Process Analytical Technology
Application in precipitation processes
Marjatta Louhi-Kultanen, Research Lecturer, Docent in Industrial Crystallization, Department of Chemical Technology, Lappeenranta University of Technology, Finland
Precipitation is a commonly used purification method when a pure crystalline Active Pharmaceutical Ingredient, excipient or intermediate with specific particle properties needs to be isolated from a multi-component process solution after a synthesis, extraction or bio-process.
The main factors affecting precipitation processes and in-line process monitoring when Process Analytical Technology (PAT) based on in-line / on-line spectroscopic methods is applied, is discussed here.
Precipitation
The main purposes of crystal engineering of pharmaceutical compounds are usually to control crystal morphology, crystal size distribution and polymorphism.
The two main precipitation methods are:
Reactive crystallisation and
Precipitation by adding a precipitant chemical which decreases the solubility of the crystallising compound.
In the case of reactive crystallisation, the reaction can be as follows:

where
A, B reactants
C precipitate (poorly soluble solute /solvate / hydrate)
D forming solute having high solubility
There are several examples in reactive crystallisation in the pharmaceutical industry of base-acid compound systems where a change in pH by adding an acid / base precipitates the drug compound. One example of precipitation using a precipitant chemical is salting out of proteins by adding an inorganic salt into the aqueous protein solution.
Precipitation processes of fine chemicals are usually operated in semi-batch mode, i.e. one or more chemicals are added by pumping them into the mixing reactor continuously. In general, temperature control is required in order to control the properties of the forming crystals and / or if the processing is highly endothermic or exothermic.
The main factors affecting precipitation are as follows:
The solution:
• Selection of precipitants / reactants
• Selection of solvents
• Additives (habit modifiers, stabilisers)
• In-reactive crystallisation: reaction kinetics vs. crystallisation kinetics
• Reactant / precipitant concentrations
The operation conditions:
• Residence time (impact on supersaturation level)
• Mixing system (impeller type, mixing intensity, concentration / temperature distribution in the crystalliser, which is also important in order to avoid undesired side reactions)
• Reactant / precipitant feeding (mass flow rate, size of the feed tube, hydrodynamic conditions at the feeding point)
Supersaturation
The driving force of crystallisation is supersaturation, which is expressed by the difference or ratio of the actual and equilibrium (saturated solution) concentrations. While chemical potential is the true driving force, in practice, in industrial crystallisation, concentration based expressions are sufficiently accurate for most applications. The amount of solute required to make a saturated solution for a given condition is called the solubility. The solubility of organic compounds in different solvents is difficult to predict and, therefore, the solubility is usually determined experimentally. The principle used to determine supersaturation is shown in Figure 1. In the case of reactive crystallisation, the product yield can be obtained from the equilibrium concentration line (reactant A) shown in Figure 1. It should be remarked here that the solubility of the precipitate C can be dependent on the solution composition. The solubility is not necessarily constant as plotted in Figure 1.
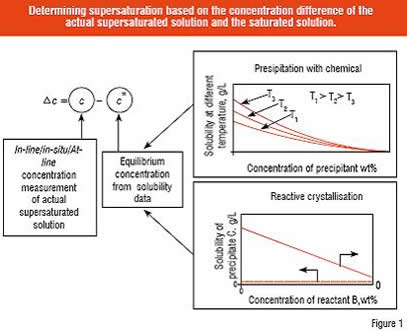
Supersaturation mainly determines the rate of crystal growth and nucleation. The general rule is: the shorter the residence time, the higher the supersaturation. Proper control of the crystal growth rate is important to obtain correctly sized crystals. Nucleation dictates the final number of particles in the batch, i.e. the higher the nucleation rate, the smaller the crystals. Moreover, it is an essential factor regarding the forming of polymorphs: what is the supersaturation level when first nuclei are formed in the solution (primary nucleation). In order to obtain the required properties of a crystalline product, it is crucial that the supersaturation level be controlled effectively by the desupersaturation rate, which is mainly determined by the crystal growth rate and the available crystal surface area. In addition, the level of supersaturation also affects the agglomeration, shape and purity of the crystals.
In-line process monitoring
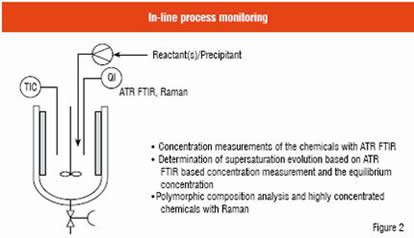
ATR FTIR spectroscopy allows simultaneous monitoring of admixture concentrations and Raman spectroscopy gives information on the polymorphism of product crystals during the semi-batch process. Figure 2 shows schematically an in-line process monitoring system when these two methods are employed. The ATR FTIR and Raman probes are immersed directly into the solution in the crystalliser. For the spectral treatment, in many cases multivariate models must be developed for the calibration modelling in order to be able to quantify the different species in the model compound system at the used temperature range.
The strength of in-line ATR FTIR spectroscopy in monitoring solid-liquid suspensions is that it is a back-reflective analysis method, i.e. crystals present in the suspension do not disturb the analysis even in high density suspensions. In addition, it is a method which can allow monitoring of several compounds at the same time so that they can be quantified simultaneously. Furthermore, its resolution is usually sufficient for the analysis of crystallisation processes. The quantification of different compounds is very useful especially for controlling precipitation systems, which usually contain different reactants, impurities, side-products, co-solvents, etc. as well as the crystallising compound. On the other hand, relatively complex multivariate models are required for spectral treatment (Pöllänen et al.). In addition to polymorphism monitoring, Raman spectroscopy can be used for highly concentrated solutions (Qu et al.). Raman spectroscopy is also a good tool to investigate stability of metastable polymorphs or pseudopolymorphs in different solvents (Qu et al.). The interpretation of Raman spectra is usually easier compared to ATR FTIR. The relative quantification of Raman results is generally done based on peak height / surface area ratios. The crystalliser initially contained sodium glutamate solution and sulphuric acid was pumped into the crystalliser at a constant feed rate. In addition to ATR FTIR data, concentration determination also requires the thermodynamic model (dissolution and mass balances of solutes) which is introduced in detail by Alatalo et al. L-glutamic acid has two reported polymorphs, an ? (thermodynamically metastable) and a ? (stable) form.
