The Significance of microRNAs in Cancer
A brief overview
Ancuta Jurj, Department of Translational Molecular Pathology, The University of Texas MD Anderson Cancer Center
George Adrian Calin, Center for RNA Interference and Non-Coding RNAs, The University of Texas MD Anderson Cancer Center
In recent decades, microRNAs (miRNAs) have become pivotal in cancer therapeutic strategies, serving as compelling tools and biomarkers. With dual roles as oncogenes or tumour-suppressors, miRNAs play key functions in cancer biology. Several miRNA-targeted therapeutics gaining attention have advanced to clinical development. This surge is reflected in increased miRNA integration across oncology trials, screening, diagnostics, and drug testing. Consequently, this mini-review aims to provide a succinct overview of the evolving landscape of miRNA applications in oncology research, shedding light on their potential as a therapeutic modality.

Decades ago, microRNAs (miRNAs) were first identified in Caenorhabditis elegans (C. elegans) by Ambros and Ruvkun’s groups in 1993 and subsequently linked to cancer by Calin et al in 2002. This discovery sparked a surge in miRNA-related research, highlighting their pivotal role in tumourigenesis and leading to diverse applications, from biomarker development to potential RNA therapeutics. Structurally, miRNAs are small noncoding RNAs (ncRNAs), composed of single-stranded RNA molecules typically 19-24 nucleotides (nt) in length. Their biogenesis initiates in the nucleus, where primary miRNAs (pri-miRNAs) are transcribed from RNA transcripts and cleaved by RNA polymerase into precursor miRNAs (pre-miRNAs), which mature into single-stranded miRNAs approximately 22 nt long. Each step of miRNA biogenesis relies on essential components such as RNA polymerase II (Pol II) for transcription, RNase III enzymes (DROSHA and DICER1), and members of the Argonaut family (AGO2).
miRNAs play crucial roles in modulating various biological processes, exhibiting distinct spatial and temporal expression patterns. In addition to their conventional function in post-transcriptional repression of specific target proteins by promoting mRNA decay or inhibiting translation, miRNAs have been observed to influence transcriptional activation, epigenetic regulation, and upregulation of translation. In this mini-review, we aim to provide a concise overview of the role of miRNAs in cancer biology.
miRNA and cancer – a glimpse behind these two pioneers
As is widely recognized, cancer constitutes a genetic ailment affecting a growing number of individuals worldwide. It is characterised by a cascade of mutations occurring in tumour suppressor genes, facilitating uncontrolled cell proliferation, and impeding cell death. Recent findings have unveiled the role of miRNA dysregulation in bolstering cancer progression. Intriguingly, a single miRNA has the capacity to bind to numerous targets, potentially exceeding several hundreds, encompassing diverse functionalities such as transcription factors, receptors, and vectors. Ample evidence underscores the marked differences in miRNA expression between normal and tumour tissues, strongly implicating their involvement in tumour progression, development, invasion, and metastasis.
Do miRNAs play a dual role?
As mentioned previously, miRNAs are implicated in modulating all cancer hallmarks, functioning either as oncogenes or tumour suppressors. However, certain miRNAs exhibit context-dependent roles, acting as oncomiRs in some malignancies while serving as tumour suppressors in others. For instance, in certain cancers, members of the let-7 family function as tumour-suppressor miRNAs, impeding invasion, metastasis, epithelial-to-mesenchymal transition (EMT), and self-renewal. Conversely, when their expression profile is elevated in the tumour microenvironment, let-7 family miRNAs can adopt oncogenic roles. Another example is miR-146a-5p, which demonstrates a dual role as both an oncomiR and a tumour suppressor in several cancers. Similarly, miR-186 exhibits a dual function, serving as both a diagnostic and therapeutic target, and playing pivotal roles in oncogenesis, cell migration, invasion, cell death, metastasis, and drug resistance. In gastrointestinal cancers, miR-9 has been identified to play a dual role as well. These observations underscore the heterogeneity of cancer and the dual role of miRNAs in malignant pathophysiology. However, further studies are imperative to elucidate the specific roles of miRNAs and the underlying mechanisms governing their regulation.
microRNAs and cancer as a full body disease
There are studies supporting the hypothesis that miRNAs can directly modulate genes encoding hormones or enzymes responsible for hormone maturation and metabolism. Furthermore, miRNAs indirectly modulate hormone-mediated cell signaling transmission by targeting hormone antagonists or receptors.
