Transforming Clinical Trials
Harnessing wearables for Real-World Data (RWD) and Real-World Evidence (RWE) collection and analysis
Rashmi Gurnani, CEO, AKT Health Analytics
Wearable technology is redefining clinical trials by enabling continuous, real-time collection of real-world data (RWD). This innovation enhances the generation of real-world evidence (RWE), improving patient-centricity, data accuracy, and trial efficiency. As adoption grows, wearables promise to accelerate decision-making, reduce costs, and shape the future of evidence-based healthcare.

Clinical trials are at a pivotal juncture in 2025. For decades, clinical trials have largely relied on controlled environments and episodic measurements that do not fully reflect the variability of patient behaviour and treatment effects in the real world. With the rapid advancement of wearable technologies—ranging from smartwatches and biosensors to continuous glucose monitors and patches—we are now capable of collecting high-quality, real-time health data outside clinical settings. These data streams, termed real-world data (RWD), are generating real-world evidence (RWE) that has the potential to reshape how therapies are developed, validated, and monitored post-launch.
As someone involved in both clinical research and digital health strategy, I have closely followed and supported the integration of wearables into decentralised clinical trials. This article outlines the landscape, use cases, and scientific knowledge required to convert wearable data into actionable RWE.
Understanding Wearables as RWD Engines
Wearable technologies—such as smartwatches, biosensors, fitness trackers, and skin patches—are revolutionising how we collect health data in clinical research. These devices enable continuous, real-time monitoring of vital physiological and behavioural parameters, including heart rate, oxygen saturation, respiratory patterns, sleep quality, activity levels, and more. Unlike traditional trial methods that rely on infrequent, clinic-based measurements, wearables provide a more comprehensive and patient-centric view of health over time.
Real-world data (RWD) generated by wearables reflects how patients function in their natural environments. This enhances the quality and applicability of real-world evidence (RWE), supporting outcomes that are more representative of everyday life. For instance, a smartwatch can detect atrial fibrillation episodes outside of clinic hours, while an activity tracker can capture decline in mobility in neurodegenerative conditions—both offering early intervention opportunities.
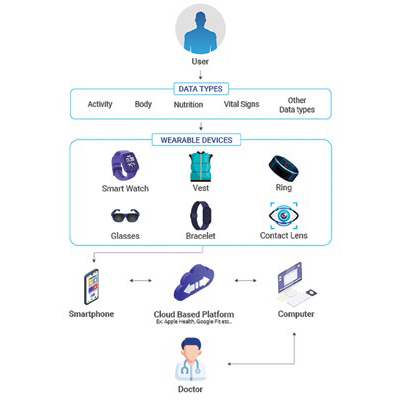
Critically, these data are passive and non-invasive, reducing patient burden and improving trial compliance. Wearables also empower decentralised and hybrid trial models, allowing remote participation and expanding access to diverse populations. As long as the devices are clinically validated and integrated into robust data ecosystems, wearables can serve as reliable engines for capturing RWD that fuels stronger, more meaningful RWE.
Applications in current clinical trials
a. Cardiovascular Trials
Apple’s Heart Study, involving over 400,000 participants, utilised the Apple Watch to detect atrial fibrillation (Perez et al., 2019). The ability to capture heart rate irregularities in daily life significantly improved early detection and patient engagement.
b. Neurology & Parkinson’s Disease
In Parkinson’s trials, wearables like the Verily Study Watch or GaitUp sensors have captured tremor severity, stride length, and bradykinesia with high reliability (Arora et al., 2021). These measures offer more frequent and objective assessments compared to clinic-based scales.
c. Sleep and Respiratory Conditions
Wearable oximeters and actigraphy bands help monitor nocturnal desaturation in obstructive sleep apnea or COPD patients. Real-world measurements provide better insight into night-to-night variability and therapy adherence.
d. Oncology Supportive Care
Activity trackers are increasingly used to monitor fatigue and mobility changes in cancer patients undergoing chemotherapy. These metrics correlate strongly with patient-reported quality of life and treatment tolerance.
Regulatory Considerations and Data Quality
As wearable technologies become more prominent in clinical trials, regulators are beginning to embrace their potential—but with caution as there is limited information on devices available. Agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) now recognise the value of RWD and RWE in supporting regulatory decisions. The FDA's Digital Health Center of Excellence has been instrumental in developing frameworks that support the use of wearables for safety monitoring, endpoint generation, and decentralised trial models.
However, regulatory acceptance is reliant on rigorous validation. Devices must undergo both analytical validation—to ensure accurate, consistent readings—and clinical validation—to confirm their data meaningfully reflect health outcomes. For example, a wearable that measures heart rate must demonstrate reliability across populations and conditions, and its output must correlate with clinical endpoints.
Data quality is equally critical. Wearable data must be secure, auditable, and compliant with Good Clinical Practice (GCP). Sponsors must also ensure informed consent procedures clearly explain data usage, especially given privacy laws like GDPR and HIPAA. Additionally, handling data loss due to device non-compliance or connectivity issues requires robust protocols and contingency plans. Only through disciplined governance can wearable data be deemed credible for regulatory use.

Data management and analytics
Wearable data arrives in high frequency (e.g., minute-by-minute heart rate or movement etc). Managing this requires:
1. Scalable infrastructure
Cloud-based platforms that can ingest, filter, and standardise continuous streams of data as huge amounts of data is passing to the servers.
2. Pre-processing and feature extraction
Algorithms must identify meaningful events—such as tremors, arrhythmia, or sleep cycles—from raw accelerometer or photoplethysmography (PPG) signals.
3. AI and machine learning
Machine learning models can identify digital biomarkers and even predict flare-ups in chronic diseases like heart failure or Multiple Sclerosis.
