ETHICAL BUSINESS STIMULATES BEST VALUE IN HEALTH ECONOMY
Thomas Cueni, Director General, International Federation of Pharmaceutical Manufacturers and Associations (IFPMA)
Pharmaceutical business performance is widely perceived to be about robust sales, successful launches, financial results, profit margins, and the like. However, a very crucial measure of success relates to ‘how’ the industry conducts its business and achieves its mission. Ethics and building patients’ trust are at the core of how the R&D-based biopharmaceutical industry regulates itself today. In this opinion piece, Thomas Cueni, Director General of IFPMA, explains why the R&D-based biopharmaceutical industry has adopted the New Code of Practice and why it makes good business sense to win and retain the trust patients place in healthcare professionals, medicines and vaccines produced by the R&D-based biopharmaceutical industry.
Seventy years ago, average life expectancy globally was 47; today it is 72. Seventy years ago, 50 million people worldwide were infected with smallpox; today none. The disease is eradicated. Thanks to initiatives such as Gavi (the Vaccine Alliance), more than 80 per cent of children now get early immunisation. The world’s state of health is better than ever, even though headlines often focus on the negative: Zika, outbreaks of Ebola and the lack of a cure for Alzheimer’s. Successful biomedical research has, along with modern sanitation and global political will, contributed significantly to this phenomenal global health progress.
Yet, the pharmaceutical industry is often criticised in the public debate for the values that guide its behaviour. One wonders why is the pharma industry so exposed in the public debate? Perhaps the answer lies in the core nature of our business. Unlike any other industry that sells consumer goods, our innovations can save lives. This leads to raised expectations within society for the industry to meet the highest standards of safety and effectiveness in medicines as well as to ensure ethical business conduct. Companies that are virtually as one in putting “patients first” in their mission statements should not be surprised if the criticism is harsh when something goes wrong.
Return on Ethics
While it’s easy to prove the impact of bad commercial decision-making, ethics however, also impacts the bottom line. An analysis by Ethisphere1, an institute defining and advancing the standards of ethical business practices, has shown that there could be a 6.4 per cent “ethics premium” on the stock-market. Ethics and financial value are tied together. According to the World Bank and the European Bank for Reconstruction and Development (EBRD), more than 70 per cent of small and medium-sized enterprises (SMEs) in transition economies perceive corruption as an impediment to their business and one-third of SMEs see corruption as a major business obstacle.2 The consequences include higher consumer distrust, unfair business conditions and/or inaccessible market opportunities, stifled innovation and investment, burdensome regulations, and differing standards across borders leading to elevated business cost and legal risks. There is, therefore, a strong business and economic case for ethical conduct, since companies generally, and SMEs in particular, are the engine of economic growth but need to operate and innovate within ethical corporate environments.
In the “ethics business”, our industry has been and remains ahead of the curve. I’m proud to be associated with the world’s largest public-private partnership to strengthen ethical business practices in the medical device and biopharmaceutical sectors: The Business Ethics for APEC SMEs Initiative.

As the premier forum for facilitating economic growth, cooperation, trade and investment in the region, APEC (Asia-Pacific Economic Cooperation) has shown great leadership. The potential rewards of increasing business ethics are considerable since APEC countries account for approximately 60 per cent of world GDP (US$19.254 trillion) and about 47 per cent of the world trade. Not only does the APEC Business Ethics initiative addresses a major challenge facing SMEs in target sectors and developing economies (e.g., in the identification and cost-effective implementation of the highest standard of ethical business practice), but it sets out a collaborative vision for the APEC region as a whole. This vision showcases how ethical business can support innovation, as well as strengthen economic growth, increase competitiveness, foster cross-border trade, reduce public waste and mitigate regulatory burdens. And critically, it can strengthen health systems and improve patient outcomes, build public confidence and trust in healthcare systems, promote access to life-saving and enhancing products, and boost innovation capabilities.
The results are incredible. In five years, the initiative has achieved nearuniversal adoption of codes of ethics by medical technology and biopharmaceutical industry associations within APEC economies, more than doubling the total number of codes from 37 to 77, including within ten economies where they previously did not exist (Chile, China, Indonesia, Malaysia, Peru, Philippines, Singapore, Chinese Taipei, Thailand, Vietnam). High-standards of business ethics are now part of normal business in 18,000+ companies, making real progress in raising the ethics bar and laying a solid foundation for trust in the healthcare sector. The speed of this achievement is better understood when recognising that the region’s initial 37 codes in these sectors took over three decades to develop. These efforts will continue to take shape and will be given a further impulse at the 2019 APEC Business Ethics for SMEs Forum, Santiago de Chile, in September 2019. As industry co-chair, I look forward to taking part in these discussions and bringing to the table IFPMA’s contribution.
While the core of the innovative biopharmaceutical industry’s work is the discovery of new medicines and vaccines, it also needs to develop, promote, sell and distribute these in an ethical manner and in accordance with the rules and regulations for medicines and healthcare. It’s not just what pharmaceutical innovation achieves that matters, but how the industry goes about achieving it.