Recent evidence suggests an important network of interactions between miRNAs and ERα (estrogen receptor-α), coordinating cellular responses to estrogen. Certain miRNAs modulated by ERα, such as (pri-) miR-17-92 and miR-206a-363, have been identified as targets that downregulate ERα expression at the protein translational level. Another study highlights the significance of miR-149-5p, which inhibits the regulatory activity of the transcription factor SP1 under 17β -estradiol therapy, while its decreased profile promotes the expression of hnRNPA1, regulating the loading of let-7 miRs into EVs. Regarding androgen receptors (AR), miR-let-7c indirectly suppresses receptor activity by targeting c-Myc. Direct associations between miR-185 and miR-205 with AR expression have been described. In prostate cancer, AR regulates the transcription of miR-21, enhancing tumour cell growth, while a confirmed connection exists between miR-185-5p and AR in clear cell renal cell carcinoma. miR-21 and miR-206 show increased expression profiles in hormone-dependent cancers.
miRNAs also play a crucial role in regulating stress responses. Hyperthermia, among the stress-induced factors, suppresses the expression of miR-23a, leading to elevated levels of the pro-apoptotic protein NOXA and subsequent apoptosis. Furthermore, a downregulated profile of miR-23a is associated with CDK5 inhibition, resulting in increased expression of HSP70 in stressed cells. In response to environmental stress, the tumour suppressor p53 modulates the expression of miRNAs at both transcriptional and processing levels. Consequently, an increased number of cancers harbouring p53 mutations exhibit specific alterations in miRNA processing and transcriptional activity. Loss of p53 function facilitates tumour progression by impeding stress response mechanisms. Interestingly, despite their increased expression, some of these miRs have been identified as oncomirs but paradoxically act as tumour suppressors, thereby inhibiting cancer progression.
Extracellular microRNAs as biomarkers for therapeutic clinical trials
Extracellular vesicles (EVs) are small bilipid entities secreted by various cell types and can be detected in body fluids such as blood, milk, urine, cerebrospinal fluid, and saliva, typically ranging in size from 50 to 200 nm. While EVs serve as a general term, they can be categorised into exosomes, microvesicles, and apoptotic bodies based on their distinct biogenesis pathways. Exosomes originate from inward budding of multivesicular bodies (MVBs), microvesicles originate from the outward budding of the plasma membrane, while apoptotic bodies result from cell membrane fragmentation during cell death. Recent attention has been focused on these vesicles, particularly exosomes, revealing that critical bioactive molecules such as nucleic acids (DNA, mRNA, miRNA), specific proteins, and lipids are packaged into EVs during biogenesis. These molecules are subsequently transported from donor cells to target cells, underscoring the importance of EVs as natural transporters facilitating cell-to-cell communication. In this context, EVs possess the potential to influence the phenotype of target cells, whether normal or tumour, and contribute to processes associated with cancer hallmarks. Numerous diseases are linked to dysregulated expression of miRs, and importantly, the presence of miRs in bodily fluids is associated with disease progression. Exosomal miRs, in particular, hold promise as biomarkers for various pathological conditions, offering insights into disease pathogenesis and potential mechanisms of repair. However, a critical question arises from the limited understanding of the miRNA content packaged into EVs, highlighting the need for further investigations.
Exosomal miRNAs show potential as biomarkers for early detection and prognosis across a spectrum of cancers. In non-small cell lung cancers, the miRNAs let-7b-5p, let-7e-5p, miR-23a-3p, and miR-486-5p have been identified for early diagnosis, while exosomal miR-451a shows promise as a prognostic marker for patients across stages I to III. Additionally, exosomal miR-4257 and miR-21 serve as recurrence-specific biomarkers in non-small cell lung cancers. In glioblastoma, the presence of exosomal miR-320 and miR-574-3p distinguishes patients from healthy individuals, with exosomal miR-301a correlating with pathological grades and recurrence. In prostate cancer, exosomal miR-182 and miR-183 are markedly expressed, while miR-1290 and miR-375 show promise for prognosis. In breast cancer, miR-223-3p is associated with detection of biomarkers and is linked to histological types, nuclear grade, pathological stages, and lymphatic invasion. Moreover, exosomal miR-373 holds potential as a biomarker in triple-negative breast cancer.
