Industry 4.0-Revolutionising Hot Melt Extrusion
Smart manufacturing for enhanced efficiency and quality
Nimbagal Raghavendra Naveen, Department of Pharmaceutics, Sri Adichunchanagiri College of Pharmacy, Adichunchanagiri University.
Prakash Goudanavar, Department of Pharmaceutics, Sri Adichunchanagiri College of Pharmacy, Adichunchanagiri University.
Srikruthi Kunigal Sridhar, Department of Pharmaceutics, Sri Adichunchanagiri College of Pharmacy, Adichunchanagiri University.
Incorporating Industry 4.0 into Hot Melt Extrusion (HME) processes offers improved efficiency and quality in pharmaceutical manufacturing. Automation, data exchange, and AI enablereal-time optimisation, while digital twins foster innovation. This synergy promotes smarter manufacturing, ensuring competitiveness and high-quality products.

Hot Melt Extrusion (HME) has emerged as a groundbreaking technology in numerous industrial sectors, with a particular emphasis on its transformative impact within pharmaceutical research and manufacturing. Through its seamless integration with Industry 4.0 principles—namely, automation, data exchange, and Artificial Intelligence (AI)—HME has redefined the landscape of production processes. In recent years, HME has ascended as a cornerstone technology for drug delivery applications, owing to its automated and cost-effective scale-up properties. By significantly reducing labour costs and capital investment, HME has become an indispensable tool for pharmaceutical scientists across the globe. Moreover, the ongoing advancements in HME, bolstered by a comprehensive understanding of material science and process engineering, empower pharmaceutical scientists to craft highly efficient and durable products.
One of HME's most noteworthy achievements lies in its ability to address prevalent challenges faced by pharmaceutical scientists, including issues related to poor water solubility, bioavailability, and the physical and chemical stability of active pharmaceutical ingredients (APIs), thereby surpassing conventional techniques. At the heart of HME lies the extrusion process, a fundamental element that propels its efficacy. This process commences with the meticulous preparation of solid raw materials, encompassing polymers and pharmaceutical compounds, which undergo melting within an extruder barrel. Through a meticulously controlled environment, these materials are uniformly mixed to form a molten mass. Following this stage, the molten material undergoes a cycle of cooling and solidification, culminating in further refinement processes such as cutting and finishing to yield the final product. Regulatory bodies, recognising the transformative potential of HME, fervently advocate for the implementation of Quality by Design (QbD) and Process Analytical Technology (PAT) within this domain. These strategic tools, which include Raman and Near-infrared (NIR) spectroscopy, play a pivotal role in enabling real-time quality evaluation and process monitoring, thus harmoniously aligning with the principles of Industry 4.0.
Smart Manufacturing technology in HME Process
Smart manufacturing technologies are transforming HME, revolutionising efficiency, quality control, and productivity in pharmaceutical research and manufacturing. Machine Learning (ML), among these technologies, offers significant benefits in HME processes but also presents challenges, particularly concerning model transferability. Achieving good transferability, as observed with Partial Least Squares (PLS) and Linear Discriminant Analysis (LDA) models, ensures effective monitoring of filler particle size across various grades of Polylactic acid (PLA). Similarly, soft sensor models exhibit promising transferability in monitoring melt viscosity across diverse materials and equipment. Challenges arise, however, when monitoring physicochemical properties like API content, where direct relationships with real-time process measurements may be limited. Despite these challenges, integrating machine learning into HME holds promise for optimising process parameters, predicting product quality, and facilitating real-time decision-making in pharmaceutical manufacturing. Ongoing research endeavours aim to overcome these challenges, fostering broader adoption of machine learning in HME and industrial processes.
Digital twin is also one of the smart technologies related to HME process where HME utilises co-rotating twin screw extruders like blending Active Pharmaceutical Ingredients (API) and polymers. This process occurs in heated barrels, where mechanical and thermal energy facilitates polymer plasticisation around API particles. Temperature-controlled zones ensure uniformity, with screw configurations for feeding, kneading, dispersion, conveying, and metering. Gravimetric feeders ensure precise material delivery. HME is a well-established process increasingly adopted in pharmaceutical continuous manufacturing. It achieves efficient mixing, enhancing product uniformity and bioavailability, with advantages including solvent-free processing, fewer steps, lower costs, scalability, and consistency. Challenges include compound degradation and recrystallisation, hindering continuous processing adoption due to investment costs and changeovers. Digital twin technology offers a solution by creating virtual replicas of HME processes. This enables real-time monitoring, predictive modelling, and scenario analysis, optimising manufacturing and predicting API manufacturability. Ultimately, digital twins enhance efficiency, quality control, and decision-making in pharmaceutical manufacturing, aligning with industry innovation goals.
The combination of HME with 3D printing, particularly Fused Deposition Modelling (FDM), offers a promising method for producing controlled-release tablets. In this research, a co-rotating twin-screw extruder with 11-mm diameter screws was utilised to create fused filaments suitable for 3D printing. With precise temperature control facilitated by the extruder's design, which includes an L/D ratio of approximately 40 and eight electrically heated zones, the study aimed to assess the suitability of various pharmaceutical polymers for 3D printing. By integrating FDM-based 3D printing with HME, the researchers aimed to develop controlled-release tablets with customised drug delivery profiles [Figure 1]. Furthermore, comparing the drug release profiles of 3D printed tablets to conventional tablets provided valuable insights into the performance of 3D printed pharmaceutical formulations, highlighting the potential of this approach for advancing pharmaceutical manufacturing and drug delivery systems. (Figure 1).
