Precision Medicine
Redefining genomic breakthroughs and 3D printing in pharmaceutical R&D
Rahul Shukla, Department of Pharmaceutics, National Institute of Pharmaceutical Education and Research (NIPER)
Precision medicine is revolutionising research and development (R&D) using genomic tools and 3D printing. Genomic understanding allows creation of targeted therapies, while 3D printing enables tailoring of therapeutic delivery systems and stipulated production. Such newer techniques are harbour safe, efficacious, and personalised regimens, reconstructing the modern healthcare perspective.

Precision medicine (PM) is aimed at significantly reforming the pharmaceutical industry through advancements in pharmacogenomics, polygenic risk assessment, and precision molecular diagnostics. To establish PM's effectiveness within the pharmaceutical product development, profound research is required to demonstrate that PM can enhance therapeutic outcomes across diverse patient populations without exacerbating health disparities. Moreover, PM interventions should provide therapeutic benefits over contemporary treatments. A crucial aspect of PM is gaining innovative therapeutics in economics that entails PM modalities are cost-effective and possess potential to have developmental impact. The success of PM also hinges on societal acceptance, which necessitates addressing concerns regarding data governance and maintaining reliability in AI-driven treatment customization. Securing and upholding the trust of healthcare providers, patients, and regulatory bodies is essential for PM's success. The advancement of PM is prominently fortified by multi- omics technologies, such as genomics and pharmacogenomics, and the utilisation of big data analytics, particularly through AI. For PM to be effectively integrated into clinical practice, it is crucial to amalgamate it with electronic health records (EHRs). As the evidence supporting PM grows, there is an urgent need for real-world data to verify its clinical utility, cost-effectiveness, and impact on health equity.
3D printing is increasingly contributing to personalised medicine and development of customised therapeutic delivery systems. This technology enables tailoring of treatments based on individual patient characteristics, including anatomy, disease state, and genetic profile. Various 3D printing techniques offer distinct advantages based on their applications, materials, and resolution requirements. For instance, fused deposition modeling (FDM) uses thermoplastic filaments, making it versatile for fabricating drug-loaded matrices and scaffolds in tissue engineering. Stereolithography (SLA) involves solidifying liquid resin with light exposure, achieving highresolution prints suitable for intricate drug delivery systems and microfluidic devices. Selective laser sintering (SLS) uses lasers to fuse powdered materials, allowing precise control over structure porosity for drug-loaded implants and scaffolds. Inkjet-based 3D printing deposits bioinks with cells or therapeutic agents onto substrates, enabling the creation of complex tissue constructs and drug-loaded microcapsules. In tissue engineering and regenerative medicine, 3D printing is crucial for creating complex tissue structures. Bioprinting techniques, which layer bioinks to incorporate cells and supporting materials, have successfully produced functional tissues like skin, cartilage, and blood vessels. These bioprinted tissues are valuable for drug testing, disease modelling, and potential patient-specific tissue replacements. Furthermore, 3D printing has revolutionised prosthetics and orthotics by enabling the rapid production of customised devices with enhanced fit, functionality, and aesthetics. Unlike traditional, labour-intensive methods, 3D printing allows for quick prototyping and personalization of devices.
Challenges associated with precision medicine and 3D printing
Despite its potential, 3D bioprinting faces challenges, particularly in replicating the functionality of complex secretory organs. While it allows precise customisation of anatomical structures, creating functional versions of organs such as the liver and pancreas remains difficult. Advances in technology, materials, and cellular biology are needed to achieve both structural and functional accuracy. Another challenge is designing 3D bioprinted organs for paediatric patients, whose bodies continuously grow. Bioprinted organs must adapt and grow with the child, which may require periodic replacements. This adds complexity to the design and maintenance of bioprinted tissues. Recent advancements in 3D printing are significantly transforming precision medicine, offering innovative ways to tailor treatments to individual patient needs. This technology is revolutionising how we approach drug delivery, tissue engineering, and diagnostics, leading to more personalised healthcare solutions.
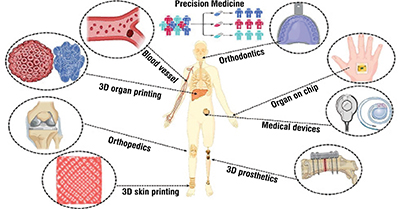
In the realm of drug delivery, 3D printing allows for the creation of customised medication systems that cater to individual patient profiles. Unlike traditional methods, which often rely on standardised treatments, 3D printing enables the production of drug formulations designed to meet specific therapeutic requirements. Techniques such as Fused Deposition Modeling (FDM) and Stereolithography (SLA) are particularly noteworthy. For example, FDM can be used to manufacture tablets with controlled release profiles, ensuring that drugs are delivered at precise rates and locations within the body. This is particularly useful for treatments that need to bypass stomach acid, such as those for ulcerative colitis. Additionally, SLA has been employed to produce polymeric microneedle patches that deliver insulin transdermally. These patches are designed with great accuracy to ensure effective insulin administration, improving diabetes management. In tissue engineering, 3D printing has enabled the fabrication of complex tissue structures that closely mimic human tissues. Techniques like bioprinting involve layering bioinks composed of cells and other materials to create functional tissues, including skin, cartilage, and blood vessels. Recent developments include the creation of bilayered scaffolds using GelMA hydrogels. These scaffolds have an upper layer that delivers growth factors such as TGF-beta-1 to promote cartilage repair, while the lower layer contains hydroxyapatite to support bone regeneration. These advancements enhance the ability to repair and regenerate damaged tissues, offering new treatment options for various conditions. The fusion of 3D printing with biopharmaceuticals has also led to the development of sophisticated drug delivery systems. For instance, scaffolds made from polymers like PLA and coated with peptides derived from bone morphogenetic proteins (BMP-2) have shown promise in enhancing bone growth. Similarly, multiunit implants composed of polycaprolactone and melanin have been designed to release insulin in response to light. These implants use light to control the release of insulin, providing a novel approach to diabetes treatment. 3D printing has also improved diagnostic capabilities by enabling the creation of detailed, patient-specific anatomical models. These models offer a more accurate representation of a patient’s internal structures, aiding in surgical planning and reducing the risk of complications during procedures. By providing a clearer view of a patient's anatomy, 3D-printed models help surgeons prepare more effectively and perform surgeries with greater precision. Despite these obstacles, 3D bioprinting holds significant promise. It has shown remarkable progress in developing customised drug delivery systems and tissue scaffolds tailored to individual patient profiles. Examples include drug-loaded matrices and scaffolds designed for controlled release and enhanced efficacy. In tissue engineering, bioprinted models offer a more accurate assessment of pharmaceutical effects, reducing the need for animal testing and leading to more ethical drug development.
