Drug Discovery in Academia
An evolving model
Edward Holson, Medicinal Chemist III, Stanley Center for Psychiatric Research, The Broad Institute of MIT and Harvard, USA
As economic pressures mount on the current industrial model of drug discovery and development, there is growing momentum in academic institutions to enter the fray of drug discovery and development. There has always been a symbiotic relationship between academia and industry in the discovery and elucidation of new biological targets; however, the delineation of the roles of academia and industry are beginning to blur.
Over the past decade there has been increasing pressure on the pharmaceutical industry to increase economic efficiencies in drug discovery. This has been driven by several factors including the increased costs of R&D, lower productivity, patent expiration, generic competition and the increasing difficulty in bringing new chemical entities to market.
The extraordinary investment requirements have also led the industry to focus on only those disease areas which have high commercial potential and are deemed prosecutable. Many feel that this has led to a risk-averse behaviour which has hindered innovation in the field and created a focus on what are considered “druggable” targets. This has created a narrow view of biological space and has left major “third world” diseases orphaned.
While there has always been synergy between industry and academia in drug discovery, there is growing momentum within the ranks of academic and non-profit organisations to fill this innovation and therapeutic development gap through adoption of more conventional drug discovery and development approaches. Incorporation of disciplines and infrastructures that were the domain of industry is at the leading edge of this new paradigm in academia.
The Stanley Center for Psychiatric Research within the Broad Institute of MIT and Harvard is providing a forum to explore such a model. The Stanley Center was founded to discover the human genes that confer risk for bipolar disorder and schizophrenia and to use this information to develop new diagnostic tests and treatments for these illnesses. Consistent with this mission and key to this emerging ‘academic drug discovery’ model are industry veterans, who are making a transition from industry back into this new academic environment.
Integrated role of medicinal chemistry
The Stanley Center medicinal chemistry group is a small focussed group of four computational and medicinal chemists with both Big Pharma and biotech experience. The primary goal of this group was to implement a fully functional medicinal chemistry group mirrored on the capabilities of industry with limited permanent staff to allow for fiscal and staffing flexibility as projects evolved. Rather than following a fully integrated model supported by full-time staff, we adopted a hybrid virtual biotech model wherein we formed a core competency internally and outsourced the majority of analogue and bulk chemical synthesis.

The medicinal chemistry group functions to collaborate with multiple academic groups at various stages of the discovery process, including high throughput screening, probe compound development, lead optimisation, formulation, PK / ADME and in vivo efficacy experiments.
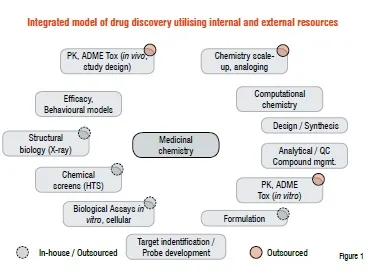
Shown in Figure 1 is a ‘chemo-centric’ view of the various disciplines integrated within the Stanley Center. The medicinal chemistry group focuses on hypothesis-driven compound design, synthesis, data analysis and the development of new approaches to drug discovery. Resources are augmented through the use of several Contract Research Organisations (CROs) for analogue and bulk chemical synthesis. Academic collaborators constitute core biology and in vivo efficacy (behavioural models). External vendors are used for more traditional drug development disciplines such as PK / ADME for both in vitro and in vivo studies.
The majority of our biological studies including in vitro binding, enzyme and cellular assays are done in collaboration with academic groups at local institutions (MIT, Harvard and Massachusetts General Hospital). The majority of our target identification, high throughput screening and compound management activities are conducted in collaboration with established core facilities at the Broad Institute. The goal was to develop an infrastructure which would allow a group of two internal medicinal chemists and four to six external chemists to efficiently process 200 to 300 compounds per year across multiple projects. By careful selection of CROs, we hoped that over time we would have a push back of ideas from our contractors and active participation in design and data evaluation. Ultimately, we hoped to establish our external contractors in chemistry as an extension of our team and leverage their capabilities as deep into our process as possible making them indistinguishable from internal resources.
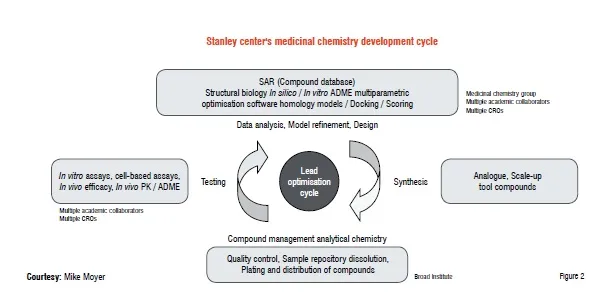
Medicinal chemistry development cycle
Our approach is similar to an industrial template in the overall flow but differs in the organisational arrangements of the different disciplines. Our design and development cycle is shown in Figure 2 and mirrors the cycle found in an industrial setting.
