Nanocarriers in Breast Cancer Therapy
Myth or miracle?
Nalla Usha Kumari, Department of Pharmaceutics, National Institute of Pharmaceutical Education and Research
Neelesh Kumar Mehra, Department of Pharmaceutics, National Institute of Pharmaceutical Education and Research.
Breast cancer (BC) is a genetically and clinically heterogeneous cancer with diverse molecular, immunochemical, morphological, and clinicopathological features with different responses to therapy. Cautious harmonisation of nanocarriers can improve BC therapy by reducing the adverse effects, metastasis, and relapse. Nevertheless, how far this advanced technology serves the challenges posed by the heterogeneity of BC is still a matter of ongoing research and development. This article emphasises the current position of nanocarriers in BC therapy and provides future suggestions for the clinical translation of nanocarriers.
Breast cancer is a formidable public health challenge in the world. Based on origin, BC is divided into carcinomas (which develop from the terminal ducts and lobules of the breast) and sarcomas (which develop from connective tissues that support ducts and lobules). Adenocarcinomas represent 95 per cent of BC cases, and triple negative BC is the most molecularly diverse and biologically aggressive subtype of BC with the lack of ER, PR, and HER2 overexpression. It affects younger and premenopausal women and has high metastatic potential. Recent advancements in BC therapy, such as radiotherapy, endocrine treatment, surgical techniques, and chemotherapy, have improved the survival of patients. Surgery is ineffective in case of recurrence and metastasis. Conventional chemotherapy is unable to offer selective therapy, leading to off-target effects and toxicity, whereas radiotherapy affects neighbouring healthy cells. 20-30 per cent of the patients develop metastatic cancer, and One of the main reasons are tumour heterogeneity, drug resistance, and chemotherapy failure. Keeping these in mind, novel approaches are required to tackle BC effectively.
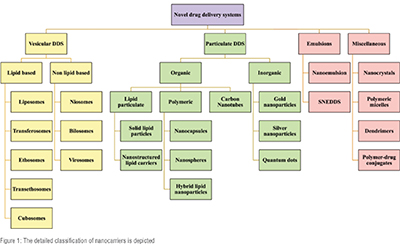
Nanocarriers are colloidal dispersions of drug carriers in the submicron range with exclusive properties like a high surface area to volume ratio, favourable pharmacokinetics, etc., that increase the efficacy and reduce the toxicity of the chemotherapeutics while conquering biological barriers and overcoming drug resistance. They are of vesicular, particulate, emulsion, and miscellaneous systems. The detailed classification of nanocarriers is depicted in Figure 1. Vesicles are typically made up of a polymer backbone with properties allowing for self-assembly into vesicular structures. These drug delivery systems include nanocarriers based on lipids and non-lipids, capable of loading hydrophilic and lipophilic substances for controlled drug delivery. The term "particulate system" encompasses a broad range of nanoscale structures, including polymeric, lipid-based, micellar nanoparticles, dendrimers, quantum dots, carbon nanotubes, nanorods, and nanoparticles made of silver, gold, or iron. These systems, composed of natural and synthetic polymers or other biomaterials, offer significant benefits across various applications in regenerative medicine and drug delivery. The nanocarriers exhibit advantages like reduced drug dose, off-target effects, degradation and improved solubility, pharmacokinetic parameters, therapeutic efficacy, increased stability, controlled drug release, etc. Various chemotherapeutics, including natural products, vaccines, proteins, mRNA, siRNA, DNA, diagnostic agents, etc., can be delivered to the targeted site using nanocarriers. These nanocarriers provide targeted therapy and protect the encapsulated components from degradation. Further, targeted therapy with spatial and temporal control on the drug release decreases the drug dose and improves therapeutic efficacy. (Figure 1)
Nanocarriers: Myth or Miracle?
