SARS-CoV-2 is highly transmissible and infectious coronavirus which is spreading across the globe at an alarming rate. As of 18 May 2021, there are 163,642,990 confirmed cases and 3,390,316 deaths reported from COVID-19. Here, we review the current knowledge regarding the immunopathogenesis and potential therapies for management of COVID-19.
In December 2019, a pneumonia outbreak occurred in Wuhan city of China that quickly spread across the world and posed serious public health emergency. On 9 January, 2020 novel coronavirus severe respiratory disease syndrome coronavirus-2 (SARS-CoV-2) was identified as the cause of outbreak in Wuhan. Later on 11 February 2020, the World Health Organization (WHO) named the disease caused by SARS-CoV-2 as coronavirus disease-2019 (COVID-19). Coronaviruses infect humans and animals and are associated with various respiratory, gastrointestinal, and neurological disorders. SARS-CoV-2 is the seventh coronavirus that is recognised to cause infection in humans. The other six coronaviruses are known to cause only mild symptoms. However, there are two notable exceptions: SARS-CoV and Middle East respiratory syndrome coronavirus (MERS-CoV). SARS-CoV is responsible for an epidemic that started from China in the year 2002-2003. This outbreak resulted in 8000 infections and 774 fatalities across 37 countries. MERS-CoV was first detected in 2012 in Saudi Arabia and caused 2,494 confirmed cases and 858 fatalities. SARS-CoV-2 shows 79.6 per cent sequence identity with the SARSCoV. However, bat coronavirus RATGI3 seems to be its closest relative, showing over 96 per cent sequence similarity. SARS-CoV-2 is highly contagious and causing more infections and deaths than both SARS-CoV and MERS-CoV. As a result, WHO declared COVID-19 a pandemic on 11 March, 2020.
Pathogenesis of COVID-19
SARS-CoV-2 is respiratory virus and mainly transmits through droplets produced when an infected person sneezes or coughs. However, recently US Centers for Disease Control and Prevention acknowledged COVID-19 as airborne disease which means that it can be transmitted by respiratory fluids also. Respiratory fluids also known as aerosols are fine droplets that are produced during respiration. Therefore, a person can be infected on inhalation of aerosols formed when a COVID-19 patient exhales, speaks, sings, shouts, coughs and sneezes. SARS-CoV-2 can cause mild infection to severe respiratory failure in infected patients. Typical symptoms associated with SARS-CoV-2 infection are fever, dry cough, fatigue, weakness, chest tightness, loss of smell and taste, pain and aches, sore throat, diarrhoea, chills, rashes, headache, conjunctivitis and in severe cases dyspnea and Acute Respiratory Distress Syndrome (ARDS). ARDS is chronic respiratory disorder characterised by difficulty in breathing, hypoxia, and pulmonary edema. SARSCoV-2 infection in children and young adults is mostly asymptomatic. But the older population (> 60 years) particularly those having the comorbidities (hypertension, diabetes, cardiovascular diseases) are at high risk of developing the severe disease. Sex related differences also determine the clinical outcome of COVID-19 as men are more prone to infection than women and also have a high mortality rate. In severe cases, COVID-19 also results in multiple organ failure which is characterised by acute liver failure, Cardiovascular Diseases (CD), neurological disorders, Acute Kidney Injury (AKI) and several hematological abnormalities. Once inside the body the first step involved in the pathogenesis of COVID-19 is the binding of the SARS-CoV-2 to host receptor Angiotensin Converting Enzyme 2 (ACE2) that is expressed on host cells viz. lung cells. The binding of SAR-CoV-2 to ACE2 receptor allows the entry of viral genetic material into host cells followed by their replication and multiplication. The newly formed viral particles then exit the infected cell and start invading other adjacent epithelial cells resulting in lung damage. Apart from lung, ACE2 receptor is also expressed by almost all tissues e.g. heart, intestine, kidney, bladder etc..
