Modernising Asia's Drug Regulations
AI and Quantum Computing as catalysts for harmonised pharmaceutical oversight
David Braga, CEO, 5th Order Cyber Corporation
Bharat Rawal, Department Head of Grambling State University’s Department of Computer Science and Digital Technologies
Larry Braga, CMC Regulatory Affairs Consultant
The Asia-Pacific pharmaceutical market faces fragmented regulations, slow approvals, and weak enforcement. This article examines how AI and quantum computing can modernise processes, enhance security, and drive regulatory harmonisation, improving global competitiveness and patient access to innovative therapies.

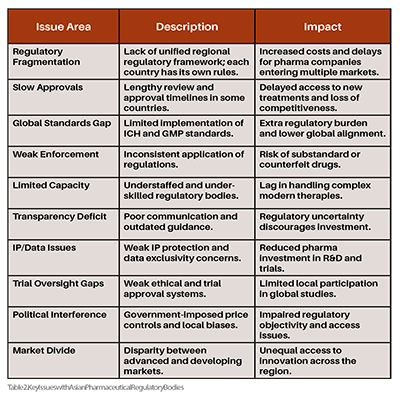
Regulatory Fragmentation Across Countries
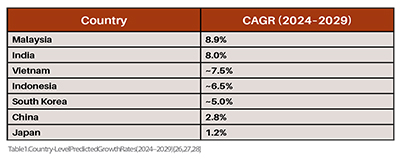
Although Asia’s pharmaceutical sector produces close to half of the world’s specialty generics, biologicals, and active pharmaceutical ingredients (APIs) Table 1, it lacks a unified regulatory framework akin to the EMA in Europe or the FDA in the U.S. Each country has its own rules, standards, and approval pathways. American regulatory pathways consistently outperformed the EU and Asia, achieving accelerated approval through earlier application submissions and more streamlined review mechanisms at the US regulatory body. As a result, multinational pharmaceutical companies must navigate multiple, often conflicting regulatory environments, which increases the time and cost of drug registration and compliance. (Table 1)
Slow Drug Approval Timelines
Regulatory review and approval processes can be significantly slower in some Asian countries (e.g., Indonesia, India, and the Philippines) than in Western countries. The Asian pharmaceutical industry was generally considerably slower than other regions, primarily due to delays in regulatory reviews after new drug application (NDA) and extended clinical development timelines. Delays in access to new treatments for patients; loss of global competitiveness in innovation and early-market access.
Limited Harmonisation with Global Standards
Some Asian regulatory authorities have not fully adopted or implemented International Council for Harmonisation (ICH) () guidelines or Good Manufacturing Practices (GMP). There is a disparity in, for example, all twelve Asian regulatory systems with China, India, Indonesia, Japan, the Philippines, Singapore, and Vietnam, where e-submissions are entirely paperless due to advancements in regulatory agility, regional collaboration, and digital transformation. In contrast, the remaining economies still require some form of physical documentation to be submitted. Regulatory submissions may need substantial localisation or re-validation, from prioritising these markets.
Inconsistent Enforcement of Regulations
Where standards do exist, enforcement is uneven due to underfunded regulatory agencies, lack of trained staff, or political interference. Although global attention is often directed toward counterfeit or fake medications, a significant issue is the prevalence of substandard or poor-quality medicines. Regulatory agencies in Asian nations often do not have adequate infrastructure to guarantee pharmaceutical products’ quality, effectiveness, and safety. Variable drug quality, counterfeit medications, and diminished public trust in healthcare systems.
Insufficient Regulatory Capacity
Many agencies are understaffed and lack the technical expertise or digital infrastructure to evaluate complex new therapies (e.g., biologics, mRNA, AI-driven drugs). The Asian pharmaceutical regulatory bodies often have redundant and conflicting requirements that are not removed in lieu of implementing good reliance practices. This poses major concerns because of duplicative efforts with minimal value in regulatory oversight and the inability to keep pace with global innovation trends, particularly in precision and personalized medicine.
Limited Transparency and Communication
Regulatory decisions are not always publicly explained, and guidelines are often not published or updated regularly. There is a lack of a value framework for assessing health technologies in Asian settings. There is a contrasting spectrum of transparency and communication expertise at different timelines of development, from relatively mature systems in Taiwan, Singapore, Hong Kong, Malaysia, and Thailand to emerging frameworks in India, Indonesia, the Philippines, and Vietnam, and nascent systems in Pakistan. This creates uncertainty for pharmaceutical companies and investors, making it difficult to plan a regulatory strategy.
