Moving from R&D to Manufacturing Excellence
Bart Moors, Business Consultant, Pharmaceutical Industry SEA, Siemens AG, Belgium
Relative separation of manufacturing and R&D (siloed thinking) continues to characterise the pharmaceutical industry. Managing operations in a holistic manner by investing on a common platform that meets the existing standards and integrates innovative technologies across plants and different functional departments can help the pharma industry keep ahead of future trends.
Despite the innovations and advances in science, the pharmaceutical industry has been more used to incremental change in manufacturing than quantum leaps that anticipate the future. Initiatives like Lean manufacturing, Six Sigma and other operational excellence programmes attracted the attention of the pharmaceutical industry a few decades later than in other industries.
The pharmaceutical industry is facing many challenges in order to compete in the market and to remain profitable in a sustainable manner. However, a wide range of commercial, regulatory and competitive pressures are accelerating the changes. For this reason, the drug industry has to embrace the principles of the Toyota Management System. More pharma companies are slow in implementing a systematic approach to improve there processes for research and development, manufacturing and the supply chain of a drug. Still many companies remain wary of the drastic changes, especially when it comes to introducing new technologies or changing processes.
Pharmaceutical industry – Current status At present, the pharmaceutical industry is characterised by a fragmented and siloed organisations. R&D, manufacturing, sales & marketing and other support functions are considered as independent activities, which miss a collaborative and synchronised approach. Barriers between these activities are high and it is not easy to bring them down. The same fragmentation even exists in the broader landscape which is surrounding the pharmaceutical industry. Huge discrepancies exist between pharmaceutical companies, healthcare organisations, regulatory authorities and patients. This environment fails to take customers’ (and other stakeholders’) needs into account.
One of the reasons for the success of other industries has been their ability to close the gap between suppliers, companies and the customer allied with a focus on advanced technological processes resulting in big improvements in the production quality, time to market and product innovation.
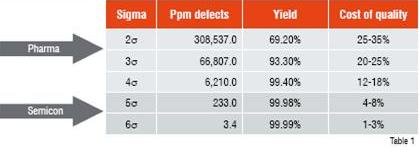
Industries such as semiconductors, food and beverages, chemicals and petrochemicals are more advanced in breaking down the barriers between stakeholders, aligning processes throughout the entire value chain and introducing innovative technologies and concepts like Process Analytical Technologies, Six Sigma, Lean Manufacturing etc.
Part of the reason for this was that the regulatory context of pharmaceutical manufacturing did not facilitate the introduction of change. Now, however, the situation is changing with the US Food and Drug Administration (FDA) recognising that stringent regulatory regulations have generated high barriers for pharmaceutical manufacturers to adopt state-of-the-art manufacturing process within the pharmaceutical industry.
Trends, challenges and opportunities
The pharmaceutical manufacturing sector has been inherently conservative in its approach to change. Historically, the regulatory framework, with its reliance on batch inspection has deterred innovation and maintained the silo-behaviour. Closing gaps between R&D and manufacturing will be a key strategy in responding to a range of drug development and quality concerns and for the ability to achieve manufacturing excellence. Closing the gaps between patient and heathcare systems will also be increasingly important for pharmaceutical companies in order to be more responsive to patient needs and demands. This will need a more flexible manufacturing concept to stay competitive. Coming from the times where the profit was high and where the pharmaceutical industry could relay on a wealthy portfolio of patents we determine today a lot of pressure on this industry by healthcare, by generic drugs manufacturing companies and by a lack of new developed drugs. This means that comparing the KPI’s of the pharma industry with the others there is an enormous bridge to gap.
An investigation shows the following average values for the pharmaceutical industry;
Scrap rework – plan for 5 – 10%
Utilisation level 15%
Cycle times 720 hours
Concepts like Lean Manufacturing, Total Performance Manufacturing (TPM) and Six Sigma are indispensible tools to achieve these goals.

Technologies and concepts to close the gap between R&D and Manufacturing
We see companies moving away from batch manufacturing with off-line product testing to continuous manufacturing with an integrated quality design into the process. Disposable manufacturing technologies will also hasten time to market, reduce or eliminate cleaning in place or Steaming in Place activities and reduce lead time, which are still very high in the pharmaceutical industry compared to other industries.
Such technologies provide a greater degree of flexibility, both in terms of scale and location and ease of use for operations. Cell chips are enabling the study of the reaction of an individual cell through the miniaturisation of different kinds of analytics and detectors. Micro reaction technology permits the synthesis of active pharmaceutical ingredients on a small-footprint, lab-bench scale. Because of continuous mode of production, the same annual product volume can be produced as when using larger-scale batch methods. An additional advantage of this technology is the elimination of the up-scaling effort and technology transfer.