Process control of precipitation
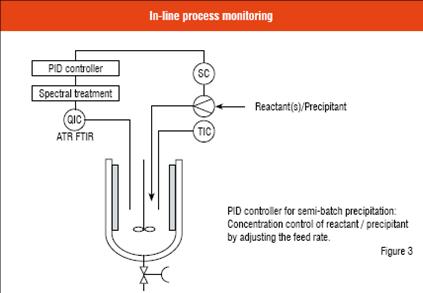
In crystallisation systems where the supersaturation levels can be determined, a feedback process control loop of the PID controller (Proportional-Integral-Derivative) can be built based on in-line concentration measurement with ATR FTIR, as shown schematically in Figure 3. As explained in the previous two sections, the aim can be to adjust the feeding rate of the reactant / precipitant based on the obtained momentary supersaturation level drawn from the ATR FTIR and solubility data and compare it to the set value. This approach makes it possible to use different precipitation policies in a controlled manner. It should be pointed out that seeding, i.e. usage of seed crystals with a desired polymorph, or other methods to induce the crystallisation may possibly be required for trouble-free processing and to ensure good uniformity between products obtained from different batches.
Future expectations
Precipitation processes are complex unit operations and are challenging to control. A summary of the fundamentals of precipitation is presented along with some basic aspects concerning in-line monitoring and process control of precipitation. In many pharmaceutical applications IR and Raman spectroscopy have been proven to be powerful tools for real-time measurements of solid-liquid suspensions. It can be expected that in future these spectroscopic methods will be more commonly utilised for process control purposes.
Acknowledgements
The author would like to thank the Crystallization Group and Chemometrics Group of Lappeenranta University of Technology for kindly providing the PAT (in-line monitoring) and multivariate data discussed in this work. The Academy of Finland (projects No. 122828 and 117155) is thanked for financial support.
References
1. Crystallization Technology Handbook, Mersmann, A., (Ed.), 2nd Edit., New York, Marcel Dekker, 2001.
2. Handbook of Industrial Crystallization. Myerson, A.S., (Ed.), 2nd Edition. Boston, Butterworth-Heinemann, 2002.
3. Palosaari, S., Louhi-Kultanen, M., Sha, Z., Industrial Crystallization, in Handbook of Industrial Drying, Mujumdar, A., (edit.), Taylor & Francis Group, LLC, 2007, pp. 1203-1224.
4. Pöllänen, K., Reinikainen, S.-P., Häkkinen, A., Louhi-Kultanen, M., Nyström, L., ATR-FTIR in monitoring of crystallization processes: Comparison of indirect and direct OSC methods, Chemometrics and Intelligent Laboratory Systems, 76(2005)1, 25-35.
5. Pöllänen, K., Reinikainen, S.-P., Huhtanen, M., Häkkinen, A., Louhi-Kultanen, M., Nyström, L., IR spectroscopy together with multivariate data analysis as a process analytical tool for in-line monitoring of crystallization process and solid state analysis of crystalline product, Journal of Pharmaceutical and Biomedical Analysis, 38(2005)2, 275-284.
6. Pöllänen, K., Häkkinen, A., Reinikainen, S.-P., Louhi-Kultanen, M. and Nyström, L., A study on batch cooling crystallization of sulphathiazole: process monitoring using ATR-FTIR and product characterization by automated image analysis, Trans.I.ChemE, 84(2006)A1, p. 47-59.
7. Qu, H., Kohonen, J., Louhi-Kultanen, M., Reinikainen, S.-P., Kallas, J., Spectroscopic monitoring of carbamazepine crystallization and phase transformation in ethanol-water solution, Industrial & Engineering Chemistry Research, in press.
8. Qu, H., Louhi-Kultanen, M., Kallas, J., Additive Effects on the Solvent-Mediated Anhydrate/Hydrate Phase Transformation in a Mixed Solvent, Cryst. Growth & Des. 7(2007)4, pp. 724-729.
9. Qu, H., Kohonen, J., Louhi-Kultanen, M., Reinikainen, S.-P., Kallas, J., Spectroscopic monitoring of carbamazepine crystallization and phase transformation in ethanol-water solution, Industrial & Engineering Chemistry Research, accepted.
10. Qu, H., Louhi-Kultanen, M., Kallas, J., Solubility and stability of anhydrate/hydrate in solvent mixtures, International Journal of Pharmaceutics 321(2006)1-2, pp. 101-107.
11. Qu, H., Louhi-Kultanen, Rantanen, J., Kallas, J., Solvent-Mediated Phase Transformation Kinetics of an Anhydrate/Hydrate System, Cryst. Growth & Des. 6(2006)9, pp. 2053-2060.
12. Alatalo, H., Kohonen, J., Qu, H., Hatakka, H., Reinikainen, S.-P., Louhi-Kultanen, M., Kallas, J., In-line monitoring of reactive crystallization process based on ATR-FTIR and Raman spectroscopy, Journal of Chemometrics, accepted.












