Advancing Nanovaccines through Transdermal Delivery
S. Neelufar Shama, Professor & Head, Department of Pharmacognosy, Scient Institute of Pharmacy
Nanovaccines represent a promising frontier in immunisation, with transdermal delivery offering a needle-free, patient-friendly approach. The integration of nanotechnology and transdermal delivery systems represents a significant advancement in the field of vaccination. This article explores the latest innovations in nanovaccine development and transdermal systems, examining their potential to revolutionise vaccination. Future prospects and challenges in advancing this technology toward widespread clinical adoption are also discussed.
Vaccination has been one of the most successful public health interventions, reducing the burden of infectious diseases worldwide significantly. However, traditional vaccine delivery methods, predominantly through intramuscular or subcutaneous injections, pose challenges such as needle phobia, need for trained personnel, and the risk of needle-stick injuries. Nanotechnology, with its ability to engineer materials at the molecular scale, offers innovative solutions to these challenges, particularly through the development of nanovaccines. When combined with transdermal delivery systems, nanovaccines present a novel approach that could transform how vaccines are administered, enhancing both efficacy and accessibility.
The Promise of Nanovaccines
Nanovaccines are vaccines that utilise nanoparticles to deliver antigens to the immune system. These nanoparticles can be engineered to encapsulate antigens, ensuring their stability and controlled release. Nanoparticles can also enhance the immune response by targeting specific cells or tissues, thereby improving the vaccine’s efficacy. The small size of nanoparticles allows them to interact with the immune system more effectively, mimicking pathogens and promoting a robust immune response.
Several types of nanoparticles are being explored for use in nanovaccines, including liposomes, polymeric nanoparticles, and inorganic nanoparticles such as gold or silica. Each type offers distinct advantages in terms of stability, biocompatibility, and the capability to carry multiple antigens or adjuvants. The adaptability of nanoparticles allows for the design of vaccines that are tailored to specific diseases, potentially enabling more personalised approaches to immunization.
Transdermal Delivery: A needle-free alternative
Transdermal delivery involves administering vaccines through the skin, bypassing the need for needles. The skin is an attractive target for vaccination due to its abundance of antigen-presenting cells, which play a vital role in initiating immune responses. Traditional transdermal delivery methods, such as patches or creams, have been limited by the skin’s barrier function, which prevents most molecules from penetrating deeply enough to reach the immune cells.
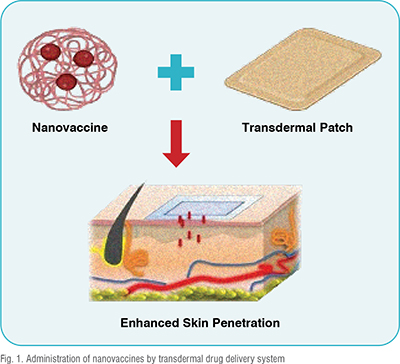
However, recent advances in transdermal technology have overcome these limitations. Microneedles, for example, are tiny needles that can pierce the outer layer of skin to deliver vaccines directly to the underlying immune cells, devoid of pain. These microneedles can be coated with nanoparticles containing antigens, ensuring that the vaccine is delivered precisely where it is needed. Dissolvable microneedles, which dissolve after administration, offer an additional advantage by eliminating the need for sharps disposal. (Figure: 1)
Innovations in nanovaccine formulations for transdermal delivery
The combination of nanovaccines and transdermal delivery systems has led to several innovative formulations. One approach involves the use of lipid-based nanoparticles, such as liposomes or solid lipid nanoparticles, which are known for their biocompatibility and ability to fuse with cell membranes. These nanoparticles can be incorporated into microneedle patches, allowing for efficient transdermal delivery of the vaccine. Recent studies have demonstrated the potential of these systems in eliciting strong immune responses against a group of pathogens, including influenza and COVID-19.
Another promising formulation is the use of polymeric nanoparticles, which can be designed to release antigens in a controlled manner over time. This sustained release can enhance the immune response by providing continuous stimulation to the immune system. Polymeric nanoparticles can also be engineered to respond to specific triggers, like changes in pH or temperature, ensuring that the vaccine is released only when it reaches the target site within the skin.
Inorganic nanoparticles, such as those made from gold or silica, are also being explored for transdermal vaccine delivery. These nanoparticles offer unique properties, such as the ability to enhance the visibility of the vaccine in imaging studies or to provide adjuvant effects that boost the immune response. For example, gold nanoparticles have been shown to enhance the delivery and presentation of antigens to immune cells, leading to stronger and longer-lasting immunity.
