Breaking Bottlenecks in Continuous Biomanufacturing
Clog-Free, Ultra-High-Density Perfusion Filter with Continuous Host Cell Protein (HCP) Removal
Abstract:
Continuous biomanufacturing offers the potential for higher yields and consistent quality, yet conventional filters clog rapidly at high cell densities. A new microfluidic perfusion system enables clog-free operation under ultra-high-density conditions, continuously removes dead cells, and reduces host cell proteins by 50 per cent. This approach extends production runs to 50 days and improves biologics consistency.
1. What are the primary reasons conventional perfusion filters fail under ultra-high-density conditions, and how does your system fundamentally alter this dynamic?
Perfusion technologies such as Tangential Flow Filtration (TFF) and Alternating Tangential Flow (ATF) are typically membrane-based, with pore sizes around 2 µm. At high cell densities (>5 × 10⁷ cells/mL), these membranes are prone to fouling, often beginning early in culture, progressively worsening, and consistently leading to product retention. To mitigate this, operators usually employ redundant filters, but even so, continuous perfusion runs are rarely sustained beyond ~25 days.
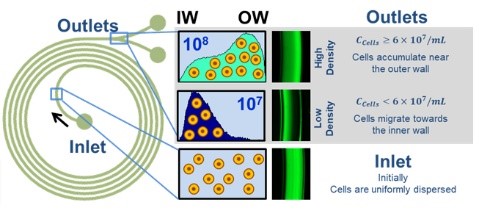
Our team has developed a non-membrane alternative based on high-density inertial microfluidics, a phenomenon we first reported. The channels are ~100× larger than mammalian cells, effectively eliminating clogging even at densities up to 1 × 10⁸ cells/mL. This enables perfusion processes to be extended to ~50 days, roughly twice the duration achieved with current systems.
Importantly, the device also facilitates selective clearance of dead cells, removing ~50 per cent per 2 VVD. This reduces host cell protein (HCP) accumulation in the bioreactor by about half compared to conventional perfusion cultures. This reduction in HCP load translates into cleaner harvest streams and improved downstream purification yields, supporting more efficient continuous biomanufacturing.
2. Could you walk us through the engineering principles behind the microfluidic perfusion system that allow for sustained clog-free operation?
Inertial microfluidics is attractive because it operates in a unique regime between laminar-flow microfluidics and large-scale turbulent systems. This intermediate flow regime enables throughputs 10²–10⁴ times higher than conventional microfluidic approaches, making the technology viable for industrial-scale applications. Over the past decade, inertial microfluidics has been widely applied to cell separation, but primarily in dilute suspensions (<1 × 10⁷ cells/mL). At higher densities, conventional inertial focusing at the inner wall becomes unstable, with particle streams dispersing until separation is lost.
Our group identified a distinct regime in rectangular spiral channels where inertial focusing occurs at the outer wall instead stabilized by particle–particle interactions at elevated concentrations. This phenomenon enables robust separation even at high cell densities while maintaining industrially relevant throughput. Moreover, the system also differentiates live from dead cells: dead cells are selectively removed at ~50 per cent clearance per 2 VVD. This reduces host cell protein (HCP) accumulation within the bioreactor, offering both longer culture stability and a cleaner harvest stream for downstream processing.
This discovery effectively extends inertial microfluidics from research-scale cell separation into a true biomanufacturing platform, bridging the gap between laboratory innovation and industrial continuous processing.
3. Continuous systems often face hurdles when moving from bench scale to large-scale production. How has your design addressed scalability without compromising performance?
One of the biggest hurdles in continuous bioprocessing is translating elegant bench-scale systems into industrial operations without losing efficiency or robustness. Our design tackles this by relying on a principle that scales naturally in biomanufacturing — throughput by parallelisation. Each module delivers 100–10,000 times the throughput of conventional microfluidic devices, and instead of stretching channel dimensions and risking shifts in flow dynamics, we simply replicate identical modules that operate under the same fluidic conditions as the original design. This “numbering-up” strategy preserves separation fidelity and the underlying fluid physics, ensuring that the performance demonstrated at the bench is reliably reproduced at production volumes. With throughput this high, we are now planning to scale the system to support bioreactors up to 2000 L, paving the way for GMP-ready, industrial-scale adoption of continuous cell separation.