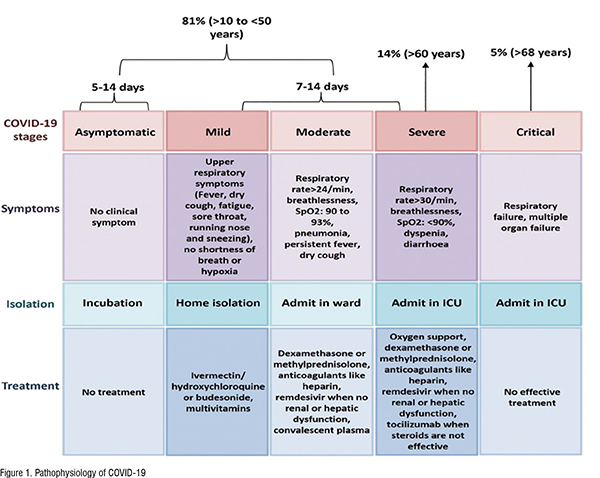
Immunopathogenesis of COVID-19
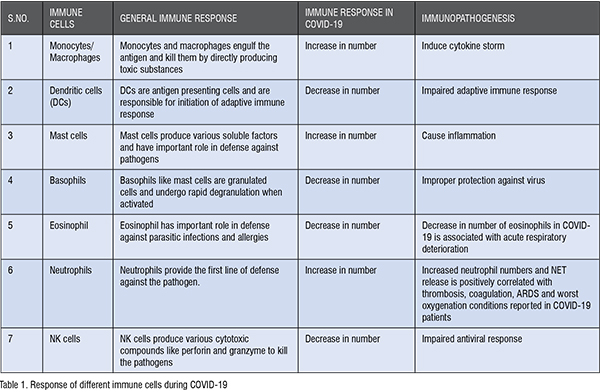
Viral infections activate our immune system. Immune system which constitutes of special organs, cells and chemicals protects our body from invaders like viruses, bacteria and other foreign bodies. Immune system is mainly characterised under two categories: Innate and adaptive immune system. Innate immune system is the first line of defense and is non-specific to pathogens. On the other hand, adaptive immune system is the second line of defense and is pathogen specific. Well-coordinated innate and adaptive immune responses generally protects against various types of bacterial and viral infections. However, in case of SARS-CoV-2 immune response gets hampered that results in immune pathology. Immune system is activated when immune cells recognised certain molecular patterns associated with the pathogen. Activated immune cells then secretes various soluble molecules called the cytokines and chemokines. These cytokines/chemokines have role in recruitment of various innate immune cells such as monocytes/macrophages and neutrophils at the site of infection. These immune cells further produce cytotoxic substances to eliminate viral infection. Although these substances protect from viral infection but can also cause damage to the host tissues. In case of COVID-19 there is exaggerated production of cytokines (cytokine storm) resulting in uncontrolled influx of monocytes and neutrophils in the lung tissue. The cells notably the neutrophils and macrophages then produce toxic substances which cause acute lung injury and ARDS resulting in respiratory failure. COVID-19 also impaires the production of interferons which are chemicals secreted by host cells in response to viral infection. After pathogen recognition, host cells produce interferons that have important role in eliminating the virus but SARS-CoV-2 through several mechanisms inhibits the generation of interferons and thus escape the host immune response. SARS-CoV-2 infection also modulates the activity of various immune cells (Table 1).
Potential Therapies for COVID-19
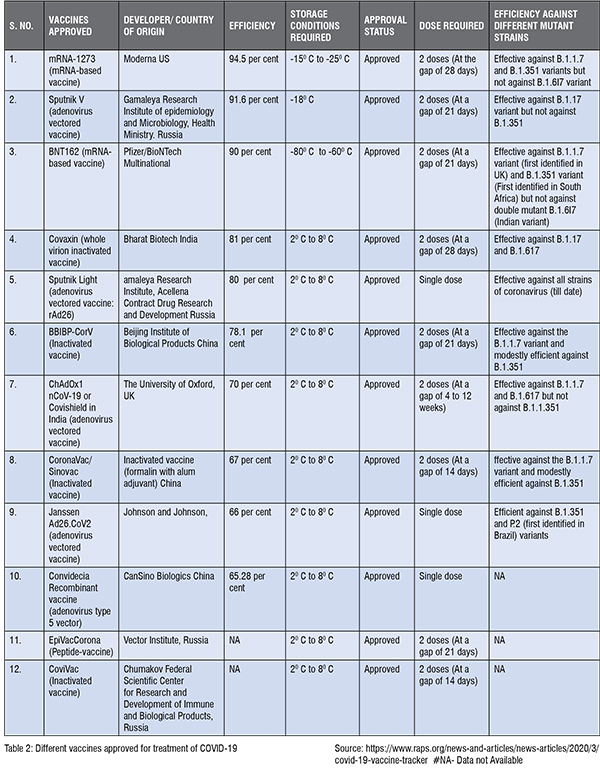
Till now there is no effective therapy available for the prevention of SARSCoV- 2 infection. Nevertheless, several drugs such as antivirals, immune modulators and antibiotics have been repurposed for combating COVID-19. Currently, human clinical trials on 92 vaccines and 419 therapeutic drugs are going on for management of COVID-19. Drugs like chloroquine, hydroxychloroquine, remdesivir, tocilizumab, etc. received intense attention worldwide as they showed positive results in preliminary studies. Remdesivir was the first drug authorised by the Food and Drug Administration (FDA) for the treatment of severe COVID-19. Remdesivir perturbs viral RNA replication. Data from the placebo-controlled trials showed the clinical effectiveness of remdesivir in shortening the recovery time of hospitalised COVID-19 patients who require oxygen support. Although there are clear indications of its efficacy against COVID-19 but no study till now has evidenced the role of remdesivir in mitigating the mortality rate. But due to lack of other potential treatment options remdesivir is in extensive use in hospitals for patients (with comorbidities) having severe COVID-19 disease. Chloroquine and hydroxychloroquine are used for the treatment of malaria and now found to be effective in the management of COVID-19 in some clinical trials. Chloroquine / hydroxychloroquine prevents the entry and exit of SARS-CoV-2 virus from the cell and have immunosuppressive properties. Chloroquine and hydroxychloroquine had shown the promising results in preventing COVID-19 in various clinical settings during first wave. But current data from several randomised controlled trials evidenced no clinical benefit of these drugs in treatment of COVID-19 during second