Lack of Data Protection and IP Enforcement
In some countries (e.g., India, China), concerns persist about enforcing intellectual property (IP) rights and data exclusivity. Asian countries, especially China and India, have been scrutinised for how they protect the rights of intellectual property (IP) and the issue of proper IP protection and enforcement, especially in the pharmaceutical industry. IP violations across borders pose a complex challenge for the Asian Pharmaceutical Industry, requiring a thorough grasp of the various issues and potential remedies. This discourages global pharma from investing in R&D or conducting clinical trials in those regions.
Inadequate Clinical Trial Infrastructure and Oversight
Some Asian countries face weak ethical oversight and slow trial approvals, limiting local participation in global studies. Therapies evaluated through clinical trials in high-income countries may not be suitable for direct use in low- and middle-income countries, as differences in sociocultural norms, genetics, and environmental conditions can influence outcomes. Additionally, these therapies often face significant financial and logistical barriers to implementation in such settings. This limits access to innovative drugs and delays regional trial data generation needed for local approvals.
Political and Economic Interference
Government price controls, local manufacturing preferences, and nationalistic policies can interfere with regulatory independence. In the Asian pharmaceutical industry, the goal of implementing compliance or compliance management is typically to ensure that organisational and managerial activities align with established laws, standards, and regulatory requirements. Although considerable research has explored public health collaboration between China and Southeast Asian nations, much of it places disproportionate focus on the influence of major powers, often overlooking the contributions and agency of Southeast Asia's small and medium-sized states. Reduced objectivity and delays in decision-making; challenges for foreign companies in market access.
Emerging vs. Mature Market Divide
While countries like Japan, South Korea, and Singapore have advanced systems, others like Myanmar, Vietnam, and Bangladesh still develop fundamental regulatory capacities see Table 2. These Asian nations have made substantial investments in biomedical sciences to strengthen their knowledge base. However, they have faced challenges in transforming these scientific advancements into a globally competitive biomedical industry. The impact of foreign direct investment (FDI) on Asian domestic economies tends to vary, largely due to notable differences in institutional frameworks, industrial bases, and resource endowments across the region. As a result, this creates uneven development across Asia, with innovation concentrated in a few hubs (Table 2).
How ai and quantum can help asian regulatory challenges
Harmonisation and Standardisation
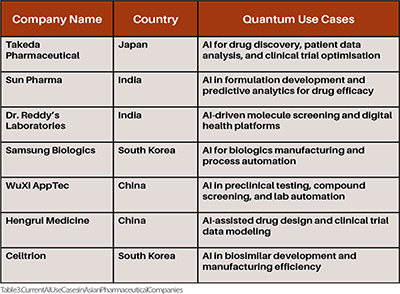
AI can analyse and align disparate regulatory frameworks across countries, proposing harmonised models based on global best practices Table 3. In drug discovery, AI leverages machine learning to predict compound efficacy and safety with high precision, enabling faster, cost-effective early development and drug repurposing compared to traditional methods. Quantum computing may enhance and expedite the drug discovery and development process, but actual product approvals will continue to depend on conventional empirical evidence from laboratory and clinical studies. Quantum data might become more integrated into the regulatory processes and an additional tool for decision-making. Still, it will only replace traditional methods after substance control validation and regulatory changes. (Table 3)
Faster Drug Approval Processes
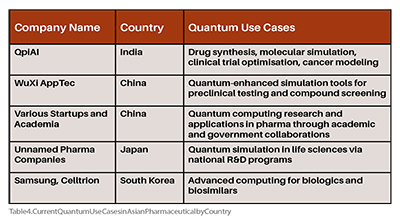
AI-driven systems can automate portions of dossier review and safety checks, reducing human bottlenecks. The adoption of AI in Asina healthcare began with automating and digitising data-intensive processes, progressing to robotics integration and ultimately enabling the broader application of advanced AI technologies to enhance medical innovation. Quantum computing can accelerate molecular simulations, shorten preclinical and clinical development timelines, and generate faster submissions. Virtual clinical trials could reduce the need for extensive physical testing, speed up drug development, and reduce costs. As quantum computing matures, it could revolutionise drug discovery by enabling the simulation of complex molecular interactions beyond the reach of classical computers, potentially leading to the discovery of new drug classes and therapies. (Table 4)
Improved Regulatory Capacity
AI tools can augment regulators' decision-making by providing predictive modeling, anomaly detection, and clinical outcome forecasts. Adherence to AI regulatory compliance correlates with AI implementation’s perceived effectiveness and data integrity. AI-powered platforms can also offer training modules and real-time guidance for regulatory staff. Regulatory processes may become more streamlined, utilising adaptive clinical trials, accelerating pathways, and better integrating digital data to reduce the time and cost of bringing new drugs to market.
