Gene to GMP Challenges and Opportunities in Gene Therapy Manufacturing
Sarang Brahma, Principal Scientist, Asklepios Biopharmaceutical (AskBio) Inc
Gene therapy is a form of life-saving therapeutic technique for patients suffering from genetic disorders. Adeno associated viral (AAV) delivery of healthy genes to cure the underlying genetic disease is currently the most prominent mechanism of gene therapy products that are commercially available. The bio-manufacturing process of these products is highly specialized and complex from a technological standpoint. Challenges arising from this complexity result in the prohibitive cost of these lifesaving therapies for patients. Understanding the nature of challenges and converting them into opportunities of innovation, collaboration and cues for regulatory changes could usher in a golden age for gene therapy.

Gene Therapy is a medicinal technique which delivers gene(s) to treat or prevent a genetic disease. Healthy genes often deemed as cargo are typically single or double stranded DNA which are delivered using viral vectors like adeno associated virus (AAV), lentivirus or retrovirus. The use of non-viral vectors like plasmids, lipid nanoparticles (LNP) or polymers like polyethylamine (PEI) is possible but relatively limited. Once delivered to the cells, the DNA either as nascent or integrated into the host cell genome can transcribe and translate into active protein to restore the normal function of the defective gene. Most gene therapy treatments tend to be a one-time dosing strategy which is expected to treat the patients for their lifetime. Gene therapies can be combined with gene editing approaches using siRNA, CRISPR, Zinc Finger Nucleases (ZFN) or TALENs for silencing or repair of the mutated genes. The discussion for this article will focus on AAV-mediated gene therapies which are the leading vector type and dominate the gene therapy market share due to their effectiveness and safety.
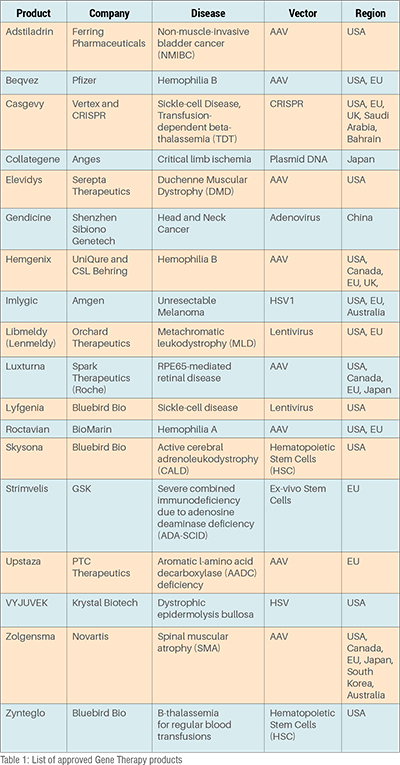
The global gene therapy market was valued at USD 10.47 billion in 2024 and is poised to exceed US$ 54 billion by 2033 with a CAGR of 19 per cent with approximately 10-12 products expected to be approved each year from 2025. Worldwide, the USA leads with 18 exclusively gene therapy products approved followed by 10 in the EU and under 5 for Asia and Australia (Table 1). A substantial number of indications for the products are for rare (1 in 2000 patients according to WHO) or ultra-rare (1 in 50,000 patients globally according to WHO) diseases which have a strong genetic etiology.
The adoption of gene therapy in Asia has been slower compared to the USA and EU largely due to factors such as high cost of these treatments which can be in excess of USD 1 million per patient and with limited public or private health insurance support most patients are priced out to benefit from them. Although the research, manufacturing, and clinical infrastructure as well as investments are gaining momentum, the Asian markets are yet to be primed for having research pipelines and commercial scale manufacturing of these products. The regulatory landscape of clinical trials and approvals advanced gene therapy products is evolving in several countries with only some countries like China and Japan having consistent prior experience and however the growth perspective for cell and gene therapy products in Asia remains optimistic with several countries like Japan, India, Singapore, China and South Korea cultivating investments (fiscal and work force), expanding access to clinical trials and updating the regulatory framework which could see a potential growth of 20-30 per cent with a market share over US$ 10 billion over the next 10 years.
Gene therapy manufacturing
The typical manufacturing process for the gene therapy AAV viral vectors involves the following steps–
Upstream cell culture and harvest
• The upstream AAV manufacturing consists of culturing of cell lines (e.g HEK or Sf9) which have high transfection efficiency, robust growth characteristics and can support manufacturing of high titers of AAV.
• Triple plasmid transfection with the therapeutic, RepCap and Helper plasmids using a transfection reagent like PEI. The therapeutic plasmid delivers the gene of interest, RepCap delivers the Rep and Cap genes required for AAV replication and capsid formation. The AAV serotype can be selected by altering the Cap sequence in RepCap. The Helper plasmid delivers helper genes like E4, E2a and VA essential for vector production and replication in the cell line.
• Following transfection and culture for 48-72 hours, the cells are harvested, depth filtered and lysed to release the AAV. The released AAV vectors are subjected to the downstream purification steps.