Enhancing efficiency through automation and robotics in HME
Automation and robotics play a crucial role in advancing HME processes, particularly in pharmaceutical manufacturing. In HME, automation technologies streamline the production process by precisely controlling parameters such as temperature, pressure, and material feeding. This automation ensures consistent product quality and reduces the need for manual intervention, enhancing efficiency and productivity. Robotic systems complement automation by performing various tasks within the HME workflow. These tasks include material handling, loading and unloading of raw materials, and operating the extruder itself. Robots execute these tasks with speed and accuracy, minimising errors and optimising the production process. Moreover, automation enables real-time monitoring and control of the HME process. Advanced sensors and monitoring systems continuously collect data on key parameters, allowing for immediate adjustments to maintain optimal conditions and product quality. This real-time feedback loop ensures that the HME process operates at peak efficiency. Furthermore, automation facilitates seamless integration with other manufacturing processes, such as downstream packaging and quality control. Automated systems can transfer extruded products to packaging lines, conduct quality inspections, and ensure batch traceability, thereby streamlining the entire production workflow. Overall, automation and robotics in HME enable pharmaceutical manufacturers to achieve higher levels of productivity, reduce costs, and improve product quality. These technologies pave the way for advanced manufacturing techniques, driving innovation and competitiveness in the pharmaceutical industry.
Technical challenges
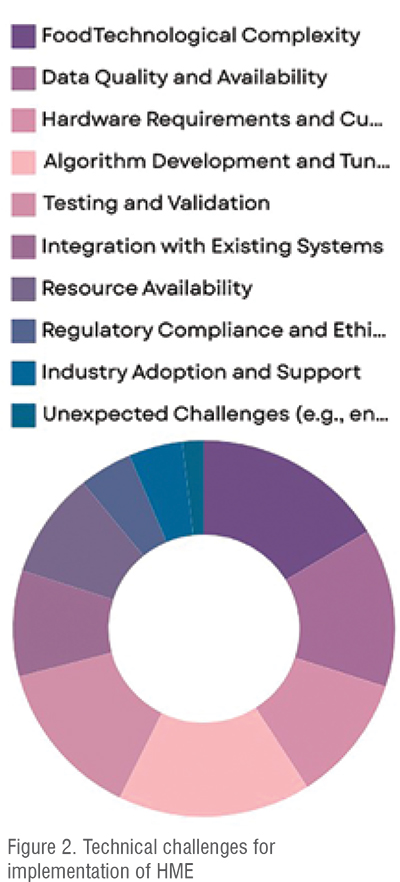
Several factors significantly influence HME implementation rates. Technological complexity, constituting 18 per cent, encompasses the intricacies of HME machinery and the sophistication of control systems required for precise operations. Algorithm development and tuning, also at 18 per cent, are crucial for optimising process parameters to ensure efficient material flow and consistent product quality [Figure 2]. Testing and validation, making up 15 per cent, are paramount for verifying product integrity and adherence to regulatory standards, essential for market approval and consumer safety. These factors collectively shape the landscape of HME adoption, highlighting the importance of technological advancements, process optimisation, and regulatory compliance in driving its widespread implementation. (Figure 2).
Future Perspectives and Conclusion
Looking ahead, the integration of Industry 4.0 principles into Hot Melt Extrusion (HME) processes signals a significant shift towards smarter manufacturing for improved efficiency and quality. Combining automation, data exchange, AI within HME holds immense potential for revolutionising pharmaceutical manufacturing and beyond. Industry 4.0 technologies will empower HME processes to become more autonomous and adaptable, continuously optimising parameters in real-time to ensure consistent product quality and operational efficiency. AI algorithms will analyse extensive data sets, offering valuable insights into process optimisation and predictive maintenance, ultimately reducing downtime and expenses. Moreover, the introduction of digital twins will enable the creation of virtual replicas of HME equipment and processes, facilitating simulation, optimisation, and scenario analysis. This digital representation of the production environment will expedite prototyping and foster the development of new formulations, accelerating innovation in drug delivery systems. As pharmaceutical companies transition towards continuous manufacturing, Industry 4.0-enabled HME processes will play a central role in meeting the industry's evolving demands. Continuous monitoring and control, facilitated by advanced sensors and analytics, will ensure compliance with regulatory requirements and enable real-time product release. In summary, the integration of Industry 4.0 principles into HME processes stands poised to transform hot melt extrusion into a smarter, more efficient, and agile manufacturing process. By embracing smart manufacturing technologies, pharmaceutical companies can strengthen their competitiveness, drive innovation, and deliver top-tier products to satisfy the needs of patients and regulatory bodies alike. The evolution of HME lies at the intersection of technology and pharmaceutical science, where smart manufacturing illuminates the path towards enhanced efficiency and quality in drug delivery.















