Advanced Drug Delivery Strategies for Inflammatory Bowel Disease
Julia Mantaj, Lecturer, Biomedical Science, Anglia Ruskin University
This article explores innovative drug delivery strategies for the treatment of inflammatory bowel disease (IBD). Novel approaches based on enteric-coated microneedle pills, nanoparticles, liposomes, prodrugs, hybrids, and biological systems will be discussed with regards to improving therapeutic outcomes and reducing side-effects in patients with IBD.

Crohn’s Disease (CD) and Ulcerative Colitis (UC) are the two main forms of IBD affecting more than 10 million people worldwide. These are chronic and progressive conditions, characterised by destructive inflammation of the intestinal tract. With time, the intestinal wall in IBD patients becomes ‘leaky’, and harmful bacteria from the gut microbiota can enter the blood circulation triggering an immune response and intensifying gut inflammation (Figure 1).

Symptoms of IBD include fever, anaemia, weight loss, diarrhoea, rectal bleeding, abdominal pain, and the urgency to evacuate the bowels. IBD is recognised to significantly impact the quality of life (QoL) of patients. Presently, IBD is not curable. Much effort has been made in developing therapeutic strategies for the treatment of IBD aiming to reduce symptoms, maintain clinical and endoscopic remission, and prevent long-term disability. These therapies target the intensified inflammation process by decreasing the body’s immune response (i.e., immunomodulators azathioprine and methotrexate) or blocking the inflammation (i.e., biologics infliximab and ustekinumab). Despite the significant advancements in disease treatment, conventional drugs have major limitations such as severe adverse reactions (e.g., nausea, fever, higher risk of infections, diabetes) due to systemic absorption leading to therapy failure or ineffectiveness. Furthermore, a considerable proportion of IBD patients are unresponsive to the advanced therapies (biologics) or lose the response over the course of the disease. A study carried out by Gibble et al revealed that over 60 per cent of IBD patients responded inadequately to their first advanced therapies within 1 year after initiation. Consequently, over time, surgical removal of the inflamed bowel part is necessary. Approximately 23-45 per cent of UC and up to 80 per cent of CD patients will require colon surgery at some point during their disease.
The major challenge in developing IBD therapeutic strategies is the delivery of therapeutics directly to the inflamed colon site. Traditional intestinal delivery systems are stimuli-responsive meaning that drugs are released following certain stimuli i.e., changes in pH, temperature, transit time, or presence of enzymes under healthy physiological conditions. However, these stimuli are different in IBD, and it is still unclear how they contribute to disease pathophysiology.
In recent years, a plethora of innovative strategies have been reported for the treatment of IBD aiming to achieve site-specific drug delivery to the inflamed tissue, thereby reducing adverse drug effects and improving the efficacy.
Enteric-coated microneedle pills
Initially developed for the transdermal delivery of small drug molecules and macromolecules, enteric-coated microneedle pills have gained attention in recent years as oral dosage forms. A research team at the Massachusetts Institute of Technology and Massachusetts General Hospital Harvard Medical School developed a capsule coated with drug-loaded microneedles capable of injecting drugs directly into the intestinal lining following ingestion (Figure 2).

Animal studies demonstrate that the dosage form delivered insulin more efficiently compared to subcutaneous administration with no harmful side-effects observed. The microneedle capsules safely passed and were excreted from the gastrointestinal tract (GIT) making it suitable for use in the inflamed GIT in IBD. The incorporation of enteric coating onto microneedles enables the pills to protect incorporated drugs from the harsh acidic environment of the stomach and deliver them directly to the site where they are required. In a pre-clinical study, Rani Therapeutics tested anenteric-coated microneedle pill loaded with the TNF-αblocker adalimumab. The study results showed that the technology protects adalimumab from the acidic and enzymatic degradation of the GIT and directly delivers it to the site of inflammation where it slowly releases the drug. In summary, enteric-coated microneedle pills represent a promising drug delivery strategy, especially biological compounds for the therapy of IBD. However, further rigorous research is necessary to evaluate their clinical long-term effectiveness and safety in the management of IBD.
Nano-delivery system strategies
Nanoparticulate (NP) systems represent a promising approach to delivering drugs to inflamed colonic tissues, thereby offering the advantage of reducing the drug dose and systemic side-effects. Macrophages, neutrophils, and M cells that are present in inflamed intestinal regions can easily take up the NPs due to their small size (1-100 nm). Triggered by inflammatory cytokines (e.g., TNF-α, IL-1α, IL-6), in the inflamed colon, the intestinal permeability is increased due to the loss of integrity of intestinal tight junctions, cell-to-cell contacts, and immune cell infiltration that is also referred to as ‘leaky gut’. The loss of intestinal integrity enables the NPs to easily penetrate mucosal tissues and transport drugs to the inflamed colon sites. The NPs can be designed to display distinctive attributes and physicochemical properties (size, surface charge, ligands, targeting moieties) that can contribute to the extended retention of drugs at inflammation sites due to e.g., nanoparticle adhesion to mucus (Figure 3).

