Single-use / Disposable Technology
Considerations for Biopharmaceutical Facility Design
Craig Sandstrom, Principal Process Engineer Fluor Corporation USA.
Single-use / disposable technology offers a cost-effective and flexible process design for biopharmaceutical manufacturing facilities.
The biopharmaceutical industry is rapidly adopting single-use / disposable technology (SU / DT) for clinical, product launch, and commercial production facilities to provide cost-effective and flexible process capabilities (Langer and Ranck 2005). These recent technologies have requirements, advantages and disadvantages that differ from Traditional Reusable Technology (TRT). These differences need to be understood so that the advantages that SU / DT offers can be realised in new and renovated biopharmaceutical manufacturing facilities.
State-of-the-art
SU / DT was originally applied to solution storage applications in the form of disposable bags and small scale filtration applications in the form of disposable capsule filters. SU / DT is now well established in disposable flow paths, and larger scale depth filtration, cartridge filtration and viral filtration applications.
Disposable bags have also been successfully used as part of a seed bioreactor train (Morrow 2006) and intermediate and final bulk product storage (Voute et al, 2004). Current opportunities for implementing in SU / DT are summarised in Table 1.
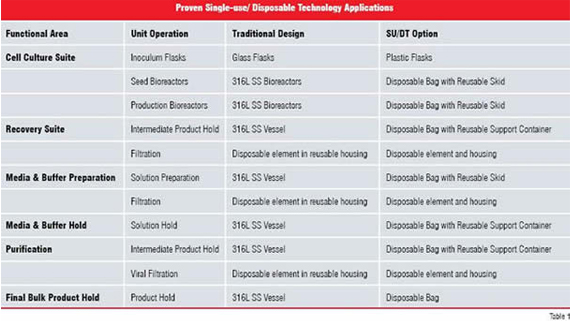
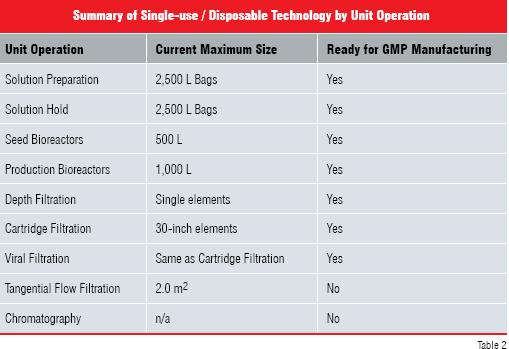
The technology is rapidly evolving and in the near future should include small-scale tangential flow filtration, chromatography and centrifugation applications. More recently, a number of vendors have started to develop larger scale (up to 1,000 L) production bioreactors, tangential flow filtration (up to 2.0 m2), depth filtration, cartridge filtration (up to 30 inch elements) and viral filtration. Additionally, there are a number of potential chromatography/ membrane chromatography devices being developed. Table 2 summarises the current maximum size available for the different biopharmaceutical unit operations employing SU / DT and indicates applications that are currently used for GMP manufacturing.
SU / DT and TRT
There are a number of fundamental differences between facilities designed for SU / DT and TRT. The primary difference is that the SU / DT-based facility will have a lower initial cost, but potentially higher operational cost, than a TRT-based facility. The overall economic evaluation is very complex, but h4ly influenced by the operational frequency, system size and complexity. Studies show SU / DT to be most economical for simple systems that are used at low frequency (Sandstrom 2003, once per week or less). Thus, simple applications such as buffer hold, media hold and intermediate product hold applications, and less frequently used seed bioreactor applications are ideal for SU / DT.
An important feature of disposable bags is their relatively low initial costs. This is very attractive on projects where the candidate product is still in clinical trials, or for facilities that experience recurring modification. Disposable bags offer a great deal of flexibility for contract manufacturing, clinical trial and initial product launch facilities that require frequent reconfiguration.
Traditional biopharmaceutical manufacturing facilities are piping-and-clean room-intense. Innovations developed to address these issues have driven layouts based upon equipment adjacencies to minimize piping runs, locating process equipment—to the extent possible—in non-classified manufacturing space. While this minimises cost, it results in multistory facilities with relatively inflexible designs. In contrast, single-use technology tends to involve portable equipment and flexible hose connections.
These issues drive a very different type of layout based on material flows and have primarily involved extensive use of clean rooms. As the SU / DT technology has become more accepted, disposable bags used for buffer and media storage have moved from clean rooms to unclassified space, with a further reduction in capital cost.
