Adoption of Blockchain in the Pharmaceutical Supply Chain
Enhancing transparency and security
Hamed Taherdoost, Founder, Hamta Business Corporation; Associate Professor and Chair, RSAC, University Canada West, & Director, R&D, Q Minded | Quark Minded Technology Inc.
The pharmaceutical supply chain faces challenges in transparency, security, and counterfeit drug prevention. Blockchain technology offers a decentralised, tamper-proof solution for tracking and verifying drug authenticity. This article explores the adoption of blockchain in pharma supply chains, highlighting its benefits, challenges, and potential for improving regulatory compliance and patient safety.

The pharmaceutical supply chain is inherently complex, involving multiple stakeholders, from raw material suppliers and manufacturers to distributors, pharmacies, and patients, creating vulnerability to inefficiencies, counterfeiting, and data silos. Blockchain technology, with its decentralised ledger, immutable records, and smart-contract capabilities, offers a transformative approach to address these challenges by enhancing transparency, traceability, and security across the end-to-end drug lifecycle (Akram et al., 2024). Recent studies have highlighted blockchain’s potential to streamline regulatory compliance, reduce operational costs, and build trust among partners while mitigating counterfeit risks (Riedel, 2024). Early pilots, such as IBM and Walmart’s endto-end pharmaceutical tracking initiative demonstrate tangible benefits in product authenticity and handling compliance. However, widespread adoption remains constrained by integration hurdles with legacy systems, high initial investments, and evolving regulatory landscapes. This article explores the adoption of blockchain in pharmaceutical supply chains, examining its benefits, real-world use cases, implementation strategies, key challenges, and future outlook, providing a roadmap for stakeholders aiming to leverage decentralized technologies for enhanced supply chain resilience and patient safety.
The need for transparency and security
Pharmaceutical supply chains are plagued by fragmented information flows, creating blind spots that facilitate counterfeit drug distribution. Studies reveal that up to 50 per cent of medications in certain developing regions may be substandard or falsified, directly endangering patient health (Riedel, 2024). Traditional centralised databases often lack real-time visibility and are vulnerable to tampering, complicating recall processes and regulatory reporting. The U.S. Drug Supply Chain Security Act (DSCSA) mandates product tracing but relies heavily on paper-based or semi-digital processes, delaying traceability by weeks (Akram et al., 2024). In this context, supply chain stakeholders require a unified, tamperevident data platform that provides end-to-end visibility, instant verification of product provenance, and automated audit trails to safeguard public health and comply with stringent regulations.
Fundamentals of blockchain technology
Blockchain is a distributed ledger main tained by a network of nodes, where each block contains a set of transactions cryptographically linked to the previous block, ensuring immutability and auditability. Permissioned blockchains, commonly deployed in pharmaceuticals, restrict network participation to vetted entities, balancing transparency with data confidentiality. Smart contracts, self-executing code on the blockchain, automate compliance and enforce agreed-upon business rules, enabling immediate execution of actions such as payments or shipment verifications, when predefined conditions are met (Akram et al., 2024). This architecture eliminates single points of failure, reduces reliance on intermediaries, and ensures a single source of truth that all participants can trust.
Key benefits for pharmaceutical supply chains
The pharmaceutical supply chain faces numerous challenges, including counterfeit drugs, regulatory compliance, and inefficient tracking systems. Blockchain technology offers transformative solutions by providing enhanced transparency, security, and efficiency. By creating an immutable and decentralized ledger, blockchain ensures accurate tracking of pharmaceutical products from production to delivery, reducing the risk of counterfeit drugs entering the market. Additionally, it improves data security through cryptographic techniques, ensuring that sensitive information is protected from unauthorized access or tampering. The integration of smart contracts also automates compliance processes, making it easier to adhere to stringent regulations. These benefits contribute to a more reliable and cost-effective supply chain, ultimately improving patient safety and trust in the pharmaceutical industry (Figure 1).
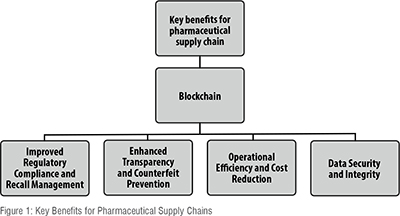
Enhanced traceability and counterfeit prevention
Blockchain provides immutable, timestamped records for every transaction, enabling stakeholders to trace a drug’s journey from manufacturing through distribution to the patient (Akram et al., 2024). Real-time visibility of temperature logs, custody transfers, and compliance checks prevents diversion and ensures product quality, particularly for temperature-sensitive biologics.
Improved regulatory compliance and recall management
Automated audit trails and time-stamped records facilitate rapid recall response by pinpointing affected batches and locations within minutes, rather than days or weeks (Riedel, 2024). Smart contracts can enforce DSCSA or Falsified Medicines Directive (FMD) requirements, triggering alerts when discrepancies arise.