Current applications and clinical progress
The development of nanovaccines for transdermal delivery is still in its early stages, but several promising candidates are advancing toward clinical trials. One of the most notable examples is the development of a transdermal influenza vaccine using lipid-based nanoparticles in a microneedle patch. Preclinical studies have shown that this vaccine can induce strong immune responses with minimal side effects, and early-phase clinical trials are currently underway.
BNT162b2, an mRNA vaccine, encodes a full-length, membraneanchored S protein. Both mRNAs have been embedded in lipid Nanoparticles to ensure optimal distribution to target cells. The Moderna and BNT vaccines were the first to be licensed for SARSCoV-2. Novavax of Maryland employed baculovirus/Sf9 technology to generate full-length SARS-CoV-2 S glycoproteins. For injection, an adjuvant, saponin-based Matrix-M1 is used, to solve the problem of earlier protein subunit vaccines failing to elicit a cell-mediated immunity response. However, the main issue with these vaccinations is that they cannot be stored at ultralow temperatures ranging from -80 to -60°C for Pfizer-BioNTech vaccines and -50 to -15°C for Moderna vaccines. Moreover, the Pfizer-BioNTech vaccine may be frozen only at -25 to -15°C for a period of 2 weeks, requiring specialised logistics and storage equipment. On the other hand, Moderna vaccine can be refrigerated at 2-8°C for 30 days.
Another area of active research is the development of nanovaccines for cancer immunotherapy. Transdermal delivery of cancer vaccines could provide a more targeted approach, delivering antigens directly to the skin’s immune cells and potentially reducing the risk of systemic side effects. Several preclinical studies have demonstrated the potential of nanovaccine patches to induce strong anti-tumor responses in animal models, and efforts are ongoing to translate these findings into human trials.
Challenges and future directions
Despite the promise of nanovaccines and transdermal delivery, many challenges must be addressed to completely realise their potential. One among the primary challenges is ensuring the stability of the nanoparticles during storage and transport. Nanoparticles are often sensitive to environmental conditions like temperature and humidity, which can have impact on their efficacy. Developing robust formulations that maintain their stability under various conditions is critical for the widespread adoption of nanovaccines.
Another challenge is the need for large-scale manufacturing processes that can produce nanovaccine formulations consistently and cost-effectively. The production of nanoparticles often involves complex processes that can be difficult to scale up, and ensuring the quality and uniformity of the final product is essential for regulatory approval. Advances in manufacturing technologies, such as microfluidics and 3D printing, may offer solutions to these challenges, enabling the production of high-quality nanovaccines on a commercial scale.
Regulatory considerations also pose challenges for the development and approval of nanovaccines delivered through transdermal systems. The unique properties of nanoparticles, including their small size and ability to interact with biological systems at the molecular level, raise questions about their safety and potential long-term effects. Regulatory agencies will need to establish clear guidelines for the evaluation of nanovaccine safety and efficacy, ensuring that these innovative products meet the same rigorous standards as traditional vaccines.
Looking ahead, the future of nanovaccines and transdermal delivery systems is bright. Continued research and development in this field are likely to yield new formulations that offer even greater efficacy, safety, and convenience. The ability to design vaccines that are tailored to specific diseases, populations, or even individuals opens up exciting possibilities for personalised medicine. Moreover, the use of transdermal delivery systems could expand access to vaccines, particularly in low-resource settings where traditional injection-based methods may be less feasible.
Conclusion
The integration of nanotechnology and transdermal delivery systems represents a significant advancement in the field of vaccination. Nanovaccines offer the potential to enhance immune responses, improve vaccine stability, and enable targeted delivery to specific tissues or cells. When combined with transdermal systems, these vaccines can be administered in a needle-free, patient-friendly manner, addressing many of the challenges associated with traditional vaccination methods.
While there are still hurdles to overcome, including issues related to stability, manufacturing, and regulatory approval, the progress made thus far is encouraging. The ongoing development of nanovaccines for transdermal delivery has the potential to revolutionise how vaccines are administered, making them more accessible, effective, and personalised. As research continues to advance, we can expect to see these innovations play an increasingly important role in the future of public health.
The future of vaccination may well lie at the intersection of nanotechnology and transdermal delivery, offering new hope for the prevention and treatment of a wide range of diseases. With continued investment and collaboration between researchers, clinicians, and industry, the promise of nanovaccines delivered through the skin could soon become a reality, ushering in a new era of immunization.
References:
1. Priyanka, Mai Abdel Haleem Abusalah, Hitesh Chopra et al. Nanovaccines: A game changing approach in the fight against infectious diseases. Biomedicine & Pharmacotherapy. 2023; 167:115597.
2. Xinglong Gao, Xinlian Wang, Shilin Li, et al. Nanovaccines for Advancing Long-Lasting Immunity against Infectious Diseases. ACS Nano. 2023; 17 (24):24514-24538