SU / DT has been developed primarily for smaller scale, portable applications. Consequently, a primary feature of facilities designed around SU / DT has to deal with portable equipment.
Portable equipment requires staging space and space for equipment movement. This requires a facility with a more robust design (floors, doors and walls) to handle the portable equipment traffic. Additionally, the material flow-driven nature of the layout favours a single-level, as opposed to a multilevel, production operation.
The smaller scale purification process utilising buffers supplied in disposable bags leads to interesting material flow and layout challenges. In many instances, the equipment footprint required for the buffer containers is greater than the process equipment footprint. This can be readily visualised for a 63 cm chromatography operation supported by two 1,000 litre buffer bags; two 500 litre buffer bags, two 250 litre buffer bags and two 500 litre product bags.
The footprint for the chromatography pumping skid and chromatography column is approximately 1 metre wide by 3 metres long. Each buffer container is approximately 1.3 metres by 0.9 metres. This requires that the buffers are either stacked, temporarily staged or share the buffer staging space with adjacent unit operations to minimise the required processing area.
Warehouse, in-process storage and waste disposal are important issues to consider. By utilising significant quantities of single-use/disposable items, the warehouse and in-process storage requirements are significantly increased over facilities utilising traditional reusable technology. Space should be strategically located to assemble the single-use/disposable items near their points of use to minimise material traffic. Additionally, a room should be located near the glass-wash and waste dock to disassemble items.
Level of automation differences
Many different types of instrumentation are employed to perform online analysis (pH, dissolved oxygen, conductivity, temperature and pressure) and flow control (on/off and control valves) in biopharmaceutical applications. All of these, with the exception of temperature and pressure, require an instrument to be in contact with the process solution. Many of these online probes currently lack equivalent SU / DT counterparts. SU / DT can employ automated pinch valves for on/off flow control, but lack an equivalent flow-control valve.
However, some degree of flow control can be achieved using peristaltic pumps. This limitation frequently leads to hybrid systems that employ reusable probes with SU / DT, leading to components that still require cleaning and sanitisation. A number of vendors are now developing disposable sensors to avoid the necessity of using reusable probes. However, these appear to be a long way from being commercially available.
By their very nature, SU / DT requires more extensive set-up that readily lends itself to manual operations. Together with current limitations in instrumentation availability results in SU / DT-based facilities that favour a lower automation level. This also has the advantage of decreasing the initial project capital cost.
Intangible issues
Beyond cost, scale and layout issues, there are a number of intangible issues that are important to consider when evaluating SU / DT (Sandstrom and Schmidt 2005). These intangible considerations include:
- Mixing
- Line sizes and connection types .
- Container integrity
- “Practical” vs. “Possible”
- Leachables, extractables and chemical compatibility
- Safety
- Flexibility
- Risk
Both disposable bags and reusable vessels can be mixed, although the degree of agitation is different. Containers are vigorously agitated to rapidly blend materi als, gently agitated to provide a uniform sample and prevent segregation, and moderately agitated to enhance heat transfer. The degree of agitation can be almost arbitrarily selected for fixed vessels. However, the degree of agitation in bags is generally limited. This requires careful evaluation and procedure development for media/buffer preparation applications to ensure successful design and operations when using disposable bags.
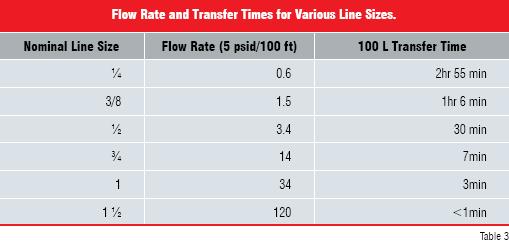
SU / DT applications have limitations on the available line sizes and connection types. Table 3 shows flow rates for water through various nominal line sizes and the corresponding time required to transfer 100 L. A significant factor is the need for an aseptic or hygienic connection. Aseptic connections are currently limited to ½-inch line sizes. Sterile tube welders are available for line sizes up to ?-inch, but their reliability has been questionable. Hygienic connections are available in larger line sizes, with 1½- inch being the current upper limit. These line size limitations impose significant practical limitations on usable disposable bag capacities.
Leachables, extractables and chemical compatibility is an issue for both traditional reusable and SU / DT. Corrosion is the primary concern in traditional reusable technology, with high chloride/low pH solutions presenting the most significant challenges for stainless steels (Peckner 1977). For SU / DT, it is very important for material compatibility testing to be performed with the process solutions of interest.