4. Statistical RWE Modelling
Time-to-event analyses, causal inference techniques, and mixed models can be used to evaluate endpoints derived from wearables.
Designing wearable-enabled clinical trials: Lessons learned
From some of the companies that have executed decentralised clinical trials, below are some essential recommendations:
● Selection of the right device: Align device capabilities with the clinical question. For instance, if step count is a proxy for functional capacity, choose a validated accelerometer with proven accuracy.
● Pilot first/beta testing: Short feasibility studies help uncover technical issues, participant confusion, or data gaps.
● Train participants effectively: Provide digital and printed guides. Include caregivers where necessary, especially for elderly patients.
● Real-time monitoring: Use dashboards to flag non-compliance or signal deterioration early.
● Define data completeness thresholds: Specify what constitutes acceptable levels of data received (e.g., 80 per cent daily wear-time) to maintain dataset integrity.
Benefits vs challenges
The integration of wearable technologies into clinical trials offers a wide range of advantages that are reshaping research methodologies. Most notably, wearables make studies more patient-centric by enabling data collection in everyday environments, reducing the need for frequent clinic visits. This convenience can enhance patient engagement, adherence, and retention. Moreover, continuous monitoring captures fluctuations in health metrics more accurately, allowing for early detection of adverse events or disease progression. This leads to more sensitive and dynamic endpoints, often requiring smaller sample sizes and offering cost efficiencies.
However, despite these promising benefits, significant challenges remain. One major hurdle is ensuring the usability of wearable devices across diverse patient populations, particularly among older adults or individuals in rural settings with limited access to technology. Data quality and consistency can be affected by poor device compliance, signal interruptions, or firmware updates. Additionally, the massive volume of data generated demands robust infrastructure and sophisticated analytics for meaningful interpretation. Regulatory pathways are still evolving, and sponsors must navigate uncertainty around the acceptability of wearable-derived endpoints. Balancing innovation with scientific rigor, patient privacy, and regulatory compliance remains critical. Only through thoughtful design and validation can the full promise of wearables in clinical trials be realised.
Case studies
1. The VascTrac Trial for Heart Failure
Patients wore wrist-based trackers post-discharge. A 500-step drop in daily activity predicted hospitalization within two weeks (Wang et al., 2022).
2. Parkinson’s mPower app study
Smartphone-based tests (e.g., voice, tap tests) coupled with wearable sensors accurately captured fluctuations in symptoms and motor instability over time (Bot et al., 2016).
3. Long-COVID digital cohorts
In a UK-based digital health study, reduced sleep quality and sustained high heart rate from wearable data corresponded to persistent post-viral fatigue symptoms (Sudre C. H. et al., 2021).
Future outlook
The evolution of wearables in clinical research is just beginning. There are several developments that are anticipated , such as:
● Multi-modal sensors: Devices that capture ECG, temperature, hydration, and even biochemical data like glucose or cortisol.
● Digital biomarkers for precision trials: AI-driven insights will define disease subtypes and predict responders, reducing trial heterogeneity.
● Hybrid study designs: Combining remote wearable data with occasional site visits to blend the best of both models.
● Label expansion based on RWE: As regulators gain confidence, we will see broader label extensions based on wearable-enabled studies, especially in post-marketing settings.
Conclusion
Wearables are no longer peripheral tools— they are becoming central to how modern clinical trials are conceived, conducted, and analysed. By enabling continuous, real-world data capture, these technologies offer an unprecedented level of insight into patient experiences outside traditional trial settings. This real-time data enhances the relevance and sensitivity of trial endpoints, supports earlier safety signal detection, and helps evaluate therapies more holistically, particularly in chronic and progressive diseases.
However, realising the full potential of wearables in clinical trials demands more than just adopting new technology. It requires robust validation of devices, transparent and ethical data governance, and alignment with evolving regulatory standards. The data collected must be accurate, interpretable, and applicable to regulatory and clinical decision-making. Furthermore, ensuring inclusivity and usability across patient populations will be key to avoiding disparities in participation and outcomes.
As the healthcare and pharmaceutical industries continue to evolve towards more personalized and digital-first approaches, wearable technology will play a foundational role. When thoughtfully integrated into trial designs, wearables can bridge the gap between controlled clinical environments and the realities of patient life—ultimately delivering stronger real-world evidence, improving patient outcomes, and accelerating innovation across the drug development lifecycle. All we wait for now is for the right regulations to come through for successful RWD and RWE generation.
References
1. Perez, M. V., Mahaffey, K. W., Hedlin, H., Rumsfeld, J. S., Garcia, A., Ferris, T., ... & Turakhia, M. P. (2019). Large-scale assessment of a smartwatch to identify atrial fibrillation. New England Journal of Medicine, 381(20), 1909-1917.
2. Arora, S., Venkataraman, V., Donohue, K. D., Biglan, K. M., Dorsey, E. R., & Little, M. A. (2021). High-frequency remote monitoring of Parkinson’s disease: using wearables to predict fluctuations. npj Digital Medicine, 4(1), 1-10.
3. Wang, R., Blackburn, G., Desai, M., Phelan, D., Gillinov, L., Houghtaling, P., & Gillinov, M. (2022). Accuracy of wrist-worn heart rate monitors. JAMA Cardiology, 7(2), 150-158.
4. Bot, B. M., Suver, C., Neto, E. C., Kellen, M., Klein, A., Bare, C., ... & Friend, S. H. (2016). The mPower study, Parkinson disease mobile data collection using ResearchKit. Scientific Data, 3, 160011.
5. Sudre, C. H., Murray, B., Varsavsky, T., Graham, M. S., Penfold, R. S., Bowyer, R. C., ... & Steves, C. J. (2021). Attributes and predictors of long COVID. Nature Medicine, 27(4), 626–631.










