DIGITAL SOLUTIONS IN DRUG DEVELOPMENT
Regulations to enable safe, effective, and high-quality solutions
Shrishaila Patil, Vice President, Statistical Programing, Navitas Data Sciences, a part of Navitas Life Sciences (a TAKE Solutions Enterprise)
From mobile medical apps and fitness trackers, to software that supports clinical decisions doctors make every day, digital technology has been driving a revolution in healthcare. As we are learning to live the virtual new normal in COVID-19 era, adoption of digital solutions has witnessed further acceleration. To keep pace with this promising innovation, the regulators must modernize its approach to regulation. Question to ask ourselves is “How to ensure high-quality, safe and effective digital health products”. This article highlights changing Regulatory landscape (European/FDA Guidelines) for enabling Safe, effective, high-Quality & trustworthy AI/Digital solutions in Pharma.
From mobile medical apps and fitness trackers, to software that supports the clinical decisions that doctors make every day, digital technology has been driving a revolution in healthcare. As we learn to live in the new normal, forced by the COVID-19 pandemic, adoption of digital solutions has witnessed further acceleration.
To keep pace with this promising innovation, regulators must modernise their approach to regulation.
The question is “How can we ensure high-quality, safe, and effective digital health products”.
This article focuses on the changing Regulatory landscape (European and FDA Guidelines) for enabling safe, effective, high-quality and trustworthy Artificial Intelligence (AI) and Digital solutions in Pharma.
With COVID-19 acting as a catalyst, just what is changing?
In the past few years, many innovations and novel approaches across Clinical Trial processes have been witnessed.
To a degree, COVID-19 has acted as a catalyst to some of these great innovations and adaptations.
Real World Data (RWD)/Evidence
- To leverage diverse data sources and analytics tools to enhance study design and protocol optimisation, capturing clinical trial data more efficiently.
- To make healthcare data both tangible and manageable using novel data analytics to extract insights, to aid a better understanding of disease. RWD can come from several sources, for example:
- Electronic Health Records (EHRs)
- Claims and billing activities
- Product and disease registries
- Patient-generated data, including in home-use settings
- Data gathered from other sources that can inform on health status, such as mobile devices
Digital Data collection methodologies: Mobile Technology, Wearables, ePRO, eCOA, etc.
- To empower robust data capture, whether reported by the patient or by leveraging novel sensors. For example, we have seen the Apple Heart Study with Stanford Medicine with over 400,000 people participating to identify irregular heart rhythms.
Conventional Clinical Trials to Virtual Trial designs
- To Improve patient recruitment, retention, and real-time access to data for improved Quality
- Sponsors need to ensure that vendors have enough industry, technological, and regulatory expertise to create a virtual trial environment
- Training is required for both the personnel and patients involved in the trial in order to understand the tools used
- A hybrid model can improve the patient experience while, at the same time, giving sponsors a low risk jumping off point to familiarise themselves with the virtual trial environment
Routine monitoring to Risk-based and centralised monitoring
- An improved and efficient approach to Clinical Trial conduct, with better oversight by moving away from 100 per cent source data verification. ICH E6 (R2) has really encouraged this approach
- Real-time remote monitoring facilitating decentralised healthcare
Digiceuticals or Digital Therapeutics
- Digital therapeutics are evidence-based therapeutic interventions driven by highquality software programs to prevent, manage, or treat a medical disorder or disease. Some examples include:
- FDA Approved Bluestar® Phone App (from Company Welldoc) for managing diabetes and is the only FDA cleared and reimbursable Software as a Medical
Device that works with a patient’s existing device.
- FDA Approved reSET to help those with opioid use disorder
Personalised Medicine
- Making sense of genomic data and personalised adaptive treatment, dosing plans for each patient
- Medical treatment tailored and adaptable to the patient based on their predicted response or risk of disease
Leveraging Big Data, Robotic Process Automation, Artificial Intelligence, Machine Learning, Deep Learning, and Natural Language Processing
- Using technologies to unlock valuable new insights from the rich and diverse data available
What we need to know is how this technological transformation impacts:
- Patients
- Study/Protocol Design
- Regulatory Compliance
- Data Quality
- Investigators
- Pharma Companies
- CROs
- Biometrics Process/CDM functionality
- Scientific/Technical/Data Standards
As a core part of clinical trial teams, each of us will have a significant role to play as we experience this exciting time of transformation.
We need to have a new mindset and adapt our SOPs, processes, and approach accordingly in order to accommodate these changes to the way that Clinical Trials are conducted, Data is captured, analysed, and reported.
