficient way. Other than this study also revealed that IR780-CSOSA micelles may independently realise the real-time diagnosis and imaging on multiple tumor models and under laser irradiation, deep penetration into tumors by IR780-CSOSA/DOX micelles can be manipulated [15]. Figure 1 shows characterisation of IR780-CSOSA micelles.
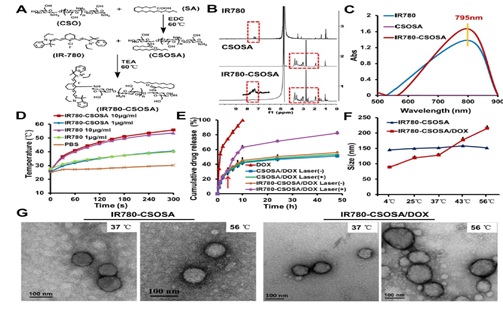
Figure 1: (A) Synthesis of IR780-CSOSA. (B) 1H NMR spectra of IR780, CSOSA and IR780-CSOSA. (C) UV-vis spectra of IR780, CSOSA and IR780-CSOSA solution. (D) Photothermal effect of IR780-CSOSA and free IR780 under laser irradiation (808nm, 1W/cm2) for 5 min. (E) In vitro NIR-triggered drug release profiles. (F) Size alteration of IR780-CSOSA and IR780-CSOSA/DOX micelles at varying temperatures. (G) Transmission electron microscope (TEM) images of IR780-CSOSA and IR780-CSOSA/DOX micelles at 37 °C and 56 °C.Open access article.
In cancer therapy PEGylated graphene oxide nanomaterials have shown promising results due to their photothermal capacity and drug loading capacity. Melo-diogo et al.,(2018)reported the use of POxylated graphene oxide nanomaterials for the combination chemo-photo therapy of breast cancer cells. They reported that POxylated graphene oxide shows the properties required for application in cancer therapy. In breast cancer cells an optimal therapeutic effect was produced by 1:3 DOX:TOS molar ratio. This drug ratio showed low impact on the normal cells. A strong therapeutic effect was induced by the delivery of DOX:TOX by POxylated graphene oxide to the cancer cells. The study concluded POxylated GO as a promising phototherapeutic and drug delivery agent[16].
Li et al,. 2018 developed organic multimodal phototheranosticnanoagents i.e. semiconducting polymer nanoparticles coated with cell membrane which can biomimetically target cancer associated fibrolasts in tumour microenvironment for enhanced multimodal imaging guided cancer therapy.
Such biomimetic nanocamouflages comprise a near-infrared (NIR)absorbing semiconducting polymer nanoparticle (SPN) coated with the cell membranes ofactivated fibroblasts. The mechanism of homologous targeting allows the activated fibroblast cell membrane coated SPN (AF-SPN) to target cancer associated fibroblasts specifically which may lead to increased tumour accumulation relative to the uncoated and cancer cell membrane coatedcounterparts after systemic administration in living mice. AF-SPN together with providing strong photoacoustic (PA) signals and NIR fluorescence for tumour detection, also generate huge amount of cytotoxic geat and single oxygen to produce combinational photodynamic and photothermal therapy which ultimately leads to higher antitumour efficacy than the counterparts[17].
References
[1] Kalyane D, Raval N, Maheshwari R, Tambe V, Kalia K, Tekade RK. Employment of enhanced permeability and retention effect (EPR): Nanoparticle-based precision tools for targeting of therapeutic and diagnostic agent in cancer. Materials Science and Engineering: C 2019.
[2] McGuire S. World cancer report 2014. Geneva, Switzerland: World Health Organization, international agency for research on cancer, WHO Press, 2015. Oxford University Press; 2016.
[3] Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA: a cancer journal for clinicians 2018;68:7-30.
[4] Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA: a cancer journal for clinicians 2019;69:7-34.
[5] Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a cancer journal for clinicians 2018;68:394-424.
[6] Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA: a cancer journal for clinicians 2016;66:7-30.
[7] Mahajan S, Patharkar A, Kuche K, Maheshwari R, Deb PK, Kalia K, et al. Functionalized carbon nanotubes as emerging delivery system for the treatment of cancer. International journal of pharmaceutics 2018;548:540-58.
[8] Damyanov C, Maslev I, Pavlov V, Avramov L. Conventional treatment of cancer realities and problems. Ann Complement Altern Med 2018; 1 (1) 2018;1002.
[9] Lucky SS, Soo KC, Zhang Y. Nanoparticles in photodynamic therapy. Chemical reviews 2015;115:1990-2042.
[10] Zhang Y, Xu S, Li X, Zhang J, Sun J, Xia H, et al. Fabrication, photothermal conversion and temperature sensing of novel nanoplatform-hybrid nanocomposite of NaYF4: Er3+, Yb3+@ NaYF4 and Au nanorods for photothermal therapy. Materials Research Bulletin 2019;114:148-55.
[11] Toraya-Brown S, Fiering S. Local tumour hyperthermia as immunotherapy for metastatic cancer. International Journal of Hyperthermia 2014;30:531-9.
[12] Muhanna N, Chan H, Townson JL, Jin CS, Ding L, Valic M, et al. Photodynamic therapy enables tumor specific ablation in preclinical models of thyroid cancer. Endocrine-related cancer 2019;1.
[13] Soni N, Soni N, Pandey H, Maheshwari R, Kesharwani P, Tekade RK. Augmented delivery of gemcitabine in lung cancer cells exploring mannose anchored solid lipid nanoparticles. Journal of colloid and interface science 2016;481:107-16.
[14] Berger D, Winterhalter B, Fiebig H. Conventional chemotherapy. The Nude Mouse in Oncology Research 2018:165-84.
[15] Tan Y, Zhu Y, Wen L, Yang X, Liu X, Meng T, et al. Mitochondria-responsive drug release along with heat shock mediated by multifunctional glycolipid micelles for precise cancer chemo-phototherapy. Theranostics 2019;9:691.
[16] de Melo-Diogo D, Costa EC, Alves CG, Lima-Sousa R, Ferreira P, Louro RO, et al. POxylated graphene oxide nanomaterials for combination chemo-phototherapy of breast cancer cells. European Journal of Pharmaceutics and Biopharmaceutics 2018;131:162-9.
[17] Li J, Zhen X, Lyu Y, Jiang Y, Huang J, Pu K. Cell membrane coated semiconducting polymer nanoparticles for enhanced multimodal cancer phototheranostics. Acs Nano 2018;12:8520-30.