Downstream purification and filtration
• The depth filtered harvested AAVs from the lysed cells are subjected to tangential flow filtration (TFF) for concentration and desalting proteins and contaminating nucleic acids.
• This is followed by affinity or capture chromatography with resins having selective affinity towards a broad range or specific AAV serotypes. These columns help eliminate host cells proteins and nucleic acids.
• The AAV are then purified further to enrich the full particles (with the correct size of DNA) and remove the empty and partial particles. This is conventionally done using Ultracentrifugation (UC) which allows separation based on mass gradients. The UC steps can be replaced by an anion exchange (AEX) chromatography which is faster, efficient and cheaper, however it does need significant development and optimisation.
• The purified and enriched full AAV particles are further subjected to polishing (SEC) steps and concentration to the target concentration using ultrafiltration diafiltration (UFDF) steps. The product can be called ‘drug substance (DS)’ at this stage.
Drug product vialing and release
• The drug substance (DS) is purified using depth filters and can be subjected to additional polishing steps to remove any particulate contaminants and is usually deemed as Drug Product (DP) after this stage.
• The drug product is vialed into single-use or multi-use vials and is labeled appropriately.
• The drug product is subjected to rigorous analytical and quality control (QC) testing before being released for use.
Challenges in Gene Therapy Manufacturing
AAV-based gene therapy products manufacturing involves precise orchestration of a highly complex biological process. Due to the biological and chemical nature of this process, cell-related and technical challenges can be abundant. Let’s look at the three most significant challenges which affect production.
Starting material quality and availability
The cell line and the three plasmids are the critical starting materials and the biological nature of these can lead to lot-to-lot variability or low yields. The challenges with cell lines are typically related to their genetic drifts across passage numbers and risks of contamination with adventitious viruses, mycoplasma or losses in growth kinetics and transfection efficiency. Companies rely on thorough master and working cell bank characterisation and storage typically at outsourced vendors for a consistent supply and quality of the cell lines. The quality and integrity of the three plasmids is crucial for a successful manufacturing campaign. The ability to manufacture GMP runs at scale hinges on successful upstream process development to optimise plasmid ratios, selection of transfection reagents / enhancers and the optimised sequences of the plasmids themselves. Genomic integrity and conformation of the plasmids, particularly the therapeutic plasmid which could have a significant number of repeat and structurally complex sequences, can be difficult to manufacture in conventional E.coli-based plasmid manufacturing platforms. Lack of plasmid integrity if not analysed using advanced techniques like next generation sequencing (NGS) can lead to a significant regulatory escalation and potential clinical holds for the manufactured products due to a potentially failed identity release test.
Importantly, the cost of manufacturing GMP-grade plasmids is significant and could constitute as high as 25 per cent of the total manufacturing costs. The average cost of a full-GMP (compared to research grade or GMP-source) plasmid is approximately US$ 250,000 for each batch of approximately 2000 litres. Thus, for clinical GMP batches, companies typically could spend millions of dollars on plasmids alone and that does not guarantee that the run will be successful. Lastly, the lead time to manufacture the plasmids could be several months to a year with a limited number of providers offering GMP-grade plasmid solutions. The challenges to maintain costs and timelines require meticulous planning and significant resources with very little margin of failure in AAV-based gene therapy manufacturing.
Comparability and consistency of quality for drug products
For gene therapy products which make their journey through the phases of pre-clinical toxicology to clinical and eventually commercial, the manufacturing processes, sites, scales and analytics evolve over time for efficiency, costs and improvements in the quality of the product. Comparability across the variables needs to be thoroughly established and is a paramount requirement by the regulatory agencies reviewing the IND or BLA submissions. Comparability studies pose several challenges wherein firstly these studies require a significant amount of material. This is especially challenging for therapies which require higher doses and could potentially decrease the amount of clinical material available for dosing. Furthermore, in certain instances, the first manufactured material could be several years older than the most recent lot manufactured with significant emphasis being laid on the appropriate storage and availability of the older lots.
Next, due to the complexity of gene therapy products, a comparability framework must be tailored for each set of lots to be compared with a risk assessment and identification of parameters for testing. This risk-based strategy often determines that release CQAs alone are not sufficient to demonstrate comparability, but additional characterisation analyses are required. This is a significant challenge with respect to the analytical capabilities available leading to financial (especially if outsourced) and timeline strains. Finally, due to the complex biological nature of the AAV-based products, inherent heterogeneity and lot to lot variability is expected to a degree. Designing comprehensive comparability studies which can correlate the CQA and biophysical attribute variabilities to clinical outcomes and satisfy the expectations of the regulatory agencies are indeed a moonshot which only a few companies can achieve ( Figure 2 ).

