How Indian Pharma Manufacturers can Close the Gap with China
Gurmeet Singh, Procurement Director, Alfa Pharma GmbH
China's pharmaceutical industry holds a competitive edge due to its large domestic market, substantial R&D investments, advanced manufacturing capabilities for the basic pharmaceutical building blocks, and robust government support. For India to enhance its competitiveness, it must boost R&D investments, reduce API dependency, and improve its regulatory environment. China continues to solidify its position in innovative drug products by way of strategic alliances with large MNCs. Indian Pharma needs to take urgent steps in order to close the gap and remain relevant on the global stage.

India and China are the two most populous countries in the world, with the combined population of 2.85 billion people comprising almost 35 per cent of the world population. Unsurprisingly, it is of interest to any major pharma company to be a part of this huge business opportunity. These two countries also boast of their traditional medicines and methods of treatment which have come down the ages and have its own importance and space in the treatment of diseases.
India presently is a world leader in generic medicine. However, China has increased its investment in research and development and has a laser sharp focus on high-tech manufacturing and the new areas of biomedicine and biopharmaceuticals. The infrastructure in China in fermentation products, basic building blocks and key starting raw materials outstrips that of India. The main biopharmaceuticals in China namely Sinopharm Group and Shanghai Pharmaceuticals have a head start with critical support from multinationals like AstraZeneca, Pfizer, and Novartis. The west can illafford to look the other way due to the strengths of Infrastructure, advancements in biopharma and the large business opportunity that exists in collaborating in China with differing business models.
The Pharmaceutical Industry Verticals
The development in the field of Pharma and medicines can rightly be gauged in the various components that go into the making of a drug product. These components are: (Table: 01)
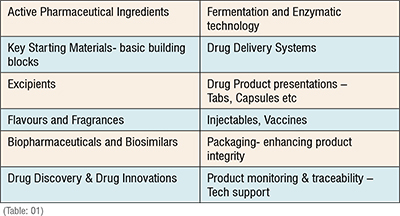
The Indian pharma sector has seen phenomenal growth during the period 1985 onwards when the industry got a window of opportunity especially between 1995 to 2005 where the process patent was respected and that gave a huge opportunity for the Indian scientist to get into reverse engineering and work on a large number of APIs. The industry also witnessed the decline of the public sector undertakings like the IDPL, HAL, SPIC during this period.
There was, however, a total absence of government support. Meanwhile, Indian companies experienced a mushroom growth of API manufacturers later graduating to finished formulations. However there were some areas where the industry took missteps, such as by dropping the manufacture of all-important basic building blocks, intermediates and key starting raw materials and going in for easy routes of economical imports from China. In doing so, they lost out vis-à-vis China and became heavily dependent to the extent of 80 per cent of the critical raw materials.
The COVID-19 impact
The world noticed the shake-up of the supply chains resulting in critical shortages impacting the fine balance between demand and supply. The western countries realised the importance of ‘reverse swing’ or reshoring and ‘friend shoring’ which gave rise to the China+1 policy as well. The Indian government too realised the importance of reworking their strategies and created the production linked incentive (PLI) scheme. The government has provided financial assistance and favourable tax treatment for manufacturers to make the products that were stopped at some point in time due to various commercial reasons. While results on the ground will be evident in the years to come, it is a step in the right direction.

Pharmaceutical Industry: Important factors for growth
1. Intellectual Capital and patent regime
2. The regulatory framework and compliance
3. Quality Control (Quality by design)
4. New Drug Discovery capability (role of AI and ML)
5. Costs(Energy, Manpower)
6. Technical Strengths
7. Infrastructure and automation (Fermentation capacities)
8. Perishables and Cold Chains
9. Preparedness for the next global disruption
10. Mergers & Acquisitions
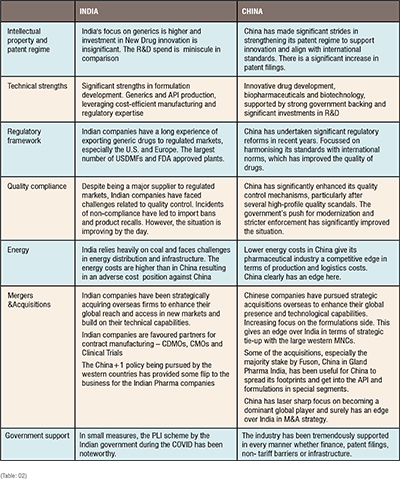
India and China – A comparative analysis
The table below compares the key strengths and weaknesses on the important parameters namely the intellectual property and patents, technical strengths, regulatory framework, quality compliance, energy and mergers and acquisitions. (Table: 02)
The Technological advancements in pharma
Technological advancements in the pharmaceutical industry have transformed drug discovery, development, manufacturing, and distribution. In addition to the broad parameters that have been discussed in the table above, we have a host of new areas that are already impacting the growth and direction in which the new innovations and developments will fast track new drugs, drug delivery systems to address the special and critical needs of humanity. The recent pandemic has given rise to innovative ways of addressing various issues from drug identification to commercialisation. Importantly, this experience will help pharma companies address the next unknown pandemic more efficiently.