Cleaning and sanitisation requirements are very different between SU / DT and TRT. A significant advantage with SU / DT is that they can be presterilised, and because they are disposable, they do not require cleaning. The clean-in-place (CIP) and clean steam systems required for TRT involve significant capital investments and their elimination, or at least minimisation, are a major part of the cost savings associated with SU / DT.
Container integrity and safety may also be an issue with SU / DT. Disposable bags need to be protected from accidental puncture and loss of container integrity. Additionally, disposable bags have a burst pressure of approximately 15 psig, and their design does not facilitate relief device installation. Finally, disposable bags will melt when exposed to excessive heat, for example, from a fire. Disposable bag integrity and safety can be enhanced using the appropriate support containers. These containers provide a ridged barrier to prevent accidental puncture and can minimise any splashing associated with loss of containment. Even with these, however, disposable bags may not be suitable for use with flammable or hazardous solutions.
Finally, the use of single-use/disposable bioreactors may be limited to GLSP and BL1-LS biosafety-level applications where closed systems require designs to “reduce the potential for biohazard release” as opposed to the BL2-LS where closed systems require designs to “prevent potential biohazard release” (NIH Guidelines 2002).
SU / DT offers a more flexible design option than TRT. This is especially important for facilities that routinely undergo reconfiguration, such as multiproduct, clinical trial and pilot facilities. A disposable bag size can be more readily changed than a fixed vessel size. Additionally, a disposable bag can be moved to accommodate process changes more easily and with less cost and associated construction time than required to remove and install new process piping.
There are risks associated with implementing SU / DT. These risks are categorised in Figure 1. The supply chain management risk can be mitigated by qualifying at least two SU / DT vendors. This risk can also be mitigated with a larger warehouse inventory to allow time to investigate suitable alternatives in the event of a supply chain disruption.
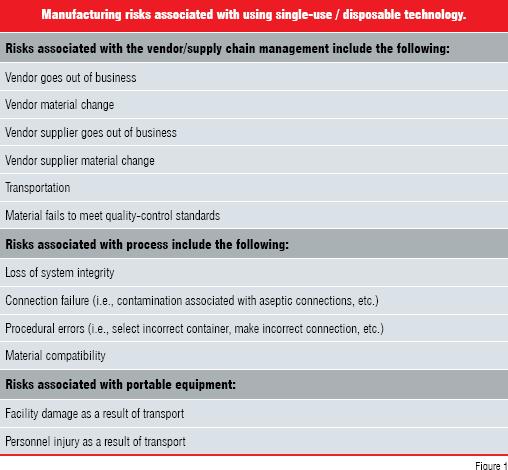
Of the risks associated with the various process applications, the breach of container integrity and failure of aseptic connections are the most serious. Integrity testing disposable bags before use is very important for applications with potential high-value product loss. Operator training and selection of robust connections are very important for applications requiring aseptic connections.
Summary
Single-use/disposable systems are now a well-established technology that offers a cost-effective and flexible process design for clinical, initial product launch, and commercial production biopharmaceutical facilities. Their requirements, advantages and disa vantages lead to different facility designs when compared with TRT. Facilities designed for SU / DT have layouts that are driven by material flow issues and favour a lower level of automation compared to facilities designed for TRT that are driven by equipment adjacencies and may have a high level of automation. Additionally, SU / DT provides flexibility to accommodate process modification that may otherwise require facility renovations, and thus minimise or avoid subsequent construction costs and lost production time. Overall, the cost advantages and flexibility makes SU / DT especially attractive for small, multiproduct facilities, clinical trial and initial product launch facilities, and pilot facilities.
References
(1) Sandstrom CE. The Economics of Storing Process Solutions. Chem. Engr. April 2003:36-45.
(2) Langer ES, Ranck J. The ROI Case: Economic Justification for Disposables in Biopharmaceutical Manufacturing. BioProcess International Supplement. October 2005, 46-50.
(3) Voute N, Dooley, T, Péron G, Lee E. Disposable Technology for Controlled Freeze-Thaw. BioProcess International Supplement. October 2004: 40-43.
(4) Morrow KJ. Disposable Bioreactors Gaining Favor. Genetic Engineering News. June 15 2006 (Vol. 26, No. 12)
(5) Peckner D, Bernstein IM. Handbook of Stainless Steels. McGraw-Hill Book Company: New York, NY, 1977.
(6) Sandstrom C, Schmidt B. Facility-Design Considerations for the Use of Disposable Bags. BioProcess International Supplement. October 1005, 56-60.
(7) NIH Guidelines for Research Involving Recombinant DNA Molecules (NIH Guidelines) April 2002