Ethical Collaboration
An efficient healthcare system depends on mutual trust between patients, healthcare professionals, regulators and pharmaceutical companies. Today, global organisations representing doctors, nurses and hospital workers all bestow a great deal of importance on the culture of ethics. And crucially, patient-centred care has emerged in recent decades as the solution for an improved healthcare systems. It represents a shift from “doctor knows best” to a new pact between patients and healthcare professionals, where the patients are actively involved at every stage of decision making regarding the effect of medicine on their well-being. However, to empower patients, the right environment is needed — one based on trust. Trust has proven to be a critical factor influencing a treatment decision. This includes whether a patient accepts a treatment and agrees to follow the prescription of medicines.
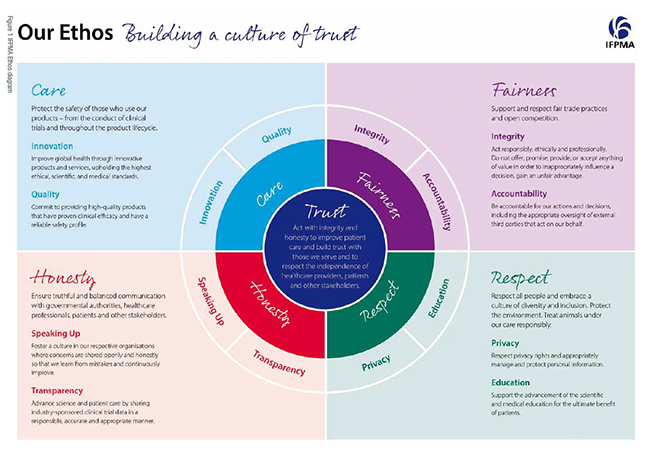
For this reason, in addition to ensuring a level playing field in terms of implementing codes of ethics, it is also essential to create consensus-based, multi-stakeholder ethical frameworks across health systems. In 2016, the world’s first economy-wide consensus framework agreements were concluded in Canada, Chile, Peru, and the Philippines, bringing together industry, healthcare professional associations, patients' organisations and governments. It is heartening to see that over the last summer, framework was also finalised in Australia, China, Mexico, Thailand, Japan and Viet Nam.
At a global level, in Geneva, the R&D biopharmaceutical industry has instigated in 2014 a consensus framework for ethical collaboration supported by patients (International Associations of Patient Organizations - IAPO), nurses (International Council of Nurses - ICN), pharmacists (the Federation of International Pharmacists - FIB), physicians (the World Medical Association - WMA) and our industry. All partners are united in a mutual interest in ensuring that the relationship between patients, healthcare professionals, the pharmaceutical sector and their representative organisations is based on ethical and responsible decision making.
These partners are part of an ethical healthcare ecosystem, with patients at the centre: because patients need to be able to trust the interactions between pharma, healthcare professionals and medical representatives. Given that what was acceptable and normal practice a few years ago may well be no longer acceptable, the IFPMA has reviewed and relaunched on January 1, 2019, its global Code of Practice as well as its ethical framework.
More Than a Code
But improving ethical conduct and increasing its undoubted benefits for the economy does not mean business and innovation should stop at codes and consensus frameworks. To be credible and in-step with societal expectation, we need to do more to reach all patients, regardless of their economic circumstances. Investment in health infrastructure, pathways to delivering services and prevention must be part of the dialogue.
Today, more than half the world’s population have to pay for their medicines out of pocket, potentially putting a huge strain on a patient’s family finances. Progress will be hampered as long as Universal Health Coverage (UHC) is not in place.
But this requires a political rewiring. Since far too often, governments worry more about the cost of treatment and prevention – but rarely do they tally the cost of inaction.
To do better, go further and faster, we’ll need partnerships, of all kinds. Partnerships are crucial to improving patient access through strengthening health systems: 90 per cent of ‘essential’ medicines are generics, and yet not all reach patients. Without access to prevention support and quality health services, global progress will be uneven. Achieving UHC means ensuring all people receive essential healthcare without risking financial hardship. There is also an important upside to this. As put so eloquently by Tedros Adhanom Ghebreyesus, Director General, at the World Health Organization (WHO): “If countries invest in making progress towards universal health coverage, they lay the foundation for progress towards ending poverty, improving gender equality, decent work and economic growth, and more.”
Achieving UHC is one of the health targets under the Sustainable Development Goals (SDGs), and will be the focus of the United Nations High Level Meeting (UNHLM) in September this year. Partnerships contribute to this by training healthcare workers, building infrastructure such as testing facilities, and delivering education to schools and community organizations to promote prevention. For instance, UHC2030 (a global platform for promoting UHC) brings together diverse stakeholders to advocate for political commitment, strengthen dialogue and facilitate knowledge sharing in order to strengthen health systems. These close partnerships could hardly have been imagined in previous decades and will only thrive with mutual trust.
References:
1. https://ethisphere.com/
2. The Business Environment and Enterprise Performance Survey (BEEPS)