Some of the new innovations that are finding space in the field of pharmaceuticals include the following:
• Artificial Intelligence and Machine Learning: Drug discovery and design and clinical trials
• Big Data and Analytics: development of biologics
• Genomics and precision medicine: personalised medicines and targeted therapies
• A larger share of biopharmaceuticals being available going forward, as the sector grows
• 3D Printing: creation of customised drug formulations and dosages tailored to individual patient needs
• Digital Health and Wearables: devices monitor patients’ health metrics in realtime.
• Blockchain: supply chain product integrity with respect to traceability of drugs in the entire supply chain.
• Advanced drug delivery systems: Nanotechnology, Nanoparticles and Nano medicine
• Robotics and Automation: will reduce human error, enhance quality control, and accelerate production timelines, along with automating manufacturing and laboratory operations
• Telemedicine and E-Health Platforms: will enable remote consultations, improving access to care.
• Regenerative medicine: - stem cell therapies will lead to new treatments.
Lessons for India
India has been slower compared to China in terms of technological advancements and gaining the traction of large western based MNCs. India has a real task on hand to bridge the gap and enter the higher value-add platforms such as new drug discovery and drug delivery systems rather than lower level contract manufacturing projects.
India must enhance its competitiveness, it must boost R&D investments, reduce API dependency by creating in-house the critical basic building blocks, the starting raw materials and improve its regulatory environment and protection of intellectual property. Fast changing geo-political situation has given a window of opportunity to the Indian Pharma to gear up and its share of the pie. Nevertheless, India will remain for quite some time the preferred destination for medical tourism. One aspect that India has to focus on is the menace of fake medicines in the market and growing concerns around antimicrobial resistance (AMR) – that the population is experiencing due to self-medication. India also needs to enhance its cold chain infrastructure to ensure drug product integrity. Meanwhile, China continues to leverage its market size, and technological advancements.
Technological advancements are not only revolutionising the pharmaceutical industry but also leading to better, safer, and more effective treatments for patients.
They are driving the industry toward a future where personalised medicine, efficiency, and innovation are at the forefront, ultimately improving global health outcomes. Here too, India needs to narrow the gap that exists between the two countries. Now is the time to act, although India needs to run faster to remain in the race. In the coming years, the government should actively support the industry by funding, in addition to the tax sops, and not shy away from guarding the industry both by way of tariff and non-tariff barriers under the permissible rules of the WTO.
It is indeed a marathon and not a sprint, and a well thought out strategic collaboration among industry,academia and government will ensure that we remain on the exponential growth trajectory.
References:
Pharma and life sciences in 2024 top 11 trends: https://www.avenga.com/magazine/pharmaceutical-industry-trends/
India’s Import Dependence on China in Pharmaceuticals: Status, Issues and Policy Options: https://ris.org.in/sites/default/files/Publication/DP%20268%20Prof%20Sudip%20Chaudhuri.pdf
China vs. India: the fight for pharma supremacy: https://www.raconteur.net/global-business/china-vs-india-the-fight-for-pharma-supremacy
WHO Director-General declares mpox outbreak a public health emergency of international concern: https://www.who.int/news/item/14-08-2024-who-director-general-declares-mpox-outbreak-a-public-health-emergency-of-international-concern
Newpaper reports and various Pharma magazines














