EDITORIAL SECTIONS
RESEARCH & DEVELOPMENT
Adopting the Power of AI to Drug Development Projects
A tremendous amount of documentation is created throughout the clinical trial lifecycle
CLINICAL TRIALS
Real-world Evidence
Hearing the diagnosis from the doctor that your child has spinal muscular atrophy SMA
MANUFACTURING
Just in Time Manufacturing Creating more sustainable supply chains
Drug development has transformed immeasurably over the past decade Even before the Covid pandemic and the conflict in Ukraine
Information Technology
Challenges and Advantages of Using Artificial Intelligence in Pharma
Many advanced largescale AI initiatives still need to be proved successful in Pharma
KNOWLEDGE BANK
Interviews
Innovation Management at Bachem
Chief Technology Officer, Bachem Holding AG
Gnther Loidl joined Bachem in He held several functions in Research and Development RD and Active Pharmaceutical Ingredient Manufacturing and became Vice President RD in Since he is CTO of the Bachem Group and a member of the Corporate Executive Committee He studied chemistry in Regensburg Germany and Leeds UK and obtained his doctorate under the supervision of Luis Moroder at the Max Planck In...
Articles
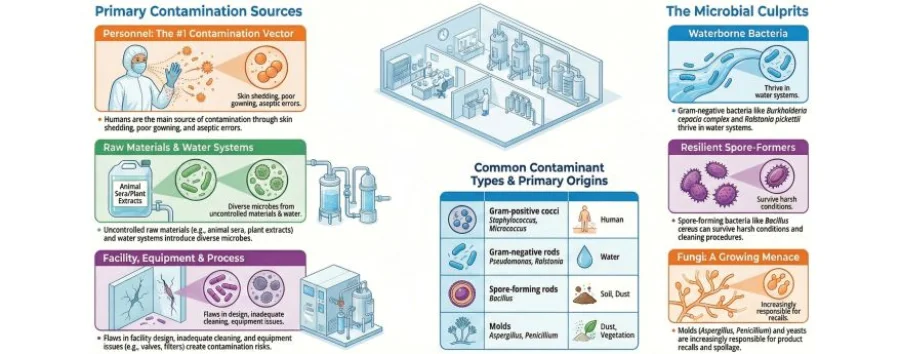
Advancing Automation and Digitalization in the Industrial Microbiology Workflow
Industrial microbiology is a critical function in the biopharmaceutical sector as its purpose is to monitor for contamination events and inform development of standard operating procedures to minimize the risk of contamination As the industry makes the shift to digitalized labs you will see more automation digitalization
Research Insights
SARS-CoV-2 Omicron Variations Reveal Mechanisms Controlling Cell Entry Dynamics and Antibody Neutralization
Severe Acute Respiratory Syndrome Coronavirus SARSCoV is adapting to continuous presence in humans Transitions to endemic infection patterns are associated with changes in the spike S proteins that direct viruscell entry These changes generate antigenic drift and thereby allow virus maintenance in the face of prevalent human antiviral antibodies
Case Studies
Tablet Coating at Low Bed Temperatures with a Novel Coating Polymer
This study shows how to obtain elegant uniform coatings with zero visible defects and a practical coating solution for heat sensitive products For More Infromation Click Here
Techno Trends
New Antibiotic by Merck that Fights Bacteria
In the present drug discovery scenario there are very few pharmaceutical companies who invest in research into antibiotics as majority prefer to focus on profitable drugs for chronic diseases Most drugs were developed years ago and older antibiotics have lost their effect as the bacteria have become resistant to them Merck has developed an antibiotic platensim
White Papers
Cloud Computing in the Laboratory Informatics Solutions and Validation Approaches
In the modern business environment, a significant portion of work is conducted using computerized systems. Many solutions are used to complete tasks, including simple desktop programs like email and Microsoft Office to enterprise-wide, global systems like ERP (Enterprise Resource Planning), MES (Manufacturing Execution System), and LIMS
TOP ARTICLES
QSAR - Changing the Landscape of Antidiabetic Drug Discovery
Smriti Sharma Bhatia is an associate professor of chemistry at Miranda House, University of Delhi, India. Her academic and research interests include Computational chemistry and drug design. Currently, her research group is focused on the design of oral drug formulations for Type 2 diabetes mellitus. She is a popular science communicator.