As we adapt, we need to continue to safeguard Data Integrity, Quality, and Security by ensuring compliance to changing requirements from Global regulatory bodies.
US FDA - Key Guidelines and initiatives:
The 21st Century Cures Act (Cures Act), signed into law on December 13, 2016, is designed to help accelerate medical product development and bring new innovations and advances to patients who need them faster and more efficiently.
As of today, more than 30 AI algorithms have been approved by the US FDA including those for the detection of diabetic retinopathy, stroke, brain haemorrhage, and atrial fibrillation.
The FDA released its Digital Health Innovation Action Plan in 2017 clarifying the FDA’s role in advancing safe and effective digital health technologies and addressing Key provisions of 21st Century Cures Act.
The FDA released its Policy for Device Software Functions and Mobile Applicationsi in 2019. This guidance explains how the Agency plans to regulate software that aids Clinical decision support (CDS), including software that utilises machine-learning-based algorithms.
The FDA is to focus regulatory oversight on higher-risk software functions, such as those used for more serious or critical health circumstances as well as Software that utilises machine-learning based algorithms, where the user might not readily understand the program’s ‘logic and inputs’ without further explanation
Software Precertification (Pre-Cert) Pilot Program:
The Software Pre-Cert Pilot Program will help inform the development of a future regulatory model. This model will provide more streamlined and efficient regulatory oversight of software-based medical devices developed by manufacturers who have demonstrated a robust culture of quality and organisational excellence, and who are committed to monitoring real-world performance of their products once they reach the market.
The goal of the program is to have tailored, pragmatic, and least burdensome regulatory oversight that assesses organisations (both large and small) to establish trust that they have a culture of quality and organisational excellence such that they can develop high-quality Software as a Medical Device (SaMD) products Figure 1.
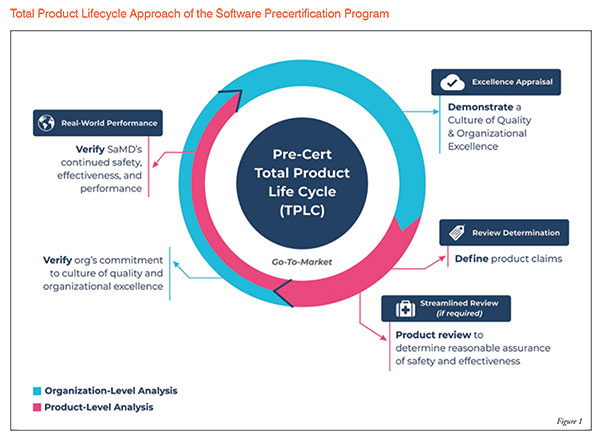
The FDA's Total Product Life Cycle (TPLC) approach enables the evaluation and monitoring of a software product from its pre-market development to post market performance, along with the continued demonstration of the organisation's excellence:
- Excellence Appraisal
- Review Determination
- Streamlined Review
- Real-world Performance
Achieving FDA Approval
FDA Approval Status could be put at risk after each update or new iteration for AI-powered therapeutic/diagnostic tools. For example: Security Updates, adding new features or functionalities, or updating an algorithm, etc. Planning to take a version-based approach to the FDA approval process might be good idea. In this approach, a new version of software is created each time the software's internal ML algorithm(s) are trained by new set of data, with each new version being subjected to independent FDA approval.
FDA's TPLC approach on AI/ML workflow:
The highly iterative, autonomous, and adaptive nature of these tools requires a new, TPLC regulatory approach that facilitates a rapid cycle of product improvement and allows these devices to continually improve, while prov Figure 2.
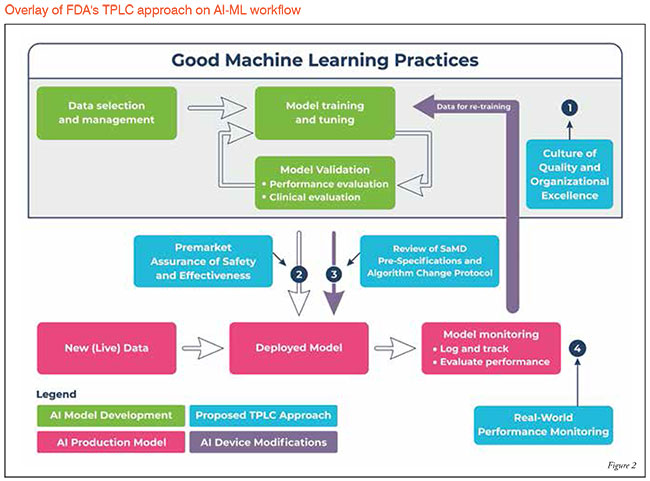
To fully realise the power of AI/ML learning algorithms, while enabling continuous improvement of their performance and limiting degradations, the FDA’s proposed TPLC approach is based on the following general principles
that balance the benefits and risks, and provide access to safe and effective AI/ML-based SaMD:
- Establish clear expectations on quality systems and Good ML Practices (GMLP)
- Conduct a pre-market review for those SaMD
- Expect manufacturers to monitor the AI/ML device and incorporate a risk management approach in development, validation, and execution of the algorithm changes (SaMD Pre-Specifications and Algorithm Change Protocol)
- Enable increased transparency to users and the FDA using post-market real-world performance reporting for maintaining continued assurance of both safety and effectiveness.