With the advances in nanotechnology, nanocarriers are being tailored to obtain optimal therapeutic properties while reducing unwanted side effects. Nanocarriers exhibit accumulation at the tumour site by extravasating leaky vasculature of the tumour (EPR effect) and reducing the off-target effects on healthy cells. The EPR effect is expected to increase the accumulation of nanocarriers by 20-30 per cent, and transcellular pathways also improve the nanoparticle's extravasation into the tumour site. They reduce the system toxicity of the drug; for instance, liposomal doxorubicin exhibited lower cardiotoxicity than free doxorubicin. The physicochemical properties of the nanoparticles, as well as the pathophysiological and pathoanatomical features of the tumours, influence the extent of tumour accumulation. However, the heterogeneity of the vascular architecture, multidrug resistance etc., remain challenging. More strategies are required to improve the effectiveness of the nanocarriers by pharmacological and physical co-treatments through active targeting/stimuli-responsive nanocarriers or a combination of nanocarriers with immunotherapy. Active targeting uses the interaction between surface-modified nanocarriers that bind with the receptors overexpressed at the target site. FunctionaliSation of nanocarriers with ligands like folic acid, hyaluronic acid, antibodies, aptamers, etc., whose receptors are overexpressed or selectively expressed at tumor site can improve the internaliSation of the nanocarriers into the cancer cells. Investigations have revealed that tumour cells exhibit greater uptake of hyaluronic acid-coated nanoparticles than normal cells. Considering the BC subtype, various active ligands can be used to modify the nanocarrier surface to enable active tumour targeting. Epidermal growth factor receptor (EGFR) targeted nanoparticles displayed greater uptake of nanocarriers by EGFR-expressing BC cells than the EGFR-negative BC cells. The folate receptor is overexpressed in TNBC, and the folate-conjugated microspheres exhibited a significant reduction in the tumour volume in the BALB/C mice model compared to the non-targeted microspheres.
Over the past two decades, extensive research has focused on stimuli-responsive nanocarriers that are responsive to intrinsic (pH, redox, hypoxia, temperature, etc.) or extrinsic (ultrasound, light, magnetic, etc.) stimuli and can produce a rapid and controlled release of the drug upon the stimulus. Contrary to the normal tissue, the tumour environment has lower pH, overexpressed enzymes, high glutathione concentration, etc., which are being exploited to rationally design different responsive structures, from polymers to hybrid nanocarriers. Upon exposure to the stimuli, these modified systems release the drug through various mechanisms. Hybrid nanocarriers are nanocomposites of two or more different materials (polymer and inorganic- or organic-based systems) that have superior properties combining the advantages of the individual components while overcoming the individual shortcomings. In our lab, we have fabricated ligand-conjugated pH-responsive chitosan-lecithin hybrid nanocarriers for stimuli-responsive targeted delivery of palbociclib to breast cancer. The hybrid nanoparticles exhibited pH-dependent drug release, greater endocytosis, and hindered cellular migration. Notably, hybrid nanocarriers are being investigated for theranostic purposes to achieve more significant therapeutic potential.
Despite the preclinical success of nanocarriers in BC therapy, their translation has exhibited limited success. Despite the advantages of improved solubility, permeability, bioavailability, circulation time, targeted delivery, etc., nanocarriers still have limitations like complex manufacturing procedures, challenges with quality control, scalability, reproducibility, characterisation tech niques, nanocarrier stability, etc., which pose challenges in translation to market. Furthermore, the opsonisation of nanocarriers, immune reactions, drug leakage, toxicity of excipients, etc., are some of the other challenges. One of the reasons for in vitro–in vivo discrepancy is the change in the physicochemical properties of nanocarriers after entering the biological system due to their interaction with the biomolecules. These interactions give a new biological identity different from the pristine nanocarriers, which impacts the nanocarriers' biodistribution, release kinetics, targeting ability, stability, etc.
The current limitations, such as stability, limited translational potential, in vitro-in vivo discrepancy, and knowledge gaps, must be addressed for the clinical translational of nanocarriers. Carrier-free nanocarriers are one option for minimising polymer toxicity, and they have high drug loading with low systemic toxicity. The balance of nanocarrier properties and tumour conditions is essential for delivering the loaded cargo to the intended site. A thorough understanding of nanocarrier properties is required for rational nanocarrier design and structural optimisation. The biomolecular interactions of nanocarrier with the biological milieu determine its fate in the body. Similarly, pathoanatomical features, tumour microenvironment, physiological process, etc., should be considered while designing nanocarriers for accurate delivery to the tumour site. One practical approach for replicating a tumour microenvironment involves utilising patient-derived xenografts to investigate how nanocarriers accumulate, extravasate, and selectively target tumours.
In summary, nanomedicine has technical limitations and is in its infancy. This novel technology is expected to transform BC therapy and improve the survival of patients. The balance between conducting thorough research on nanocarriers to ensure their efficacy and safety is crucial for facilitating regulatory approval processes. A deeper understanding of the physicochemical nature of nanocarriers and physiological conditions may result in fruitful outcomes in the clinical translation of nanocarriers and BC treatment.