4. Continuous removal of dead cells is often difficult in perfusion systems. What specific innovations in your filter technology enable this without interrupting live-cell productivity?
Continuous removal of dead cells is often cited as one of the major bottlenecks in perfusion culture, since most filter systems indiscriminately retain both live and dead populations. In contrast, the selective clearance of dead cells is a natural outcome of our technology. The microfluidic chip is designed to exploit dense suspension inertial focusing, which naturally concentrates ONLY viable cells within the flow path while leaving non-viable cells in the supernatant. As media is exchanged, the dead-cell–rich supernatant is continuously removed from the bioreactor, while the live cells remain unaffected and highly productive.
This built-in selectivity means there is no mechanical stress or direct intervention imposed on the viable cell population. Instead, the system provides a passive yet robust mechanism for continuously reducing dead-cell burden, thereby minimising host cell protein accumulation and maintaining culture health — all without interrupting live-cell productivity.
5. By lowering HCP burden at the upstream stage, what downstream purification efficiencies or cost savings have you quantified?
By selectively removing dead cells upstream, our technology is expected to reduce host cell protein (HCP) burden by ~50 per cent compared to ATF systems. This is significant because HCP is a major contaminant that drives complexity and cost in downstream purification. A cleaner upstream harvest means fewer impurities entering capture and polishing steps, which should improve column performance and extend resin lifetimes. In effect, we are shifting part of the purification burden upstream, allowing downstream chromatography to become more efficient and predictable. In monoclonal antibody production, this could translate to extended Protein A resin lifetimes and reduced buffer use in polishing steps
The benefits extend beyond quality to cost. Based on our projections, the combination of ~14 per cent higher product yield, ~45 per cent lower COGs, and improved operations with no downtime for filter changes or washing could significantly reduce buffer and resin consumption, lower labor costs, and accelerate batch turnaround. For companies scaling up to 2,000 L bioreactors, these efficiencies are expected to translate into substantial savings while supporting higher productivity and more robust supply of biologics, including antibodies and viral vectors.
6. Is the system optimised for specific mammalian cell lines (e.g., CHO, HEK293), or does it maintain broad applicability across diverse host platforms?
Our system is not limited to specific mammalian cell lines such as CHO or HEK293 but is designed for broad applicability across diverse host platforms — from mammalian cells to microbial systems and even algae. This versatility is possible because, in addition to discovering the core technology, we have developed a numerical model and scaling law that describe the fundamental physics governing cell focusing and separation. These predictive tools guide our channel designs for different host systems, enabling us to adapt the technology across a wide range of bioproduction platforms. In effect, the combination of experimental validation and model-based design makes the system platform-agnostic and future-ready for multiple biologic modalities
7. What are the main barriers - technical, cultural, or regulatory - that you anticipate for widespread adoption of continuous clog-free perfusion, and how are these being addressed?
The primary barrier to adoption is cultural rather than technical. Most bioprocess teams are accustomed to conventional ATF/TFF-based perfusion, and their standard protocols have been built around these membrane-based systems. Since our clog-free perfusion technology does not use a traditional membrane filter, it challenges existing definitions of a “filter” and raises concerns about the possibility of stray cells entering the harvest stream. To address this, we include a safeguard: a small inline filter before the harvest tank to ensure safety and provide reassurance during adoption.
On the technical side, our system inherently removes dead cells from the culture, producing a cleaner perfusion environment that leads to higher downstream purification yields. This requires some protocol adjustments, but in ways that are ultimately beneficial to process efficiency. To further protect harvest integrity, we have also developed a dead cell harvester positioned upstream of the harvest tank. Regulatory compliance is not expected to be a barrier, as our system uses USP Class VI–certified materials, is fabricated to ISO standards, and is fully compatible with GMP operations.