European Commission on Artificial Intelligence: EU has adopted a three-step Approach as described below.
1. Setting out requirements for achieving trustworthy AI
- Human Agency and Oversight: AI systems should enable equitable societies by supporting fundamental rights, and not decrease, limit or misguide autonomy
- Robustness and Safety: AI should be secure, reliable, and robust enough to deal with errors or inconsistencies during all life cycle phases of AI systems
- Privacy and Data governance: Citizens should have full control of their own data. Data should not be used to harm or discriminate against them.
- Transparency
- Diversity, non-discrimination, and fairness
- Societal and environmental well-being: AI systems should be used to enhance positive social change, enhance sustainability, and ecological responsibility
- Accountability for AI Systems and their Outcomes: Mechanisms should be put in place to ensure responsibility and accountability for AI systems and their outcomes
2. European AI Alliance
- Large-scale pilot with partners involving a wide range of stakeholders for feedback
- Members of AI high-level expert group will help present and explain the guidelines to relevant stakeholders in Member states
3. Building international consensus for human-centric AI
- Play an active role in international discussions and initiatives
Addressing the AI Black Box Issue:
- Can users understand the root cause of the negative outcome in AI solutions?
- Can users identify the training data or ML paradigm that led to AI application’s specific action?
- Incorrect Training Data can lead to misdiagnosis and/or an incorrect treatment recommendation. As a result, Clinical adoption is slow and there is a lack of trust from Patients.
- At this stage, AI can help provide data for better decision making (AI will not be fully replacing decision making processes). AI software developers will have to demonstrate to Clinicians that, with this tool, they can do a better job.
Formalised AI Use cases by American College of Radiology (ACR) Data Science Institute (DSI)
- To increase utilisation of AI adoption in Medical Imaging, the American College of Radiology (ACR) Data Science Institute (DSI) started releasing formalised use cases for how AI tools can be reliably used.
- Use cases empower AI developers to produce algorithms that are clinically relevant, ethical, and effective.
- The use cases are designed to guarantee that algorithms are applied to address clinical questions and allow for quality assessment measures that demonstrate compliance with legal, regulatory, and ethical measures.
- At present, ACR DSI has use cases for breast imaging, cardiology, musculoskeletal, neurology, oncology, paediatrics, nd thoracic.
Approved AI solution in Pharma: A Case Study
IDx-DR is an AI diagnostic system that autonomously analyses images of the retina for signs of diabetic retinopathy.
This is the first ever autonomous AI system cleared by FDA (announced on 11 April 2018) that provides screening/ diagnostic decision without the need for a clinician to interpret the image or results.
This device is a software program that uses an AI Algorithm to analyse images of the eye taken with a retinal camera called a Topcon NW400. Once the Doctor uploads the digital images of the patient’s retina to a cloud server on which IDx-DR software is installed, the software provides doctors with one of two results:
- More than mild diabetic retinopathy detected; refer to eye care professionals
- Negative for more than mild diabetic retinopathy; rescreen in 12 months
Conclusion:
With ongoing Global health concerns and the current COVID-19 pandemic situation, we need to identify and encourage forward-looking ideas, facilitate innovations, and bring them to life in order to cultivate ideas into potential solutions for our customers and, most importantly, save the lives of patients in need.
We must ensure both Patient Safety and Data Integrity when dealing with Technology.
Working professionals need to understand the changing landscape, Data sources, and Metadata, to ensure Data Quality during its processing and be able to upskill themselves to manage new technologies and tasks.
Global Regulatory bodies are actively encouraging the implementation of technology for faster drug development and are introducing Key Guidance documents and Initiatives.
Sponsors and vendors need to think innovatively in terms of their Study designs and executions plan and incorporate technology driven elements. Only then can we develop innovations beyond the current boundaries.