Key functions include:
1. Compound design and synthesis
2. Compound management—notebooks, QA / QC, tracking of compounds, information capture from CROs, registration, plating and distribution of compounds
3. Compound testing (in vitro and in vivo)
4. Data analysis and storage
5. Repeat.
While our cycle mirrors industry, the various stages of the development cycle are performed outside the traditional organisational format. Unlike a typical company, where project team members specialising in various disciplines are unified as part of a single organisation, in an academic setting disparate disciplines from distinct organisations (universities, principal investigators, etc.) are involved.
A model that lacks a clear organisational structure is highly reliant on communication and collaboration across groups. From a medicinal chemistry viewpoint, a key goal was to establish a highly efficient chemistry group by identifying “best-in-class” contract research organisations, leveraging the capabilities of the Broad Institute and establishing an efficient process to manage hundreds of compounds through the development cycle.
Lessons learnt
We began our chemistry programme by examining several different chemistry CROs. We chose to compare vendors by assigning similar chemistries to assess not only their core chemistry competency but also their communication skills, problem solving, creativity and informatics capabilities. Working on an FTE basis for a period of three months with two to four FTEs per site, we began a rolling evaluation across several companies. We conducted weekly or biweekly teleconferences along with site visits to get hands-on experience with the chemists as well as evaluate their resources and capabilities. We found a wide variation from vendor to vendor across all areas. Moreover, we found that small inefficiencies in the process (communication, problem solving, and record keeping) were amplified in our small group and created large time drains internally.
To streamline the flow of information and to centralise the capture of chemical information from all vendors, we adopted an electronic notebook (e-notebook) system established by the Chemical Biology Platform at the Broad. Originally, we transcribed varying report formats and data into the Stanley Center’s e-notebooks. We quickly found that this administrative task required a prohibitive amount of time. Ultimately, synchronisation with our vendor’s records by providing remote login (to the Broad’s e-notebook system) allowed each vendor to download experimental details and analytical data in real time. This provided several advantages:
1. A permanent record of experiment details with associated analytical data on an internal server
2. A searchable chemistry database within the Stanley Center and Broad Institute
3. A source for real-time evaluation of ongoing chemistries
4. A cornerstone for leveraging existing capabilities of the Broad Institute (compound registration, analytical (QA / QC), compound management)
5. An expansion of CROs’ capabilities and responsibilities
By integrating our compound flow into the existing informatics structure of the Broad Institute via e-notebook we were able to adopt already established protocols. Compound registration (Broad ID) assigns a unique alpha-numeric code for each compound which contains purity, batch and salt information. In addition, compound registration provides access to the Broad’s analytical capabilities. Once registered, integration of an open-source web-based image viewer (“aView”) allowed rapid annotation of analytical results and streamlined the process of compound purity assessment and re-analysis and / or re-synthesis. Moreover, this allowed us to work with the Broad’s compound management group to plate compounds efficiently for in vitro and cell-based assays.

The E-notebook system facilitated complete coordination among our chemistry contract partners and chemists working internally, thereby fully integrating them with our informatics environment. Contract chemists synthesise, document and register each compound remotely. Compounds are received pre-registered in bar-coded vials and forwarded to compound management for dissolution, preparation of stock solutions, and plating for biological assays as well as LC / MS analysis. Review and annotation of analytical data via aView prior to assaying provides a filter to remove compounds from the system that do not meet our purity requirements. What had taken hours (document, register, and collate data from various vendor sources) for each compound has now been reduced to minutes by extending our process into the hands of our chemistry CROs.
Through careful evaluation and utilisation of external CROs and by integrating into existing infrastructures at the Broad Institute, an efficient process for the first steps of the drug discovery cycle has been established. We hope with time to extend these efficiencies deeper into the drug discovery process and continue to refine this model.
Future
With the implementation of an efficient chemistry infrastructure based on an industrial model we are poised to engage and refine more of the discovery cycle in this new environment. What remains to be seen is whether this model is sustainable and how it will impact industry and academia.
Implementing a drug discovery programme creates a large burden on resources. Thus, sustaining drug discovery and development in academia will require both a shift in funding initiatives to foster this type of research as well as successful partnering between academic institutions and industry. The landscape does indeed appear to be shifting in this direction. Government funding agencies have initiated programmes geared specifically toward IND-enabling studies (NIH’s National Cooperative Drug Discovery and Development Groups Program) and towards funding for drug development activities (NIH-RAID, Rapid Access to Interventional Development). Charitable organisations and philanthropic foundations are also having an enormous impact on funding this research (The Broad Institute and the Stanley Center are just two examples). In addition, private resources are funding the earliest stages of discovery in psychiatric disease through a special outreach initiative (‘PsychHTS’) for primary high-throughput screens. Finally, academic centres focussed on specific disease areas have had major upfront investments from industry which should allow sufficient opportunity to test this new model (e.g. Vanderbilt and Johnson & Johnson). The next 5 to 10 years will define how much this new model will impact our traditional models of drug discovery and development.