Green Synthesis of Metallic Nanoparticles
Ms. Aishwarya Jain is a distinguished pharmaceutical scientist and academician with over six years of experience in pharmaceutical sciences. Currently serving as an Assistant Professor at Sinhgad College of Pharmacy, Vadgaon (Bk), Pune, she has carved a niche for herself in the domains of nanotechnology, green synthesis, and pharmaceutical innovation.
A recipient of 65+ prestigious national and international awards, including the Bharat Shiksha Gaurav Puraskar and the Women Empowerment Award, Ms. Jain is recognised as one of the leading voices in pharmaceutical education and research. Her scholarly contributions span numerous books, book chapters, and high-impact research articles published in top-tier journals indexed in SCOPUS and Web of Science.
Renowned for her commitment to academic excellence and research integrity, she also plays an active role as an editorial board member and peer reviewer for reputed scientific journals and publishing houses. Her work exemplifies a seamless blend of academic rigor and innovative thinking, particularly in green nanotechnology and sustainable drug development.
Driven by a vision to advance pharmaceutical science for global health impact, Ms. Aishwarya continues to inspire through her interdisciplinary collaborations, cutting-edge research, and passionate teaching.
The Potential of Cell Free Chromatin Inhibition in Cancer
Divya Mishra is a board certified oncologist with over 17 years’ experience in the pharmaceutical/CRO industry, clinical practice, and academia, in areas of oncology/haematology, family medicine, palliative medicine, and outcomes research. Mishra has worked across multiple geographies and diverse, multicultural settings for organisations such as Pfizer, IQVIA, and SFJ. She is currently working at ICON as a medical director and therapeutic expert for the oncology/haematology disease areas in the APAC region, based in the regional headquarters in Singapore.
Determinants of Antimicrobial Resistance (AMR) Research in South East Asia
Raja Chakraverty is a serving Scientist with the Indian Council of Medical Research with a decade long research and teaching career in Pharmaceutical Sciences with 54 publications (186 citations) and a book
Doctoral Scholar, Department of Pharmaceutical Technology
Endocrine Disrupting Compounds (EDCs)
Sintu Kumar Samanta has been working as an Assistant professor in IIIT Allahabad, India. He did Ph.D. and post-doctoral research from IIT Kharagpur and IISc Bangalore, India respectively. He is working in the area of Biochemistry and Nanobiotechnology. He published research work in several international journals and filed one Indian patent.
Pavan Kumar Gautamis presently working as a Post-Doctoral Fellow in the Department of Applied Sciences at IIIT Allahabad, India. He obtained Ph.D. from the University of Allahabad, India. His area of research is synthesis of nanoparticles for water remediation. He published his research work in many international journals.
press releasesRead more...