Regulatory consideration in precision medicine and 3D bioprinting
Regulatory oversight plays a critical role in the integration of precision medicine and 3D bioprinting within the pharmaceutical sector due to their advanced and intricate nature. In the realm of precision medicine, regulatory bodies need to assess the tools and methods employed to customise treatments based on individual patient profiles. This involves verifying the accuracy and reliability of the algorithms that process personal and genetic data, ensuring that they contribute to improved patient outcomes. Additionally, there are stringent privacy regulations to protect sensitive genetic information from unauthorised access and misuse. Clinical trials are essential to validate these personalised approaches, requiring clear protocols for data handling and patient consent to ensure ethical practices. For 3D bioprinting, the regulatory landscape is equally complex. Ensuring that the materials used, such as bioinks and scaffolds, are safe and compatible with human tissues is paramount. Regulatory authorities must oversee that the manufacturing processes for bioprinted tissues and organs are standardised and meet rigorous quality control measures. Comprehensive testing is necessary to confirm that bioprinted constructs function correctly in clinical applications, with long-term follow-up to monitor their performance and durability. Ethical issues also arise in 3D bioprinting, particularly regarding the sources of biological materials and the implications of creating advanced tissue structures. Regulations must address these ethical concerns, ensuring that bioprinting practices are conducted with respect for the origins of biological materials and that they do not lead to controversial uses. The challenge for regulators is to foster innovation while maintaining patient safety. As technology evolves, regulatory frameworks must adapt to address new developments and ensure that both precision medicine and 3D bioprinting technologies are safe, effective, and ethical.
Conclusion
Looking ahead, the future of 3D bioprinting in pharmaceuticals is bright. Continued innovation is needed to address current challenges, particularly in functional replication and growth adaptation. As technology evolves, it is expected to revolutionise drug delivery, tissue engineering, and regenerative medicine, offering new opportunities to advance patient care and therapeutic outcomes. Despite existing challenges, the transformative potential of 3D bioprinting in medical treatments remains substantial and promising. In summary, 3D bioprinting faces challenges due to the scarcity of advanced biomaterials that are both biocompatible and suitable for printing, research has shown that stimuliresponsive materials can create functional constructs, there remains a significant gap in developing materials that support cell health and provide necessary mechanical strength. Current smart constructs made with 3D printing are primarily used for tissue regeneration, drug delivery, and monitoring. Innovations, such as vascular stents responsive to heat or magnetic fields, have enhanced deployment accuracy but need additional capabilities like controlled drug release and degradation features to address full clinical requirements. Scaling these technologies from lab settings to large-scale production introduces obstacles, including the need for consistent manufacturing practices and thorough quality assurance. Since many of these constructs are still in early-stage development, further research and extensive testing are crucial before they can be approved for clinical use.
References:
[1] Q. Gao, J.-S. Lee, B. Soo Kim, and G. Gao, “Three-dimensional printing of smart constructs using stimuli-responsive biomaterials: A future direction of precision medicine,” International Journal of Bioprinting, vol. 9, no. 1, Nov. 2022, doi: https://doi.org/10.18063/ijb.v9i1.638
[2] M. E. Prendergast and J. A. Burdick, “Recent Advances in Enabling Technologies in 3D Printing for Precision Medicine,” Advanced Materials, vol. 32, no. 13, p. 1902516, Sep. 2019, doi: https://doi.org/10.1002/adma.201902516
[3] L. Alzoubi, Alaa A. A. Aljabali, and M. M. Tambuwala, “Empowering Precision Medicine: The Impact of 3D Printing on Personalized Therapeutic,” AAPS PharmSciTech, vol. 24, no. 8, Nov. 2023, doi: https://doi.org/10.1208/s12249-023-02682-w
[4] W. Sun, J. Lee, S. Zhang, C. Benyshek, M. R. Dokmeci, and A. Khademhosseini, “Engineering Precision Medicine,” Advanced Science, vol. 6, no. 1, p. 1801039, Oct. 2018, doi: https://doi.org/10.1002/advs.201801039
[5] W. Sun, J. Lee, S. Zhang, C. Benyshek, M. R. Dokmeci, and A. Khademhosseini, “Engineering Precision Medicine,” Advanced Science, vol. 6, no. 1, p. 1801039, Oct. 2018, doi: https://doi.org/10.1002/advs.201801039
[6] H. Bhuskute, P. Shende, and B. Prabhakar, “3D Printed Personalized Medicine for Cancer: Applications for Betterment of Diagnosis, Prognosis and Treatment,” AAPS PharmSciTech, vol. 23, no. 1, Dec. 2021, doi: https://doi.org/10.1208/s12249-021-02153-0