Scalability and high production costs
The complex AAV manufacturing process which is reliant on cell culture, optimal transfection and downstream purification poses a scalability challenge for commercial scales. Several gene therapy products require a high dose of > 10 viral genomes (VG)/Kg due to low or moderate efficacy with infusion doses being several milliliters of the manufactured products in 10 to 10 VG/mL. Thus, a commercially manufactured 2000-liter batch with moderate yields could be sufficient to dose only 10 patients for a 10 VG/Kg adult dose. A significant amount of material can be required for batch release analytical testing and stability testing which further reduces the amount of materials available for clinical use. The challenge for gene therapy companies globally is to propel the success observed in phase I and II clinical stage manufacturing to late stage and commercial scales. Several companies lack in-house manufacturing capabilities for phase 3 and onwards stages and finding gene therapy CDMOs to manufacture can be time consuming considering technical transfers, availability of manufacturing slots and fiscally straining with per batch costs exceeding US$ 5 million.
The challenges with AAV gene therapy manufacturing are a significant factor holding back in unleashing a golden era for these therapies and making them successful like protein biologics in the early 2000s. This was acknowledged during the 2025 American Society of Gene Therapy (ASGCT) meeting along with the fact that AAV gene therapies are at an inflection point in time. Companies which are adaptable and responsive to the current manufacturing, clinical and regulatory challenges with commitment towards making gene therapy products safe and affordable can find clinical and commercial success and longevity.
Opportunities in gene therapy manufacturing
Gene therapy products are transformational and at times the only hope for patients with terminal genetic disorders. Most commercial gene therapies have been collaborative efforts between universities, start-ups and a large pharma or biotech company. These efforts allow sharing of R&D, manufacturing process and regulatory knowledge. It also helps companies connect large-scale manufacturing and storage facilities networks, something which would be challenging for smaller companies. Success stories for such collaborations which were fruitful are University of Pennsylvania, Spark and Roche Therapeutics for Luxturnatm, University of North Carolina (Jude Samulski’s lab), Avexis and Novartis for Zolgensmatm to name a few.
Updates to technology platforms such as the introduction of stable producer cell lines, reducing the dependency on triple plasmid transfection and in turn costs are being explored by several companies. Utilisation of AI-driven tools for predicting manufacturability of AAV constructs as well as controlling manufacturing process metrics is a powerful approach to prevent manufacturing failures. For comparability-related challenges, opportunities are often offered by the regulatory agencies like the USFDA to have consultation meetings with them and get feedback on the analytical comparability prior to IND or BLA submissions. Companies are strongly encouraged to capitalise on the meeting feedback to understand gaps and mitigate potential pitfalls when they present the data to the regulators.
Unique opportunities are available for the Asia Pacific region particularly in the contract manufacturing and development (CDMO) space. Gene therapy manufacturing requires a consistent supply of high-quality plasmid starting with companies experiencing lead times of several months up to a year for GMP-grade supplies. This is a perfect opportunity for several Asia Pacific companies to employ novel DNA manufacturing strategies for high-quality GMP plasmid manufacturing. Opportunities also exist in the analytics space particularly for the tests like replication competent AAV (rcAAV), adventitious viruses, sterility and process viral clearance which are almost always outsourced by companies manufacturing these products. Offering cost-effective and rapid turnaround on the testing of these products has been offered by several CDMO in Asia and companies located in the US and Europe have been exploring to outsource these analyses harnessing the fast transport and supply chain logistics available. Although intensive investment and effort is required to offer commercial scale contract manufacturing capabilities, several Asian market players are taking that step and are being considered as an attractive manufacturing partner for US or Europe-based companies.
In summary, gene therapy products have tremendous potential to cure the unmet needs of patients suffering from genetic disorders. The bottlenecks and challenges in gene therapy manufacturing are stark, yet they are something which can be overcome with innovation, collaboration, and regulatory changes. The outlook for the future of gene therapy remains to be bright with hopes of several patients resting on its success.
References
1 OpenAI. Gene therapy mechanism. AI-generated figure created using ChatGPT. 2025. Available from: https://chat.openai.com/
2 Gene Therapy Market Size, Share & Analysis Report, 2024-2033; Nova1Advisor; Report Code: 7819; Published: Apr 2025
3 Grieger, J C et al.; Production of Recombinant Adeno-associated Virus Vectors Using Suspension HEK293 Cells and Continuous Harvest of Vector from the Culture Media for GMP FIX and FLT1 Clinical Vector. Molecular Therapy 24-2, 287 – 297 (2016)
4 Li, C., Samulski, R.J.; Engineering adeno-associated virus vectors for gene therapy. Nat Rev Genet 21, 255–272 (2020)
5 USFDA Center for Biologics Evaluation and Research (CBER); Manufacturing Changes and Comparability for Human Cellular and Gene Therapy Products, Draft Guidance for Industry; FDA-2023-D-2436 (2023)
6 Jiang, Z et al.; Challenges in scaling up AAV-based gene therapy manufacturing. Trends in Biotechnology 41-10, 1268-1281 (2023)