Over the past decade, considerable research efforts have been made to advance nano-delivery system strategies for the treatment of IBD. For example, a study by Ali et al revealed a significant accumulation of 3 nm poly(lactic-co-glycolic acid) (PLGA) NPs on the colonic mucosal surface in mice compared to 250 nm NPs. To date, the data obtained is predominantly based on in vitro, ex vivo tissue binding studies, or in vivo studies following rectal administration using animal models. To further evaluate the NP behaviour in humans, further comprehensive studies are necessary for a definitive conclusion.
Prodrugs
Prodrug systems consist of a pharmacologically active agent that is protected in a temporarily inactive form that becomes bioactive upon specific stimuli, in the case of IBD these are enzymes that are overexpressed in inflammatory tissues. Most studied prodrug systems employ 5-ASA derivates (mesalamine/5-aminosalicylic acid) that are metabolised to the active form by the N-acetyltransferase overexpressed in many IBD patients. Additionally, amino acids and carbohydrates (e.g., dextrans, cyclodextrins) have been studied as carrier systems for 5-ASA and dexamethasone. The drugs bind to the conjugate via glycosidic bonds that are cleaved by the colonic glucanase and esterase releasing the free active drug in the colon, thereby achieving target-specific drug delivery. Despite representing one of the most practical approaches for targeted drug delivery treatment in IBD, the development of novel prodrug delivery is challenging. For instance, only a few studies are reporting the utilisation of prodrug delivery systems for the treatment of IBD highlighting the immaturity of the development of these systems. Additionally, extensive safety and stability in vivo evaluation of the systems is required.
Liposomes
Liposomes are nanosized bubble structures consisting of one or more phospholipid layers designed to deliver hydrophilic and lipophilic drugs (Figure 4).
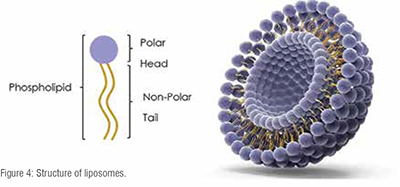
The hydrophilic drugs are encapsulated in the aqueous liposome core, whereas hydrophobic compounds can dissolve into the lipid membrane. A wide range of modified liposomal drug systems are being investigated in experimental colitis and the accumulation efficacy and the improvement of clinical IBD symptoms are being intensively evaluated. One option to specifically deliver drugs to the inflamed colon in IBD is the modification of the liposome surface with cationic lipids which results in liposome binding to mucosal tissues. For example, Myers et al developed a cationic liposome complex loaded with antisense TNF-α oligonucleotide that demonstrated a decrease in TNF-α mRNA expression in in vitro studies. These observations were further supported by in vivo experiments where the administration of the liposome complex resulted in reduced Disease Activity Index scores in a murine model of colitis. Furthermore, the prolonged circulation of liposomes in the body can be achieved via PEGylation (covalent linking of polyethylene glycol chains) that can be functionalised with various drugs for targeted drug tissue accumulation. However, cationic liposomes have shown toxicity when administered systemically due to their abnormal aggregation with proteins in the circulation. Hence, further research into liposome functionalisation to reduce systemic toxicity is required.
Hybrid delivery systems
Hybrid drug delivery systems combine the advantages of multiple carriers within a single structure. The drugs are encapsulated in a carrier, and these drug-loaded carriers are then embedded in an additional protective external compartment that degrades in inflamed tissues. This external compartment selectively dissolves or degrades in the inflamed intestinal region, releasing the embedded drug-carrier complex specifically at the target site. Xiao et al designed a hydrogel hybrid carrier system that releases CD98 antibodies incorporated in NPs. The systems demonstrated a reduction in disease severity in animal models. Furthermore, Li et al fabricated structures consisting of hyaluronic acid-modified porous silicon NPs loaded with budesonide encapsulated in a pH-responsive matrix. After oral administration, in vivo studies revealed decreased expression of proinflammatory cytokines and enhanced therapeutic efficacy.
Biological delivery systems
This novel approach exhibits significant therapeutic potential in IBD treatment involving bacteria, cells, and other biosystems for targeted drug delivery.
Bacteria
The advancement of genetically engineered probiotic bacteria offers a novel approach to utilising probiotics as drug carrier systems for the delivery of IBD medications to inflamed colon sections. Several research groups investigated the delivery of anti-inflammatory cytokine IL-10, anti-TNF-α nanobodies, and immunomodulatory proteins by Lactococcus lactis. The study findings demonstrated reduced levels of proinflammatory cytokines after oral administration. Despite the advantages of administration of genetically engineered Lactococcus lactis expressing anti-inflammatory molecules (non-invasive, allochthonous, endotoxin-free) biological containment poses a risk of transgene escape remains. Furthermore, drug delivery systems based on bacteria probably have a limited effect on IBD due to colonisations only at specific niches in the colon.
Cells
Red blood cells (RBC) offer an advantage in delivering drugs to target sites due to their features such as high biocompatibility and long circulation time in blood. A clinical trial conducted by Castro et al investigated the safety and efficacy of autologous RBCs infusion loaded with dexamethasone 21-phosphate in the maintenance of long-term remission in children with steroid-dependent CD. The endoscopic results showed a significant therapeutic effect revealing 44 per cent of disease remission in patients. Furthermore, subsequently, a sustained efficacy and safety of this treatment was reported during the next six years in 50 per cent of the steroid-dependent CD patients who continued the study. Despite these encouraging results, it should be noted that drug-loading strategies can pose a risk to the structure and function of RBCs. Additionally, further research is necessary to improve drug stability during delivery and the release of drugs immediately after reaching the inflamed tissues. Lastly, the safety of the drug-loaded RBCs in vivo necessitates extensive evaluation.
Conclusion
Novel targeted drug delivery systems not only enhance therapeutic efficacy by delivering drugs in a targeted and controlled manner directly to the affected regions of the inflamed colon in IBD but also reduce the systemic side-effects associated with conventional IBD treatment. A plethora of drug delivery systems are currently under investigation and further research is required to evaluate their efficacy and safety. Despite the challenges in delivering drugs to the inflamed colon in IBD, the new strategies hold great promise in improving IBD symptoms and the quality of life of patients.