Operational efficiency and cost reduction
By automating manual reconciliation processes and reducing paperwork, blockchain lowers administrative overhead and streamlines supplier onboarding, inventory management, and order fulfillment.
Data security and integrity Cryptographic hashing ensures data tampering is immediately detectable, while decentralized consensus mechanisms mitigate cyber-attack risks associated with centralised servers. Permissioned networks maintain confidentiality by encrypting sensitive commercial or patient data, accessible only to authorized parties (Akram et al., 2024).
Implementation strategies for stakeholders
Building a successful blockchain-based pharmaceutical supply chain requires careful planning and collaboration among various stakeholders (Figure 2). Consortium formation plays a crucial role in this process, as it involves bringing together regulators, manufacturers, distributors, and service providers. This multi-stakeholder approach ensures shared governance, clear standard definitions, and collective investment in the necessary infrastructure. To mitigate risks and ensure effective stakeholder alignment, phased pilots are recommended. By starting with targeted pilots, such as those focused on high-value biologics or specific distribution corridors, organisations can gain insights and learn iteratively before scaling up the implementation (Akram et al., 2024).
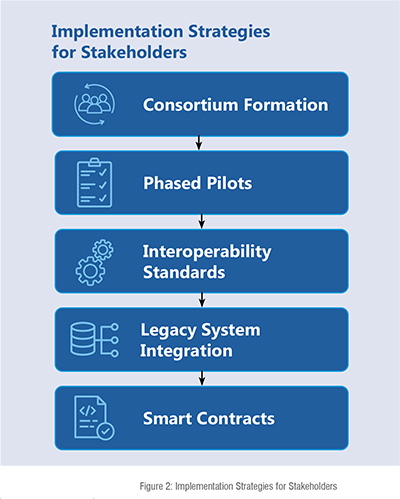
In addition to pilot projects, interoperability standards are critical for ensuring seamless integration of blockchain with existing systems. Leveraging blockchain platforms that support GS1 identifiers and EPCIS standards allows for compatibility with current traceability systems and regulatory frameworks, streamlining the transition (Riedel, 2024). Another important aspect is the integration of blockchain with legacy systems. The implementation of API-based connectors and middleware layers facilitates this process by enabling organizations to retain their existing ERP and warehouse management systems while linking them to the blockchain network, ensuring a smooth and gradual integration. Lastly, the development of smart contracts tailored to the pharmaceutical industry's regulatory workflows is essential. Co-developing smart contracts that automate processes such as DSCSA verifications, temperature compliance checks, and recall triggers helps to reduce manual intervention and enhances overall efficiency (Akram et al., 2024).
Challenges and barriers to adoption
The implementation of blockchain technology in the pharmaceutical supply chain often requires a high initial investment. Costs related to deploying the infrastructure such as node hosting, platform development, and employee training can be substantial, particularly for small and medium-sized enterprises (Riedel, 2024). However, collaborative funding models and government-backed incentives can help mitigate these financial barriers and encourage broader participation.
Another significant challenge lies in integrating blockchain with legacy IT systems. Many pharmaceutical companies rely on deeply embedded ERP and warehouse management systems, which demand extensive customization to interface effectively with new blockchain networks. This integration often requires robust middleware solutions and a phased approach to migration to ensure operational continuity.
Scalability and performance also present technical hurdles. Permissioned blockchains, though suitable for enterprise use, may struggle with throughput and latency issues when handling the high transaction volumes typical of global pharmaceutical supply chains. To address this, innovative consensus mechanisms such as Proof of Authority or hybrid models are being explored to maintain system performance at scale. In addition to technical concerns, regulatory uncertainty poses a substantial risk. The legal landscape surrounding data privacy, cross-border data transfers, and the use of digital identities continues to evolve. This uncertainty introduces compliance risks that can hinder adoption unless there is clear and consistent guidance from regulatory authorities and standards organizations (Akram et al., 2024).
Finally, organizational and cultural resistance can impede blockchain adoption. Effective implementation demands a high degree of trust, collaboration, and reengineering of existing processes among stakeholders who may have conflicting interests. Overcoming this resistance requires comprehensive change management strategies, including strong executive sponsorship, user training programs, and active engagement with all stakeholders to align goals and expectations.
Future outlook
Blockchain technology stands at the cusp of revolutionizing pharmaceutical supply chains by delivering unparalleled transparency, security, and efficiency. As pilot programs mature into production systems, stakeholders can expect enhanced patient safety, streamlined regulatory compliance, and reduced counterfeit risk. Continued innovation in consensus protocols, interoperability standards, and privacy-preserving techniques will address current limitations around scalability and data protection. Strategic consortiums, supportive regulatory frameworks, and collaborative funding models will play pivotal roles in accelerating adoption (Kumar et al., 2024). By embracing decentralized architectures and smart contracts, the pharmaceutical industry can foster resilient, trust-based supply chains that safeguard public health and drive sustainable growth.