wave. Azithromycin is an antibiotic having potential antiviral and anti-inflammatory properties also. A small non-randomised trial aimed to evaluate the effectiveness of azithromycin has revealed that azithromycin in combination with hydroxychloroquine has significant antiviral activity against SARS-CoV-2. But in contrast to this, a recent study demonstrated that there is no meaningful benefit of azithromycin in reducing recovery time and risk of hospitalisation. Another solidarity trial tested the combined effects of two antiviral drugs lopinavir and ritonavir. These drugs were initially developed against Human Immunodeficiency Virus (HIV). Lopinavir and ritonavir were also used against the SARS infection and it was observed that treatment with these drugs ameliorated ARDS at day 21. Given their effectiveness against SARS-CoV they were tested for SARS-CoV-2. However, the clinical trials showed no association between these drugs and decrease in mortality, duration of hospital stay and the need of mechanical ventilation. Therefore, clinical trials do not support the use of these drugs for hospitalised COVID-19 patients. Favipiravir which is originally designed against influenza is one of antiviral drugs that has been repurposed to halt the progression of COVID-19. Favipiravir is orally administered and appears useful in the treatment of mild to moderate SARS-CoV-2 infections. As 85 per cent of the COVID-19 cases are mild or moderate, favipiravir is considered as a potential therapy for large number of the patients. Now, a randomised controlled trial showed that favipiravir reduced the need of mechanical ventilation and duration of hospitalisation and therefore can be also used for hospitalised patients. Thus, favipiravir can be a promising therapy in decreasing the stress on global healthcare system due to COVID-19. Ivermectin is FDA approved anti-parasitic drug that has broad spectrum antiviral activity also. It is observed that ivermectin can inhibit SARS-CoV-2 replication. Various small-scattered studies back the efficacy of ivermectin in management of COVID-19. As the use of hydroxychloroquine has largely debunked for COVID-19 prevention, ivermectin is becoming wildly popular as a preventive measure for COVID-19. However, WHO warned against the use of ivermectin due to lack of convincing evidence that merits the ability of ivermectin in controlling SARS-CoV-2 infection. The interest in immunosuppressant such as corticosteroids is also refreshed for cure of COVID-19 as in the past they were widely accepted for preventing SARSCoV outbreak. However, in COVID-19 patients corticosteroids treatment resulted in poor outcomes. But later a widely publicised press release and subsequent results from RECOVERY trial noted the benefit of dexamethasone in preventing severe cases. The RECOVERY trial concluded that dexamethasone therapy reduced the death rate by one third in severe COVID-19 patients who are on respiratory support but is inefficient in preventing non-severe cases. Other corticosteroids like budesonide and methylprednisolone are also observed effective against COVID-19. Use of tocilizumab, an immunosuppressive drug mainly recommended for rheumatoid arthritis is also reinforced for COVID-19. Despite the fact that there is widespread use of tocilizumab as off-label treatment the efficacy and safety of tocilizumab in the management of COVID-19 has yet not proven. Convalescent plasma therapy in which antibodies are given to the infected person also give mixed results. Till now, it is observed that most safe, simple, and effective method for protection against coronavirus are vaccines which can control this pandemic. Several vaccines are approved for COVID-19 which are summarised in Table 2. But as there are not enough vaccines doses currently available for the entire world, we have to depend upon supportive treatment strategies till the time we vaccinate all our populations.