Enhanced Clinical Trial Oversight
AI can monitor real-time data from decentralised and adaptive trials, while quantum computing can optimise patient stratification and biomarker discovery. The Asian Drug regulatory bodies should work with the U.S. FDA, a global leader in regulatory oversight for emerging technologies from 3D-printed drugs to AI-driven medical tools, which has expanded its role by regulating Software as a Medical Device (SaMD) used for disease prevention, diagnosis, treatment, and monitoring. Regulatory processes may become more streamlined, utilising adaptive clinical trials, accelerating pathways, and better integrating digital data to reduce the time and cost of bringing new drugs to market. This leads to safer, more efficient trials that meet international standards.
Strengthened Data Integrity and IP Protection
Blockchain integrated with AI can ensure data provenance and authenticity in regulatory submissions. As Asia’s rapidly evolving economic and healthcare landscape demands effective real-world data (RWD) for regulatory and clinical decisions, critically evaluating nationspecific RWD generation and its use in producing real-world evidence (RWE) has become essential. Open-source platforms and data sharing will become more common, facilitating progress in drug development. Compliance can be ensured with world-standard data protection regulations (e.g., GDPR, HIPAA) in AI/ quantum pipelines. Quantum encryption can bolster IP security, deterring cyber breaches in sensitive clinical and pharma data ecosystems.
Transparent and Inclusive Communication
AI-driven dashboards and chatbots can offer real-time access to guidance documents and status updates, enhancing stakeholder transparency. Multilingual AI translation helps non-native speakers access updated rules. Quantum cryptography or quantum key distribution potentially yields new information processing and communication methods. Almost all quantum information processing protocols can be divided into three distinct stages: (i) state preparation, (ii) state transformation, and (iii) final measurement. By strengthening secure, accessible, and multilingual communication channels, these technologies create a foundation for proactively identifying and addressing threats in pharmaceutical supply chains.
Early Detection of Counterfeit Drugs
Computer vision and AI-enabled supply chain tracking can detect irregularities and counterfeit medications. Quantum sensors and AI together can validate drug integrity and storage conditions more precisely. Counterfeit medicines pose a global crisis, largely originating from developing nations in Asia, where weak law enforcement and low-income populations with high medical needs contribute to the problem. These emerging technologies highlight the critical need for regulatory systems to evolve, ensuring that oversight mechanisms remain effective as pharmaceutical supply chains and threats become increasingly complex.
Future-Proofing Regulatory Systems
Counterfeit medicines, often called “falsified” or “adulterated,” involve the deliberate alteration of a drug’s identity, origin, or history. This global crisis is prevalent in Asia, where weak law enforcement and low-income populations with high medical demand exacerbate the problem. AI and quantum technologies support continuous learning systems that adapt to emerging therapies like gene editing, mRNA, and precision medicine. This keeps regulatory frameworks responsive and globally competitive. Asian Regulatory bodies can rely on empirical data from laboratory experiments and clinical trials to evaluate the safety and efficacy of new drugs, medical devices, and food products. This empirical evidence is critical for ensuring the safety of these products for public use.
Conclusion
The Asian Pharmaceutical Industry stands at a pivotal crossroads in its pharmaceutical evolution. While the region has emerged as a global drug production and innovation powerhouse, outdated and fragmented regulatory frameworks threaten to stall progress. The pressing issues, ranging from slow approvals and weak enforcement to disparities in market maturity, highlight the urgent need for modernisation. Fortunately, the convergence of artificial intelligence and quantum computing offers a transformative pathway forward. These technologies are not merely tools of convenience but essential levers for regulatory harmonisation, transparency, and future-readiness. By embracing AI-driven decision-making, real-time data analysis, and quantum-enabled drug discovery and encryption, Asian regulators can leapfrog traditional barriers and align with global gold standards. The future of Asia’s pharmaceutical sector depends on its ability to build innovative, adaptive, and inclusive regulatory ecosystems. The time to act is before innovation outpaces oversight and global opportunity slips away.
Acknowledgement
We would like to thank Dr. Rawal and Larry Braga for their contributions to this article.
Conflicts Of Interest
There are no conflicts of interest.
Funding Source
There is no source of funding.