SciSparc: Subsidiary NeuroThera Labs Received Conditional Regulatory Approval for Acquisition of CliniQuantum
CiSparc Ltd Nasdaq SPRC Company or SciSparc today announced that NeuroThera Labs Inc TSXV NTLX NeuroThera a clinicalstage pharmaceutical company focused on developing novel treatments
RenovoRx Announces FDA Orphan Drug Designation Granted: Oxaliplatin for the Treatment of Pancreatic Cancer
RenovoRx Inc a life sciences company developing innovative targeted oncology therapies and commercializing RenovoCath a novel FDAcleared drugdelivery device today announced that the US Food and Drug Administratio
Alkem reports record high EBITDA, strong revenue growth in Q4 & FY26
Alkem Laboratories Ltd today announced its standalone and consolidated financial results for the fourth quarter ended March The Board of Directors took record of these results at its meeting held in Mumbai today
Curevo to be Acquired by Lilly to Advance Next-Generation Shingles Prevention
Curevo Vaccine Curevo a clinicalstage biotechnology company dedicated to developing varicella zoster virus VZV vaccines with improved tolerability today announced entry into a definitive agreement to be acquired by Lilly
Biocytogen Launches AI-Powered RenSuper™ Platform and Industry-First Fully Automated Antibody Discovery Infrastructure
Biocytogen today announced the launch of RenSuper Workstation a nextgeneration AIpowered antibody discovery platform providing offtheshelf access to a largescale experimentally validated library
LimmaTech Biologics to be Acquired by Lilly for Next-generation Vaccines Against Common Bacterial Infections
LimmaTech Biologics AG a Swiss clinicalstage biotech company developing vaccines for the prevention of lifethreatening diseases today announced entry into a definitive agreement to be acquired by Lilly
EventsRead more...
01 - 02
Jun 2026Future Pharma 2026
Renaissance Seaport District, Boston, MA, USA
02 - 04
Jun 2026CPHI Americas 2026
Pennsylvania Convention Center, Philadelphia, USA
04 - 05
Jun 2026Pharmac South 2026
Chennai Trade Centre, Nandambakkam, India
04 - 05
Jun 2026Global Bioprocessing Summit & Exhibition 2026
Berlin, Germany
09 - 11
Jun 20265th World ADC Summit South Korea
COEX Magok Lewest, 143 Magokjungang-ro, Gangseo-gu, Seoul, South Korea
09 - 11
Jun 2026World TPD & Induced Proximity Summit South Korea
COEX Magok Lewest, 143 Magokjungang-ro, Gangseo-gu, Seoul, South Korea
09 - 11
Jun 2026World Targeted Therapeutics Summit South Korea
COEX Magok Lewest, 143 Magokjungang-ro, Gangseo-gu, Seoul, South Korea
09 - 10
Jun 2026ChemE Show 2026
George R. Brown Convention Center, Houston, Texas, USA
LATEST NEWSRead more...
Ionis Partner GSK Reports Positive Phase 3 Results for Bepirovirsen in Chronic Hepatitis B
Ionis Pharmaceuticals partner GSK has announced positive pivotal results for bepirovirsen an investigational antisense oligonucleotide ASO being developed for chronic hepatitis B CHB Findings from the Phase BWell
Pfizer and Innovent Biologics Sign Global Collaboration to Advance Oncology Pipeline
Pfizer and Innovent Biologics have entered into a global strategic licensing and collaboration agreement to support the research and development of earlystage cancer medicines The partnership covers antibodydrug conjugates
Insilico Medicine and Human Longevity Partner to Develop AI Foundation Model for Longevity Science
Insilico Medicine and Human Longevity have announced a multimilliondollar collaboration to develop what they describe as the industrys first largescale AI foundation model focused on longevity science
Merz Therapeutics Enters into Agreement with Jiangxi Kvvit Pharmaceutical for INBRIJA® in Parkinson’s Disease
Merz Therapeutics and Jiangxi Kvvit Pharmaceutical have signed an exclusive licence and collaboration agreement for INBRIJA levodopa inhalation powder in mainland China Hong Kong and Macao
AbbVie Receives Positive CHMP Opinion for MAVIRET in Acute Hepatitis C Treatment
AbbVie announced that the Committee for Medicinal Products for Human Use CHMP of the European Medicines Agency has adopted a positive opinion recommending approval of MAVIRET for the treatment of acute hepatitis C virus HCV infection in adults and children aged three years and above
Gilead Sciences Receives Approval for Hepcludex as the Treatment for Chronic HDV
Gilead Sciences has announced that the US Food and Drug Administration FDA has granted accelerated approval to Hepcludex bulevirtidegmod mg for the treatment of adults living with chronic hepatitis delta virus HDV infection