Adopting productivity tools such as Knowledge Management, Product Lifecycle Management and Master Data Management tools, (MDM can also serve as a data portal), the organisations could facilitate better coordination between their R&D and manufacturing processes. It is important for information to flow not only from R&D to manufacturing but the other way round as well. The collection of knowledge through manufacturing will help the R&D organisation to accelerate the development of new processes when new products have to be designed. For example, the characteristics and behaviour of production equipment and machines in the manufacturing process can be inputs for the development process.
The Product Lifecycle Management software defines, collects and keeps track securely of all product and compliance relevant information . By providing common access to a single repository of all product-related knowledge, data and processes, PLM can speed up the innovation and launch of successful products. In the future PLM will enable companies to deliver automatically the information to support the (e-) regulatory submission and approval processes to the authorities.
Shared PLM and KM tools can work together with other solutions to help pharmaceutical companies move away from R&D and manufacturing as separate silos to deliver a more integrated and interactive manufacturing and development process, speeding the time of development. At the heart of this is the Workflow Management System and Electronic Lab Notebook (ELN)—the R&D suite that links to the production suite used in manufacturing and the PLM software. Thus, R&D dovetails into the Manufacturing Execution System (MES). This is further facilitated by the ability to use the same sample management and Laboratory Information Management System (LIMS) in both development and manufacturing.
A range of other innovative technologies complete the picture. Process Control Systems allow automation in laboratories, even for small process equipment or pilot equipment. Micro process technology introduces this technology more quickly for broad application in the laboratory and successful transfer to industrial production levels.
Finally, Process Analytical Technology (PAT) and “ The Critical Path Initiative” of the FDA will move the pharma industry towards a science-based manufacturing industry. With PAT, the pharmaceutical manufacturing environment will move towards scientific understanding of pharmaceutical materials and processes.
PAT will play an important roll in supporting companies moving from off-line quality inspections to on-line and finally towards a full real-time product release capability. The connection with Process Analytical Technologies enables companies to link all their analysers and other process measurement tools to one single system architecture. It forms a part of the process control system tightly integrated with Electronic Batch Records functions.
Knowledge management systems, data portals, totally integrated component based automation and master data systems become even more important in handling flexible, complex and dynamic interrelationships.
With this component based architecture, the requirements of the original equipment manufacturers can live up to supply their own working components and have the ability to integrate with the overall plant process system.
This requires a modular concept, based on standards, stands like OMAC and ISA S88, but also standards in the field of operation with SCADA and in the field of megatronics (Motion control).
Profinet with the supporting CBA software makes a plug and play connection possible. In this concept, the integration of Q (quality), S (supply chain) and M (asset management; OEE) aspects starting already on the lowest level, makes it easy to make this information available over the whole system and use these data to improve the manufacturing performance.
Lean, leaner, leanest!
Innovative solutions, on the one hand, close the gap between R&D and manufacturing and on the other hand, eliminate the barriers within the entire value chain, to transform the pharmaceutical industry into a more efficient business model.
Manufacturing flexibility entails supply chain visibility, production capacity and dynamic decision making, each of them being supported by the innovative technologies. Incorporating these concepts and innovative technologies into an overall strategy can be categorised as continuous improvement and Lean Manufacturing.
Determining value based on what is valuable to the customer forms the basis of Lean Manufacturing. Hence, it focusses on the silos that need to be bypassed within the pharmaceutical companies and around them. Furthermore, it keeps every activity in motion and eliminates the waste. Floorspace, work-in-progress and lead time are all interrelated. Reducing testing times is another aspect that would allow manufacturers to release products faster and reduce inventory levels.
Lean principles are very easy to understand but they are very hard to implement, especially in a regulated environment like the pharmaceutical industry where the validation and proof is required to introduce any change or new system to maintain or improve quality.
However, the present regulatory context, with its introduction of Quality by Design (FDA Guidance on Process Analytical Technology and Quality by Design) facilitates the introduction of innovative technologies (as discussed above) and concepts in order to achieve the goals of Lean Manufacturing and Six Sigma methodologies.
What is essential, is connecting Innovation, TPM, Lean and Six Sigma to business objectives directly and getting the support of management by integrating these improvement programs in a pragmatic manner. Often Lean processes can get too methodical, by which efforts can become mechanical. Further, tools and systems must be in place to support people and secure the modifications.
Conclusion
The pharmaceutical environment is characterised by disconnected processes and siloed approaches throughout the entire value chain. The key is standardising and improving the processes. This enables the flawless execution of the processes. A standardised approach doesn’t stifle creativity. It changes the way creativity is used. To stay competitive and to achieve the goals of TPM, Six Sigma and Lean Manufacturing, Pharma manufacturers must move away from the traditional approach and manage operations in a holistic manner by investing on a common platform that meets the existing standards and integrates innovative technologies across plants and different functional departments.