REFERENCES
1. Basak, S. K. (2023). From Pharmaceutical Innovation to Revenue Generation: The Asian Experience. The Journal of Law, Medicine & Ethics, 51, 62-75. https://doi.org/10.1017/jme.2023.121
2. Yan, Y., Guo, X., Li, Z., Shi, W., Long, M., Yue, X., Kong, F., & Zhao, Z. (2025). New Drug Approvals in China: An International Comparative Analysis, 2019-2023. Drug design, development and therapy, 19, 2629–2639. https://doi.org/10.2147/DDDT.S514132
3. Advancements in regulatory agility, regional collaboration, and digital transformation: insights from the Asia Partnership Conference of Pharmaceutical Associations (APAC). (2024). AAPS Open, 10(1), 14. https://doi.org/10.1186/s41120-024-00102-2
4. Robertson, J., Santoso, B., Holloway, K. A., Dartnell, J., Tisocki, K., McLachlan, A., & Smith, A. (2012). Asia Pacific Conference on National Medicines Policies. Australian Prescriber, 35(6), 190. https://doi.org/10.18773/austprescr.2012.085
5. Feeney, A. J., Goad, J. A., & Flaherty, G. T. (2024). Global perspective of the risks of falsified and counterfeit medicines: A critical review of the literature.Travel Medicine and Infectious Disease, 61https://doi.org/10.1016/j.tmaid.2024.102758
6. Nguyen, T. T. D., Lee, Y. H., Lin, Y. J., Chang, S. C., Hsiao, F. Y., Chang, C. J., & Ou, H. T. (2025). Value Framework Based on Multiple-Criteria Decision Analysis for Assessment of New Health Technologies Under Universal Healthcare Coverage System in Taiwan. Value in Health, 28(2), 241-249.
7. Guilherme, S. J., Wen-Yi Shau, Hsu-Wen, C., & Setia, S. (2024). Bridging Real-World Data Gaps: Connecting Dots Across 10 Asian Countries. JMIR Medical Informatics, 12https://doi.org/10.2196/58548
8. Huang, C., Cao, C., & Coreynen, W. (2024). Stronger and more just? Recent reforms of China’s intellectual property rights system and their implications. Asia Pacific Journal of Innovation and Entrepreneurship, 18(3), 210-223. https://doi.org/10.1108/APJIE-04-2023-0081
9. Feng, S., & Sik, C. P. (2024). Multifaceted Challenges of Jurisdictional Divergence in Cross-Border Intellectual Property Violations. International Journal of Criminal Justice Sciences, 19(1), 20-40. https://doi.org/10.5281/zenodo.19102
10. M, S. H., Acharya, S. C., Gautam, M., Silwal, S. R., Sapkota, S., Poudyal, S., Sharma, S., K, G. B., Nigar, T., Pervin, S., Gulia, S., Gunasekara, S., A F M Kamal, U., Tshomo, U., Safi, A. J., Nadeem, M. S., Masood, A. I., Sumon, M. A., Purvin, S., . . . Wijesooriya, K. (2024). Cancer research in South Asian Association for Regional Cooperation (SAARC) countries. Lancet Oncology, 25(12), e675-e684. https://doi.org/10.1016/S1470-2045(24)00518-7
11. Dai, F. (2023). The new dynamics between the state, local ideals, and (anti-) corruption: an ethnographic study of the ethical practices of China's pharmaceutical industry. Inter-Asia Cultural Studies, 24(2), 349-365.
12. Zhang, L. (2025). Shared Knowledge and Local Practice: The Evolution of Public Health Cooperation between China and Mainland Southeast Asian Countries. China Review, 25(2), 83-119. https://library.capella.edu/login?url=https://www.proquest.com/scholarly-journals/shared-knowledge-local-practice-evolution-public/docview/3222855577/se-2
13. Zhou, Y., & Sun, F. (2022). Creating Knowledge Assets under Bio capitalism: Analyzing China’s Biomedical Industry and Its Patent Networks. Economic Geography, 98(5), 411–437. https://doi-org.library.capella.edu/10.1080/00130095.2022.2070471
14. Liu, N., & Moon-Gyu, B. (2025). Linking Inward Foreign Direct Investment to Innovative Entrepreneurship: The Mediating Role of Economic Institutions in Chinese Regions. Sustainability, 17(10), 4290. https://doi.org/10.3390/su17104290
15. Allam, H. (2025). Prescribing the Future: The Role of Artificial Intelligence in Pharmacy. Information, 16(2), 131. https://doi.org/10.3390/info16020131
16. Kumar, G., Yadav, S., Mukherjee, A., Hassija, V., & Guizani, M. (2024). Recent advances in quantum computing for drug discovery and development. IEEE Access, 12, 64491-64509.
