Advancing Toxicology and Drug Testing
The use of live invertebrate models
Vitaliy Khutoryanskiy, Reading School of Pharmacy and Physicochemical, Ex Vivo and Invertebrate Tests and Analysis Centre (PEVITAC), University of Reading
Invertebrates offer ethical, cost-effective alternatives to mammalian models for toxicological and antimicrobial testing. Models like slugs, planaria, and greater wax moth larvae enable assessment of irritancy, epithelial damage, infection studies and immune responses. Their ease of use and physiological relevance highlight their growing value in preclinical research and disease modelling.

The advancement of novel chemicals and formulations across diverse applications underscores the strong need to assess their safety for humans, animals, and the environment. For an effective evaluation of the toxicological properties of these substances, the tests should provide reliable information, be economically viable, technically straightforward, and able to produce results promptly.
The use of mammals for toxicological testing of new chemicals and formulations has raised significant public concern in recent decades. This highlighted the need for alternative testing methods that are both ethically sound and scientifically reliable.
Invertebrates , comprising over 90 percent of all living animal species, present a promising alternative for replacing mammals in toxicological testing. Laboratory research involving invertebrates has a rich history, marked by numerous Nobel Prize-winning discoveries. For instance, Thomas H. Morgan used the fruit fly Drosophila melanogaster as a model, leading to breakthroughs in understanding the role of chromosomes in heredity. Additionally, the nematode Caenorhabditis elegans served as a vital model organism, contributing to multiple Nobel prizes. Research on C. elegans has unravelled similarities in genetic and molecular mechanisms of organ development and programmed cell death between humans and the organism. Studies on gene conservation between species and elucidation of fundamental mechanisms of gene regulation have also been illuminated through research on C. elegans. Furthermore, the use of green fluorescent protein in C. elegans research has facilitated investigations into touch sensitivity and gene expression.
Using live invertebrates for toxicological testing brings several advantages.
Firstly, there is no need for ethical licences, as invertebrates are often exempt from the ethical concerns associated with vertebrate research. This makes their use more socially acceptable. Additionally, invertebrates are generally cost-effective, both in terms of acquisition and maintenance in laboratory settings, compared to vertebrates. Their high reproductive rates and ease of breeding further contribute to a sustainable and readily available source for experiments. Moreover, some invertebrates share essential biological processes with mammals, providing meaningful parallels in toxicological responses. This biological similarity allows researchers to draw insights from invertebrate studies and potentially extrapolate the results to understand the effects in mammalian systems. In certain instances, experiments with invertebrates can be conducted using high-throughput approaches, offering the advantage of swiftly screening multiple formulations.
Our research group at the University of Reading is actively investigating the use of live invertebrates for toxicological screening of chemicals and pharmaceutical formulations, including polymeric excipients for drug delivery. A key method in our studies is the slug mucosal irritation test (SMIT), a method pioneered by researchers from the Ghent University. This assay has proven effective in screening formulations for their potential to induce mucosal irritation, showcasing a robust correlation between data obtained in slugs and traditional mammalian models.
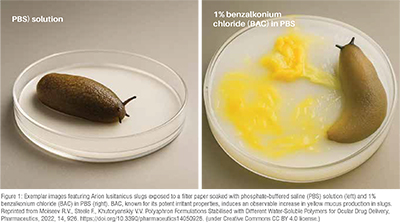
The SMIT test operates on a straightforward principle: when exposed to an irritant sample, slugs release mucus, and the quantity of this secretion can be easily quantified through gravimetric measurements (Figure 1). This approach provides a reliable and simplified means of assessing the mucosal irritation potential of various substances, offering valuable insights for the development and screening of chemical formulations. In addition, mucus production can also be assessed using fluorescence assays, as described in the literature, providing a complementary and sensitive method for evaluating epithelial disruption and irritancy.
Planaria, aquatic free-living flatworms inhabiting freshwater, marine, and terrestrial environments, stand out for their exceptional regenerative capacities. This unique feature positions them as valuable models for research in tissue regeneration and stem cell biology. Notably, aquatic planaria are easily bred in laboratory settings, enhancing their suitability for scientific studies. Moreover, their adaptability makes them particularly useful in the development of toxicological screening tests, further highlighting their significance as versatile organisms in various research domains.
An innovative planarian toxicity fluorescent assay has recently been developed as a cost-effective and rapid prescreening tool for identifying potential skin irritants. This assay leverages the protective barrier function of planarian epithelia. Upon exposure to an irritant sample, the epithelia undergo disruption, rendering them more permeable to fluorescein sodium. This increased permeability is easily quantified through fluorescence microscopy and image analysis, providing a reliable method for assessing the impact of substances on planarian skin and offering valuable insights into potential irritant effects. The principles of planarian toxicity fluorescent assay are shown in Figure 2.
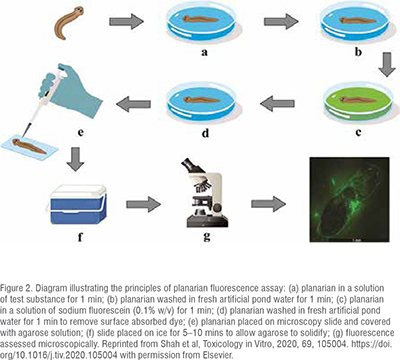
This test has been applied in investigations involving novel polymeric excipients, with results systematically compared to those obtained from in vitro cell culture assays.