miRNAs have emerged as promising biomarkers for prognostic and diagnostic purposes across various types of cancers. Achieving a comprehensive understanding of miRNA-based diagnostic approaches entails delineating tumour subtypes with similar phenotypes and uncovering new malignant sub-entities. To enhance the specificity of diagnostic tools, it is imperative to elucidate the mechanisms governing miRNA circulation in bodily fluids, including their associations with proteins, lipids, and exosomes. Moreover, exploring chemical modifications within miRNA structures holds potential for identifying novel molecules serving as biomarkers. IsomiRs, which represent miRNA variants resulting from post-maturation editing events such as modifications at the 3’-end facilitated by nucleotide transferase and 3’-exonuclease processes, offer promising avenues for enhancing tumour type specificity. By leveraging the specificity of isomiRs, more refined distinctions between tumour types can be achieved, thereby advancing miRNA biomarker development. Nonetheless, further investigations are warranted to refine and optimise cancer diagnostics.
miRNAs and small molecule inhibitors therapeutics
miRNA therapy holds promise as an adjunct to standard therapies such as chemotherapy, radiotherapy, or immunotherapy. However, its success hinges on effective delivery systems capable of transferring miRNAs into target cells. While existing delivery systems possess certain limitations, a promising strategy involves targeting miRNAs that are selectively expressed in malignant cells while remaining absent in normal cells. This approach aims to mitigate toxicity, enhance therapy specificity, and simplify the quantification of specific miRNAs, thereby optimising therapeutic outcomes.
Small molecules have garnered significant attention in the medical field, emerging as pivotal elements in drug development. Particularly noteworthy is the increasing interest in discovering small molecule inhibitors targeting noncoding RNA (ncRNA), driven by the presence of modifications in RNA molecules that render them attractive drug targets. In this regard, small molecules can be tailored to selectively target miRNAs both before and after their maturation process. A recent study offers valuable insights into small molecule inhibitors, including the identification of structures capable of binding to the tertiary folded structures of miRNAs. Understanding this concept sheds light on how stem-loop hairpin secondary structures can fold into tertiary configurations, thereby forming bulges or pockets crucial for small molecule interactions. These findings underscore the potential for developing targeted therapies that leverage small molecules to modulate miRNA function effectively.
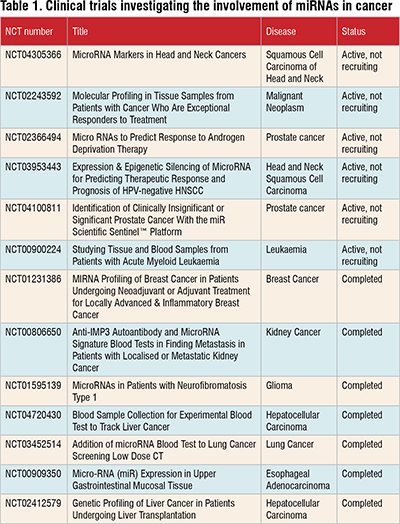
According to ClinicalTrials.gov, there are currently 417 ongoing clinical trials investigating the use of miRNAs in cancer. Among these trials, 16 focus on assessing miRNAs' prognostic significance, while 148 are specifically exploring their diagnostic potential in cancer. Notably, there are no ongoing clinical trials that directly combine miRNA research with cancer treatment using small molecule inhibitors. However, it's worth noting that the field of miRNA-based cancer therapy is continually evolving, and future trials may explore this approach.
Table 1 provides a snapshot of selected ongoing clinical trials in this area. Further research and clinical investigation are needed to fully understand the potential of miRNAs and small molecule inhibitors in cancer treatment.
Conclusions
miRNAs serve as pivotal regulators in various aspects of cancer biology. Further research is essential to fully understand and decipher the intricate processes governed by miRNAs, including their biogenesis, modulation of cancer hallmarks, and diverse functions. These complex processes offer valuable insights into strategies for combating cancer. To date, mounting evidence highlights the potential of miRNAs as biomarkers for prognosis and diagnosis across different pathologies. Moreover, the design of molecules specifically targeting miRNAs shows promise in counteracting various cancer mechanisms and ultimately improving the survival rates of patients diagnosed with various forms of malignancies.
Acknowledgements
G.A.C. is the Felix L. Haas Endowed Professor in Basic Science. Work in G.A.C.’s laboratory is supported by NCI grants 1R01 CA182905-01 and 1R01CA222007-01A1, NIGMS grant 1R01GM122775-01, DoD Idea Award W81XWH-21-1-0030, a Team DOD grant in Gastric Cancer W81XWH-21- 1-0715, a Chronic Lymphocytic Leukemia Moonshot Flagship project, a CLL Global Research Foundation 2019 grant, a CLL Global Research Foundation 2020 grant, a CLL Global Research Foundation 2022 grant, The G. Harold & Leila Y. Mathers Foundation, two grants from Torrey Coast Foundation, an Institutional Research Grant and Development Grant associated with the Brain SPORE 2P50CA127001.