17. Arshad, A. S. (2023). SIRIM Berhad: The Frontier in Medical and Health Technology. The Malaysian Journal of Medical Sciences, 30(4), 1-7. https://doi.org/10.21315/n1jn1s2023.30.4.1
18. Avramouli, M., Savvas, I. K., Vasilaki, A., & Garani, G. (2023). Unlocking the Potential of Quantum Machine Learning to Advance Drug Discovery. Electronics, 12(11), 2402. https://doi.org/10.3390/electronics12112402
19. Sembiring, M. H., & Novagusda, F. N. (2024). Enhancing Data Security Resilience in AI-Driven Digital Transformation: Exploring Industry Challenges and Solutions Through ALCOA+ Principles. Acta Informatica Medica, 32(1), 65-70. https://doi.org/10.5455/aim.2024.32.65-70.
20. Meskó, B., & Topol, E. J. (2023). The imperative for regulatory oversight of large language models (or generative AI) in healthcare. NPJ digital medicine, 6(1), 120. https://doi.org/10.1038/s41746-023-00873-0
21. Guilherme, S. J., Wen-Yi Shau, Hsu-Wen, C., & Setia, S. (2024). Bridging Real-World Data Gaps: Connecting Dots Across 10 Asian Countries. JMIR Medical Informatics, 12https://doi.org/10.2196/58548
22. Lamata, P., Niederer, S., Nordsletten, D., Barber, D. C., Roy, I., Hose, D. R., & Smith, N. (2011). An accurate, fast and robust method to generate patient-specific cubic Hermite meshes. Medical image analysis, 15(6), 801-813.
23. Schlichtholz, K., & Markiewicz, M. (2024). Relativistically invariant encoding of quantum information revisited. New Journal of Physics, 26(3), 033018.
24. Authorship, & SCIMAGO, I. R. (2023). Counterfeit medicines: relevance, consequences and strategies to combat the global crisis. Brazilian Journal of Pharmaceutical Sciences, 59https://doi.org/10.1590/s2175-97902023e20402
25. Srivastav, A. K., Mishra, M. K., Lillard Jr, J. W., & Singh, R. (2025). Transforming pharmacogenomics and CRISPR gene editing with the power of artificial intelligence for precision medicine. Pharmaceutics, 17(5), 555.
26. Asia Pacific Pharmaceutical Market Size & Outlook, 2030 Date accessed, August 2, 2025, Date published, May 28, 2025 https://www.grandviewresearch.com/horizon/outlook/pharmaceutical-market/asia-pacific
27. Key tailwinds and headwinds impacting the outlook for the Asian Pharmaceutical Market – IQVIA
key-tailwinds-and-headwinds-impacting-the-outlook-for-the-asian-pharmaceutical-market.pdf Date accessed August 2, 2025.
28. Top 10 Pharma Trends for 2024 Industry Trends Pharmaceuticals September 2024, Date accessed August 2, 2025
29. 11 Big Pharma companies are using AI for industry transformation ://www.pharmaceuticalprocessingworld.com/ai-pharma-drug-development-billion-opportunity/. Date accessed August 2, 2025
30. Big Pharma Is Investing Billions In AI-And The Value Is Finally Starting To Show . Forbes: https ://www.forbes.com/sites/saibala/2025/04/25/big-pharma-is-investing-billions-in-ai--and-the-value-is-finally-starting-to-show/. Date accessed August 2, 2025
31. Quantum Computing in Biopharma: Future Prospects and Strategic Insights: L.E.K. Consultinghttps://www.lek.com/insights/hea/us/ei/quantum-computing-biopharma-future-prospects-and-strategic-insights.Date accessed August 2, 2025
32. D-Wave's New AI/ML Quantum Cloud Service Roadmap, Implications for Drug Discovery. Biopharma Trend. https://www.biopharmatrend.com/post/877-d-waves-new-aiml-quantum-cloud-service-roadmap-implications-for-drug-discovery/. Date accessed August 2, 2025
33. Quantum Technology Monitor https://www.mckinsey.com/~/media/mckinsey/business functions/mckinsey digital/our insights/the year of quantum from concept to reality in 2025/quantum-monitor-2025.pdf. Date accessed August 2, 2025