The potential of planaria as a model organism is being explored for the development of rapid, cost-effective, and reliable tests to predict the toxicological properties of chemicals and liquid formulations. In addition to the planarian toxicity fluorescent assay, current research leverages the species' innate locomotion in solution and notable regenerative capabilities. Prior research has already demonstrated the noteworthy influence of certain chemicals on both the mobility and regenerative capacity of planaria.
Galleria mellonella, larvae of the greater wax moth, are an emerging new model for in vivo testing. These larvae possess a well-developed innate immune system with remarkable parallels to that of vertebrates, making them highly suitable for studying microbial infections, immune responses, and the toxicity of pharmaceutical formulations. Galleria mellonella, can be incubated at mammalian body temperatures and support a variety of administration routes, including injection, oral, and topical delivery. Their ease of handling, low maintenance costs, and exemption from many ethical regulations further enhance their appeal as an alternative in vivo model. This live invertebrate model has been widely adopted for assessing antimicrobial efficacy, drug safety, and host–pathogen interactions, demonstrating significant potential to complement or reduce reliance on traditional mammalian models.
The potential applications of Galleria mellonella are currently being investigated in two key areas: (1) assessing the antimicrobial activity of various compounds, and (2) evaluating their toxicological properties. Injection of pathogenic bacteria such as Staphylococcus aureus or Escherichia coli into Galleria mellonella larvae leads to rapid infection progression, often resulting in death if left untreated. The development of infection is accompanied by visible melanisation of the larvae's body, with colour changes from creamy white to dark brown or black, indicating an immune response (Figure 3).

However, subsequent injection of antimicrobial agents—such as antibiotics, cationic polymers and peptides, or nanoparticles—can inhibit infection and improve larval survival. Experimental observations frequently revealed that certain compounds exhibited toxicity in infected larvae, despite demonstrating little or no toxicity in uninfected counterparts. This highlights the importance of evaluating both antimicrobial efficacy and potential toxicity within the context of infection, as host-pathogen interactions can significantly influence the safety and effectiveness of therapeutic agents.
Excellent progress has already been made in recent literature on the use of live invertebrates for studying toxicity, disease models, and biological activities of drugs and materials. However, many promising opportunities in this area remain underexplored. The development of novel assays employing invertebrate models, including planaria and greater wax moth larvae, offers significant potential for delivering rapid, reproducible, and ethically responsible testing solutions. These models offer distinct advantages, including ease of breeding and suitability for high-throughput screening. Their broader adoption could substantially reduce—and in some cases replace—mammalian models in early-stage pharmaceutical research, aligning with the 3Rs principles (Replacement, Reduction, and Refinement).
Beyond the invertebrates covered in this article—slugs, planaria, and wax moth larvae —there is strong potential to develop new tests using other models such as Caenorhabditis elegans, Drosophila melanogaster, Daphnia magna, and Bombyx mori. These species offer established genetic tools and experimental protocols that can be harnessed to assess drug toxicity, biological activity, and disease mechanisms. Broadening the use of this approach can contribute to the development of ethical, economical, and mechanistically insightful models that enhance drug discovery and toxicological studies.
References
1. Wilson-Sanders S.E., Invertebrate Models for Biomedical Research, Testing, and Education, ILAR Journal, 2011, 52, Pages 126–152, https://doi.org/10.1093/ilar.52.2.126
2. Adriaens E., Remon J.P., 1999. Gastropods as an evaluation tool for screening the irritating potency of absorption enhancers and drugs. Pharm. Res., 1999, 16, 1240–1244.
https://doi.org/10.1023/A:1014801714590
3. Khutoryanskaya O.V., Morrison P.W.J., Seilkhanov S.K., Mussin M.N., Ozhmukhametova E.K., Rakhypbekov T.K., Khutoryanskiy V.V., 2014. Hydrogen-bonded complexes and blends of poly(acrylic acid) and methylcellulose: nanoparticles and mucoadhesive films for ocular delivery of riboflavin. Macromol. Biosci., 2014, 14, 225–234. https://doi.org/10.1002/mabi.201300313
4. Moiseev R.V., Steele F., Khutoryanskiy V.V. Polyaphron Formulations Stabilised with Different Water-Soluble Polymers for Ocular Drug Delivery, Pharmaceutics, 2022, 14, 926. https://doi.org/10.3390/pharmaceutics14050926
5. Shah S.I., Williams A.C., Lau W.M., Khutoryanskiy V.V. Planarian toxicity fluorescent assay: A rapid and cheap pre-screening tool for potential skin irritants, Toxicology in Vitro, 2020, 69, 105004. https://doi.org/10.1016/j.tiv.2020.105004
6. Buang F., Fu M., Chatzifragkou A., Amin M.C.M., Khutoryanskiy V.V. Hydroxyethyl cellulose functionalised with maleimide groups as a new excipient with enhanced mucoadhesive properties, Int. J. Pharm., 2023, 642, 123113. https://doi.org/10.1016/j.ijpharm.2023.123113












